Pre-operative planning of minimally invasive mitral valve surgery using deep-learning
Abstract
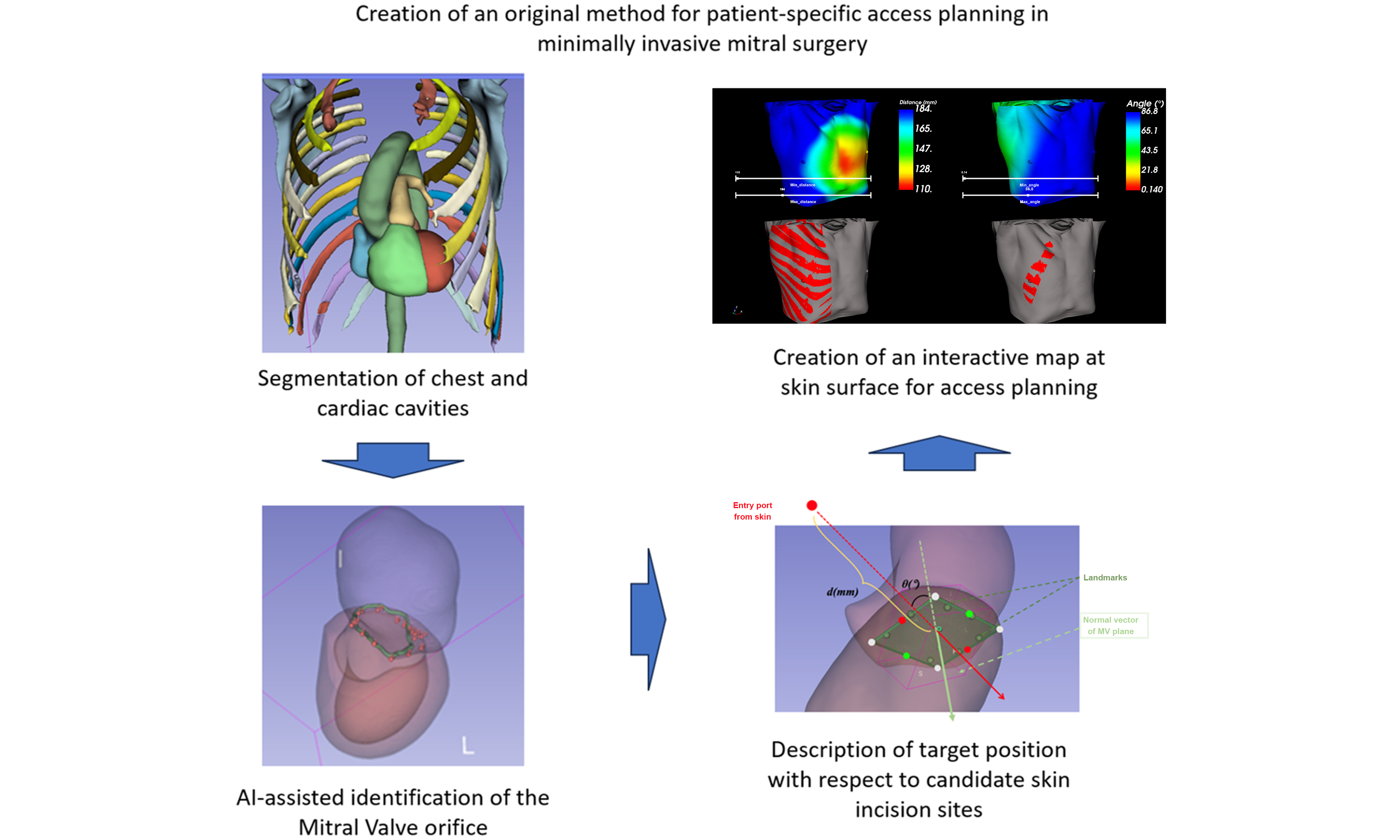
Aim: We propose a novel pre-operative planning approach based on Deep-Learning techniques in the context of minimally invasive mitral valve surgery (MIMVS), for the identification of the mitral valve and optimal thoracic working port positioning in a patient-specific fashion.
Methods: We used supervised Deep-Learning for the processing of contrast-enhanced computed tomography (CT) scans. Our algorithm consisted of four steps: segmentation on CT scans, localization of the mitral valve, creation of maps under three criteria (distance/angle between the mitral valve plane and candidate working port spots, absence/presence of ribs), and selection of optimal working port.
Results: We compared the performance of the Deep-Learning-based approach vs. the previously described semiautomatic method and conventional user’s planning (Dice mean value: 93.59). The Deep-Learning method outperformed the semiautomatic method [intraclass correlation coefficient (ICC) = 0.206]. We defined two interfaces to navigate among candidate working ports.
Conclusion: We suggest that the Deep-Learning-based approach may help the surgeon in identifying the most appropriate working port (thoracic access) for MIMVS, and to comparatively predict the features of different candidate thoracic accesses in individual patients, and help address individual anatomic issues. It may also help standardize the pre-operative planning and obtain a faster learning curve for trainee surgeons.
Keywords
INTRODUCTION
Minimally invasive mitral valve surgery (MIMVS) is widely used as an approach to the atrioventricular valves. It is associated with shorter recovery time, improved post-operative quality of life, lower blood component transfusion, and better cosmetic results. Despite these advantages, the minimally invasive approach presents substantial technical challenges. Limited surgical access, restricted visualization, and patient-specific anatomical variability can complicate the procedure, making accurate pre-operative planning essential for optimizing surgical success. Advances in medical imaging, particularly three-dimensional (3D) echocardiography and computed tomography (CT), provide detailed representations of cardiac anatomy that can support pre-operative evaluation. However, the manual interpretation of these large, complex datasets is time-consuming and subject to inter-observer variability. In this context, computational tools capable of extracting, analyzing, and integrating imaging features into the surgical planning workflow are highly desirable. The choice of the intercostal space for the operative setup and site of working/camera ports is paramount to obtain the optimal exposure of the mitral valve and post-operative results. The operative setup should be planned before the surgery. Chest CT scan is currently performed for generic operative planning, but it has not been formally employed to define the working port. The thoracic CT scan allows description of the valve position, the height of the diaphragm dome, thoracic and aortic morphology, the presence of calcification in the valve, aorta, and iliofemoral arteries, and the distance between the valve and the thoracic cage[1,2].
Deep-Learning is a subset of Machine-Learning which uses multilayered neural network to extract meaningful features and relationships required to make accurate outputs from raw data. It has demonstrated remarkable success in automating image analysis and pattern recognition tasks across diverse medical domains. In cardiac surgery, Deep-Learning methods have been applied to segmentation of cardiac chambers, quantification of valvular disease, and prediction of procedural outcomes. They can quickly locate the mitral valve, and after the valve localization, we described its positioning/distance from the thoracic cage in order to define the optimal working port. Extending these applications to the planning of MIMVS could enhance surgical precision by providing patient-specific anatomical reconstructions, identifying optimal surgical access routes, and predicting potential intraoperative challenges[3].
This preliminary work explores the role of deep learning (DL)-based approaches in the pre-operative planning of MIMVS. The aim of this study is to provide a tool to the cardiac surgeon for the pre-operative planning of MIMVS. This tool may allow greater accuracy in the surgical strategy and approach. The pre-operative planning standardization may simplify patients’ care, help reduce the operative morbidity, and accelerate the surgeons’ learning curve.
METHODS
Cardio-thoracic structures segmentation
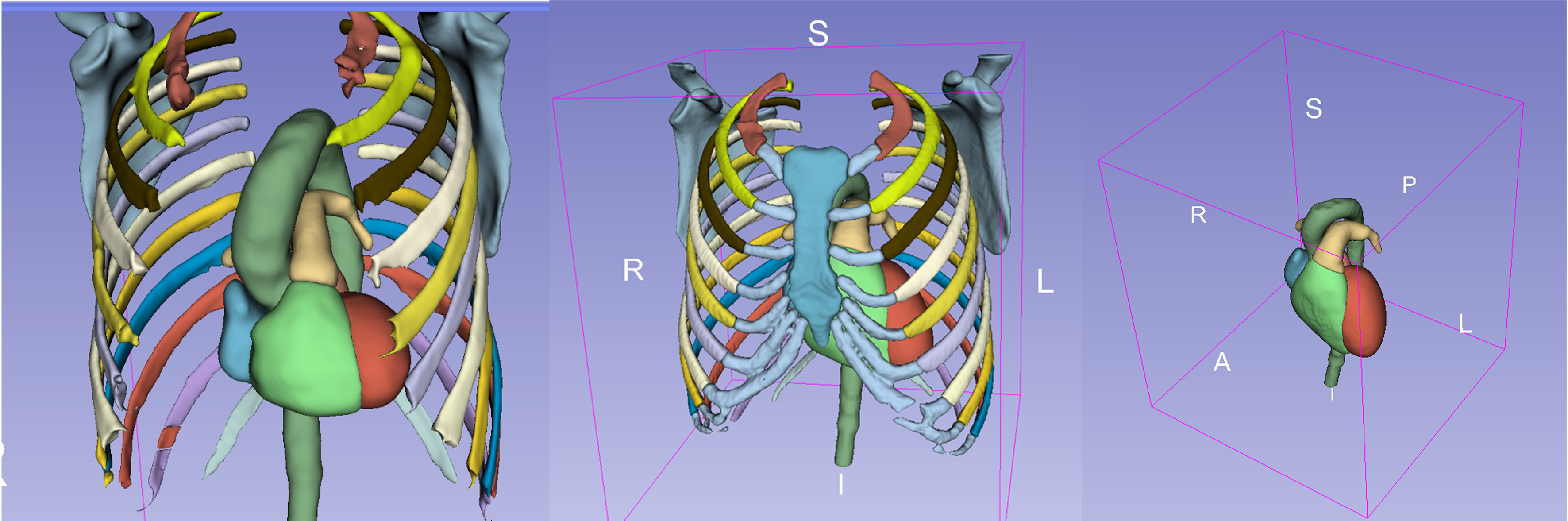
We applied Deep-Learning to train a segmentation network from the public dataset TotalSegmentator[4]. This dataset contained 1,204 CT scans sourced from various institutions, of which 317 were specifically related to the cardiac system. Of these, 201 CT scans were used for training with a convolutional neural network (CNN), 50 for validation, and 66 for testing. Additionally, we simplified the available labels to 7 cardiac system classes (including both ventricles and atria) for subsequent localization of the mitral valve and 15 classes of bone structures (including ribs) to facilitate the planning stage [Figure 1]. The latest medical segmentation network, 3D full-resolution neural network U-net (nnUNet)[5], was used for automatic segmentation. Our model was trained from scratch by the Stochastic Gradient Descent (SGD) optimizer with a batch size of 4 for 1,000 iterations. The initial learning rate was 10-3, subtracting by a factor of 0.1 every 250 iterations. All experiments were implemented in PyTorch and run on NVIDIA RTX A6000 Graphics Processing Units (GPUs).
Figure 1. All semantic classes used in CNN. CNN: Convolutional neural network; S: superior; R: right; A: anterior; I: inferior; L: left; P: posterior.
Image reconstruction was performed using the open-source 3D Slicer software[6].
Mitral valve localization
For the purposes of the present work, we employed three methods for localizing the mitral valve on CT scans. Comparing these methods was necessary to achieve more precise localization of the mitral valve, which has been the most challenging part of our work. The “manual” method consisted of identifying the mitral orifice on multiplane CT images by an experienced surgeon using fiducial points (landmarks). This allowed a representation of the area of the mitral orifice, the extraction of its center and of the average plane passing through the landmarks. This method was considered the reference gold standard.
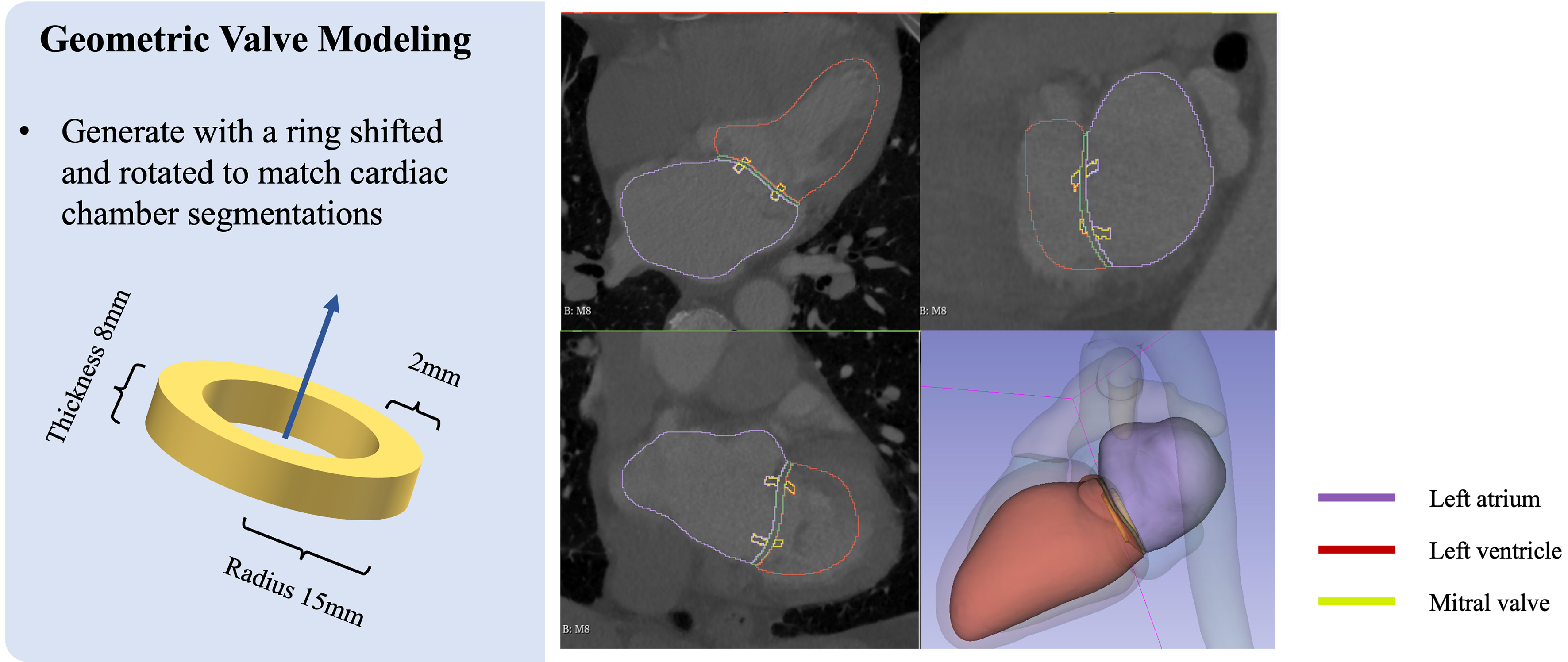
The second method (“Ring-shaped”) was based on the work of Finnegan et al., which we consider the best among previously published methods[7]. It involved a geometric representation of the mitral valve by generating a ring-shaped model, corresponding to the intersection between the left ventricle and atrium. The procedure was as follows: first, the overlapping region between the left atrium and left ventricle was identified through a dilation operation applied to the automatically segmented heart chambers (to ensure a sufficient area of overlap for subsequent analyses). Next, the center of the geometric model was determined by calculating the center of the overlap area, and the orientation was defined from the center of the left atrium to the center of the left ventricle, aligned with the direction of blood flow. Finally, the ring-shaped model representing the mitral valve was defined using a constant thickness and diameter; we chose an 8 mm thickness and a 30 mm ring diameter [Figure 2].
Figure 2. Visualization of the geometric mitral valve modeled as a patient-adapted ring. The purple region represents the left atrium, the red represents the left ventricle, and the yellow represents the mitral valve.
The third method used a 2D landmark localization network based on an attention U-net to detect the edgepoints of valve leaflets surrounding the mitral annulus[8]. By extracting 2D slices around the valve from CT scans, the model captured global structures and local textures to automatically detect the mitral valve annulus via interpolation.
To validate a segmentation model, we needed at least one model for comparison and testing. We therefore validated the model by comparing it to two others. The main difference between the Ring-shaped model and the Attention U-net model lies in their segmentation approach. In the Ring-shaped model, we localized the mitral valve using the zone of apposition between the left atrium and left ventricle, whereas in the Attention U-net model, we localized the mitral valve based on the borders of the mitral leaflet and its insertion into the mitral annulus. The main limitation of the Ring-shaped model is that it does not account for the saddle shape of the mitral valve.
We retrospectively applied the three methods to a local dataset comprising 28 contrast-enhanced CT scans from patients who had already undergone MIMVS in the cardiac surgery unit of Pontchaillou University Hospital in Rennes, France.
Method comparison
We computed the Angle (°) metric as the angular difference between the mitral valve plane obtained by the “gold-standard” manual delineation method and that obtained by either of the two other methods (“Ring-shaped” and “Attention U-net”).
All recipients of cardiac surgery provide written informed consent for the use of the anonymized personal data for research purposes. Additionally, all patients’ data are extracted from a prospective database declared to the CNIL (Commission Nationale de l’Informatique et des Libertés - National Committee for Informatics and Freedom) under the number 1207754.
Statistical analysis
We described the Distance (mm) metric as the minimal distance from each landmark to either of the other two methods. The results obtained were described as mean ± standard deviation (SD); intergroup comparison was performed using the two-tailed Student’s t test for paired data. The interobserver agreement was evaluated through calculation of the intraclass correlation coefficient. The alpha level was set at 0.05.
The working port
In a subsequent step, we developed a preliminary planning solution for surgical access (“working port”). Following the identification of the mitral valve based on the “Attention U-net” method, the solution integrated the following three criteria, which we considered the main ones for achieving optimal surgical exposure of the mitral valve: the distance between the valve and the skin, the viewing angle, and the absence of ribs and sternum at candidate skin incision sites. We generated a map of the shortest distance between the valve and each candidate skin incision site, and a map of the perpendicular viewing angle to the valve plane, while avoiding the ribs and sternum.
RESULTS
Evaluation based on the Dice coefficient on the public dataset has demonstrated that the 3D full-resolution nnUNet can accurately delineate the targeted cardiac system and rib structures [Table 1]. This capability confirms the potential applicability of our model to local datasets for further image processing in valve localization and working port selection.
Results of 3D full-resolution nnUNet segmentation network
| 3D full-resolution nnUNet segmentation results (DSC, %) | ||||||||||||
| Aorta | PA | LA | RA | Myo | LV | RV | Ribs | Scapula | Sternum | Costal cartilages | Mean | |
| nnUNet | 94.34 | 92.06 | 96.04 | 94.32 | 90.17 | 94.23 | 94.01 | 93.59 | 96.53 | 95.71 | 92.06 | 93.75 |
| 1st R | 2nd R | 3rd R | 4th R | 5th R | 6th R | 7th R | 8th R | 9th R | 10th R | 11th R | 12th R | |
| nnUNet | 94.99 | 96.17 | 95.57 | 96.39 | 95.44 | 94.82 | 93.89 | 92.97 | 92.91 | 92.54 | 90.46 | 87.00 |
Valve localization
Table 2 displays the results of comparison between each mitral valve identification method and the “gold standard” method. Difference in angle and distance between each method and the “gold standard” was considered to describe better localization of the mitral valve when closer to zero. The Attention U-net method outperformed the Ring-shaped method for both Angle and Distance parameters, the statistical significance being attained for the Distance parameter (P < 0.001). The inter-rater agreement analysis indicated poor agreement between the two methods (coefficient values of 0.206 and 0.324), further supporting the better performance of the Attention U-net approach.
Comparison of ring-shaped and attention U-net methods’ performance in the description of the mitral valve
| Variable | Ring-shaped | Attention U-net | P value |
| Angular difference vs. the gold standard | 16.3° ± 11.5° | 11.5° ± 9.4° | 0.1 |
| Distance vs. the gold standard | 5.5 ± 2.7 mm | 3.1 ± 1.3 mm | < 0.01 |
| Inter-rater agreement (Angle) | ICC = 0.206 | 0.15 | |
| Inter-rater agreement (Distance) | ICC = 0.324 | 0.49 | |
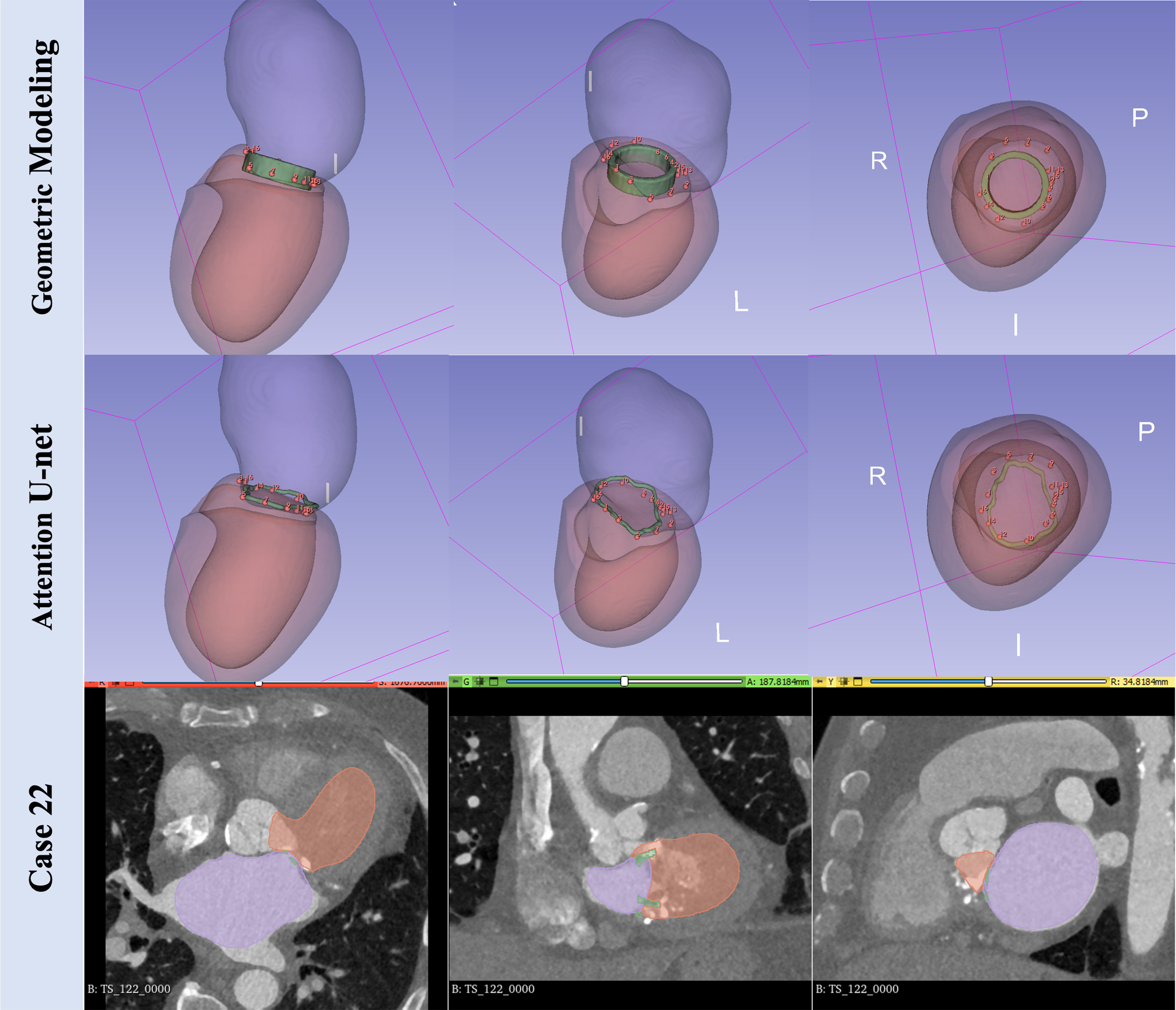
We display the visualization of the results of the geometric mitral valve on local cardiac CT cases in Figure 3. The shape, position, and orientation of the ring-shaped model are close to manual landmarks marked by the cardiac surgeon.
Planning solution: the working port
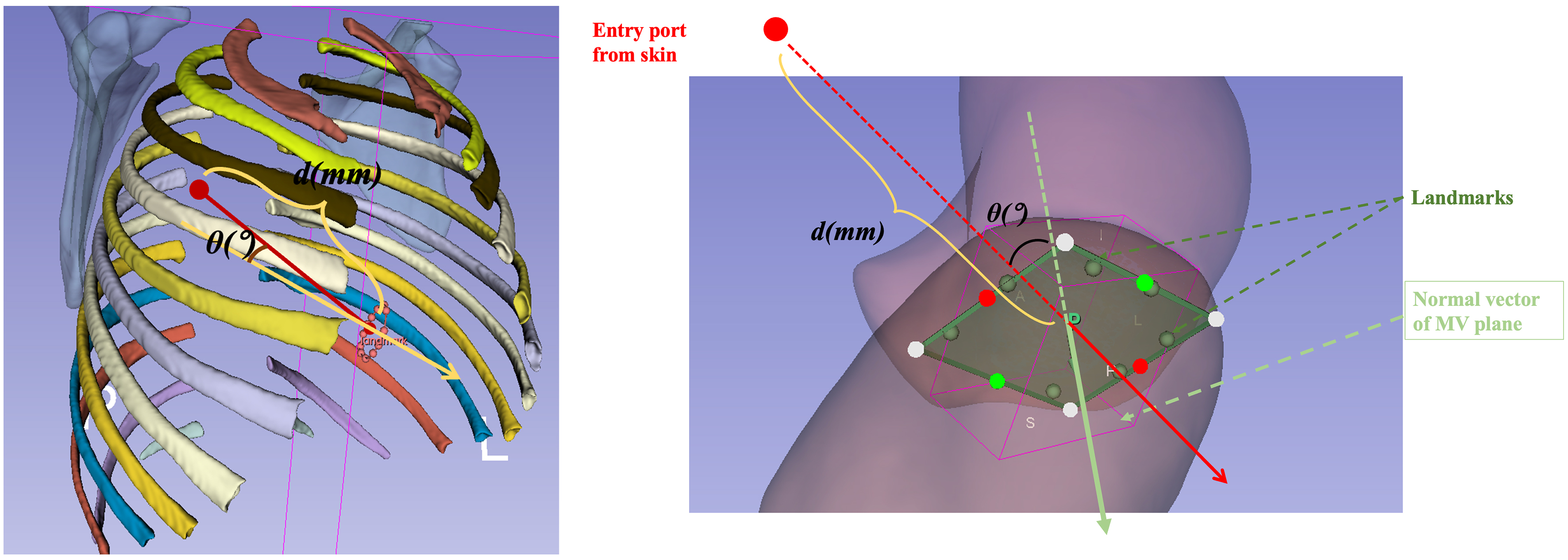
Based on the above findings, we developed a preliminary planning solution for mitral valve identification using the Attention U-net method. Three criteria were evaluated to identify the optimal working port: the distance and viewing angle between the mitral valve plane and the candidate incision site. The absence of ribs and sternum at the candidate incision site was the third criterion. d (mm) denotes the distance from the candidate working port and the center of the mitral valve, θ (°) describes the angle between the normal vector at the center of the incision and the normal vector of the mitral valve plane. The presence or absence of the ribs and sternum was treated as a binary parameter. The shortest distance between the valve and the skin at the incision, an angle as close as possible to 0, and the absence of ribs and sternum are the requirements to describe the appropriateness of the working port [Figure 4].
Planning solution: integration of parameters
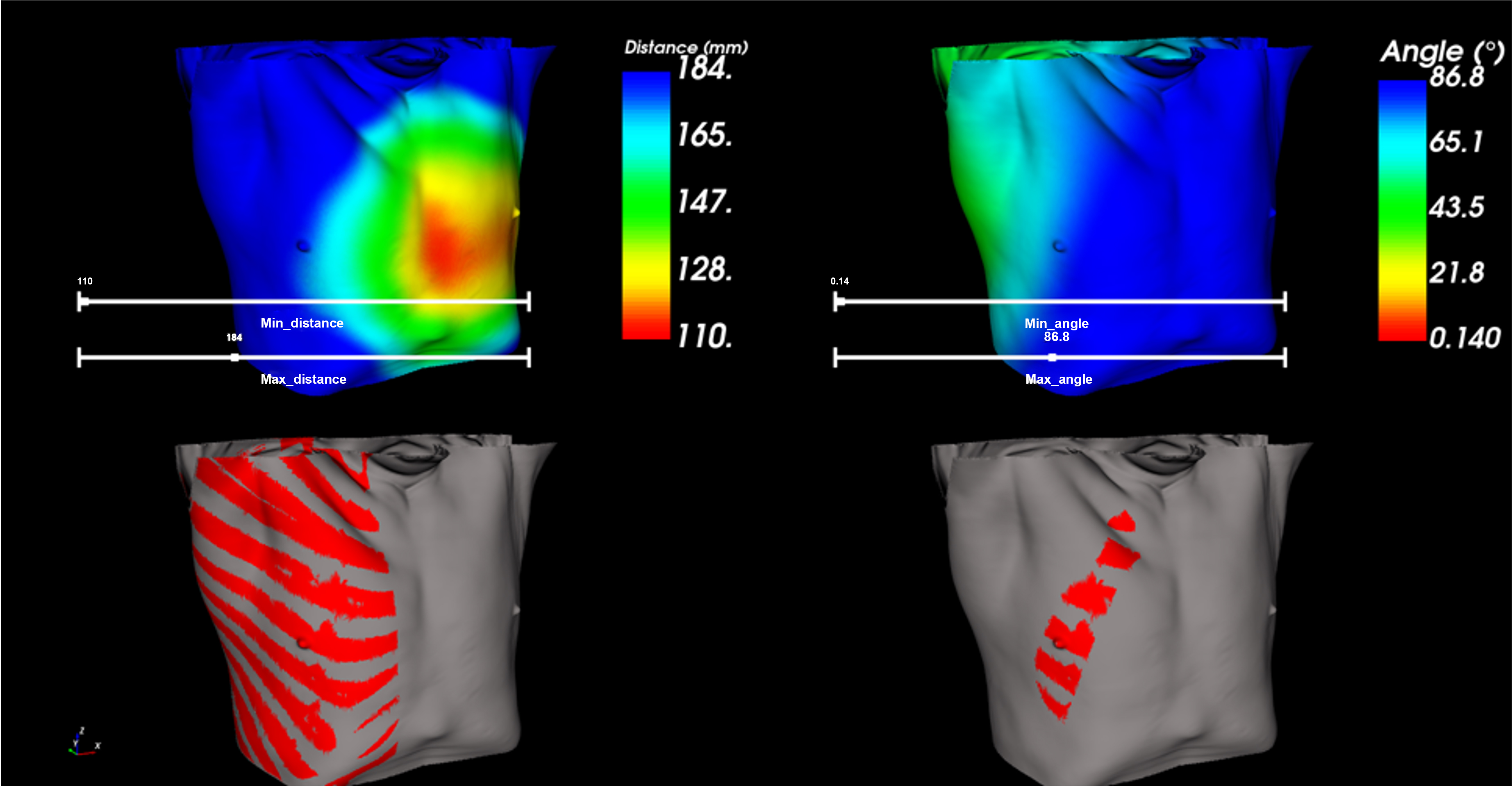
The skin area was delineated from CT scan using 3D seed point growing algorithm. Subsequently, all available points on the patient’s skin were described based on the aforementioned criteria (distance and angle with respect to the mitral valve, absence of ribs). The first representation [Figure 5] displays the distance from the skin to the mitral valve plane using a color map, the shortest possible distance being considered as optimal and being highlighted in red. The second representation illustrates the angle between any candidate incision site vector and the normal vector of the mitral valve plane, the ideal being 0 degrees, represented using a color map. The third binary representation displays the potential areas, marked in red, that avoid collision with ribs and the sternum.
Figure 5. Visualization results of optimal working ports in case No.26 by fusing the overlapping region satisfying the criteria for distance, angle, and non-collision (created using C++-based VTK code).
We introduce two approaches and their respective visualization outcomes to integrate the criteria in the identification of optimal working ports at the skin surface.
The first method, illustrated in Figure 5, employs interactive sliders for adjusting distance and angle measurements. The fusion approach ensures precise delineation of the targeted region, with the resultant combination being the intersection of areas satisfying the criteria for distance, angle, and non-collision.
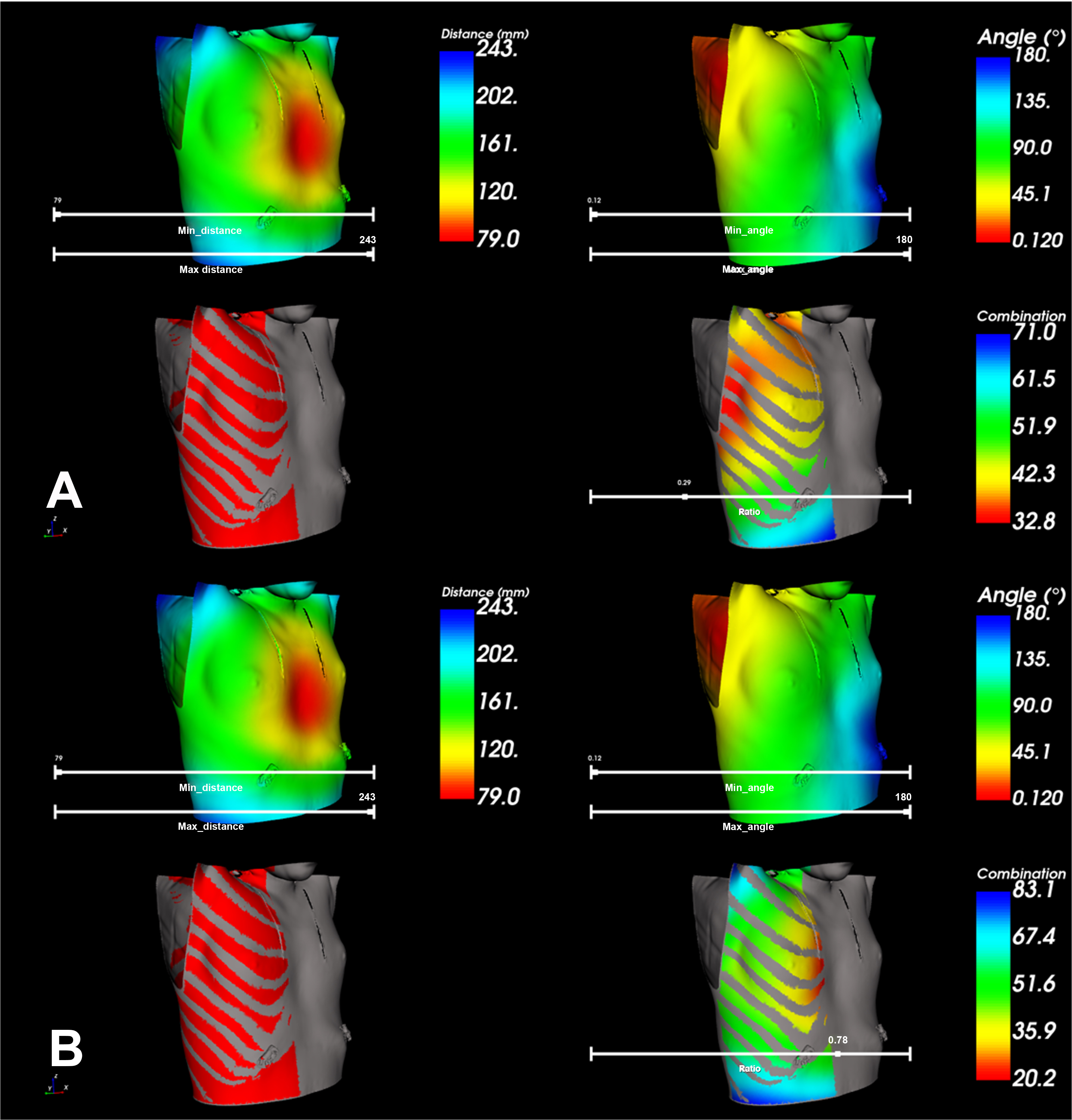
In the second approach, variable weighting is applied to balance the significance of the two main criteria (distance and angle from the working port to the mitral valve). The requested conditions for these metrics are set to be as small as possible. Therefore, these data are first normalized to a scale from 0 to 100 to standardize the input values. Following this normalization, a composite score is calculated using weighted contributions from distance and angular measurements, where
An interactive slider allows for manual adjustment of the weighting ratio, emphasizing the relative importance of distance over angle, or vice versa, according to the user’s preference. The optimal working ports with the smallest score, determined through this weighted fusion method, are shown in Figure 6.
Figure 6. Visualization of the optimal working port in case No. 27 by fusing weighted contributions from distance and angle. (A) The result with a ratio of 0.29, emphasizing the importance of angle; (B) The result with a ratio of 0.78, emphasizing the importance of distance (created using C++-based VTK code).
DISCUSSION
The diffusion of minimally invasive approaches is part of the current evolution of cardiac surgery. MIMVS is associated with shorter hospital stays, shorter recovery times, and cosmetic advantages, but it is also associated with longer operative times. Dedicated exposure techniques are required, including endoscopic strategies. The quality of exposure of the mitral valve in the MIMVS settings depends on several patient-specific factors and may influence the operative outcomes. We therefore aimed to develop a new tool for pre-operative planning in MIMVS. We hypothesized that artificial intelligence techniques may be helpful for such a purpose, similar to other medical fields. Machine-Learning, and, as its subset, the Deep-Learning are contributing to reshape patients’ care and management[3,9,10].
The present work highlights the potential of DL-based methods to enhance pre-operative planning in MIMVS. By leveraging advanced image analysis techniques, DL can extract meaningful anatomical information from complex datasets, providing surgeons with patient-specific insights that go beyond the capabilities of conventional imaging interpretation. Our model achieved an accurate segmentation of the mitral valve leaflets and surrounding anatomical structures, outperforming conventional image-processing methods and approaching the reliability of expert manual annotations. These results confirm the feasibility of integrating DL-driven analysis into the surgical planning workflow, providing surgeons with accurate, reproducible, and patient-specific anatomical reconstructions. This represents a significant step toward precision cardiac surgery, where pre-operative strategies can be tailored to the unique anatomical and pathological features of each patient. In the current pre-clinical work, we have no direct comparison with real-life results. Before using this tool in clinical applications, a robust development from a geometric point of view was required. The three main parameters chosen (angle, distance and presence/absence of ribs) are surrogate markers of the surgical exposure. We expect that the main benefits from this tool would come for patients who are obese or have chest deformities, for female patients (planning of thoracic access beneath the breast), and for surgeons who are in their learning curves. Our findings align with prior research demonstrating the value of DL in cardiac imaging and surgical planning. In particular, the ability of CNN and transformer-based architectures to perform robust segmentation of cardiac structures offers a promising foundation for generating accurate 3D reconstructions of the mitral valve and surrounding anatomy. Such reconstructions may facilitate the identification of optimal surgical access routes, reduce intraoperative uncertainty, and potentially shorten operative times. Importantly, automated feature extraction may also decrease inter-observer variability, thereby standardizing pre-operative assessments across surgical teams. Our results also highlight the potential role of DL in surgical decision-making. DL may complement the surgeon’s expertise by uncovering subtle imaging cues that influence operative strategy. Moreover, the consistency of the model’s predictions across repeated analyses reduces inter-observer variability, a long-standing limitation in pre-operative imaging interpretation.
This tool may help the surgeon obtain a patient-specific plan for the identification of the optimal working port to achieve appropriate access and exposure to the mitral valve. We took into account some variables: height, obesity, age, chest dimensions and the distance between the mitral valve and the thorax. In fact, some patients have a particular thoracic morphology contraindicating MIMVS. Jung et al. underlined that some physical characteristics are associated with suboptimal exposure of the mitral valve, especially patients with greater thoracic antero-posterior and latero-lateral diameters, patients with a high-situated diaphragm dome, and patients with body mass index (BMI) > 28,5[2].
To the best of our knowledge, this is the first work using Deep-Learning techniques on CT scan dataset for the planning of MIMVS through identification of the target mitral valve. In a systematic review over DL in cardiac surgery, Miles et al. underlined that the appropriateness and dimensions of the dataset are paramount, such as the clear definition of the analyzing method[11]. The Machine-Learning has been used mainly in medical imaging, biomedicine and elaboration of scores for prediction of mortality. There is not a single model that can be universally adapted to all purposes; therefore, we used one that we identified as the most appropriate for our specific purpose. The Deep-Learning has some advantages in this perspective; it simplifies and standardizes the imaging analysis for pre-operative planning. Despite these advantages, several challenges must be addressed before routine clinical adoption can be achieved. First, the generalizability of DL models is heavily dependent on the quality and diversity of training datasets. Many existing datasets are limited in size and may not adequately capture the variability in patient anatomy, imaging protocols, and disease presentations. Multi-institutional collaborations and the development of large-scale annotated datasets will be critical to overcoming this limitation. Second, the integration of DL tools into clinical workflows requires careful consideration of usability, interpretability, and regulatory compliance.
Our Deep-Learning model is supervised. It was trained to segment the anatomical structures from the public dataset Totalsegmentator. We then focused on the pre-operative planning of the working port for MIMVS. The proposed DL approach was more effective than the previously described geometrical method in identifying the target mitral valve. This step was then incorporated into a preliminary planning solution, which included a map of distances and angular views between candidate incision sites at the skin and the target mitral valve. Such parameters define the quality of the surgical access to the mitral valve in MIMVS. Two integration approaches of these parameters are presented [Figures 5 and 6], allowing for user-driven regulation of their respective weight in the planning of skin incision, according to surgical strategy in individual cases and the surgeon’s needs. In a further step, we foresee performing a quantitative evaluation of each approach in a series of clinical cases.
The quality of mitral valve exposure is paramount to shorten the duration of surgery and reduce intraoperative bleeding. It helps to reduce intra- and post-operative blood component transfusion and to reduce the post-operative inflammatory burden due to extracorporeal circulation. The use of contrast-enhanced CT scans is useful in order to understand the morphology of the chest, the distance between the chest and the mitral valve and the feasibility of the minimally invasive approach.
The main limitations of this study are: the low number of CT scans analyzed and the difficulty in identifying the precise mitral valve localization. Only one cardiac surgeon had worked on the valve localization. This approach was used as considered sufficient to validate the deep-Learning model, since the variability in localization of the valve annulus from one user to another is likely negligible for this research purpose. Valve localization by different surgeons would help to improve the quality of the work and limit the bias of a single surgeon. A multi-expert evaluation may give a better reproducibility for the model. The CT scans were acquired with the patients being in different positions (with arms either above or below the head). The model’s performance decreased in cases with severe imaging artifacts or rare anatomical variants, underscoring the need for larger and more diverse training datasets. The quality of the CT scan plays a role; electrocardiogram (ECG)-gated acquisitions may improve the results.
This preliminary and pre-clinical model represents the first step towards the simplification of the MIMVS. Nonetheless, the construction of this assistance tool required an original development process involving a multidisciplinary team. The next step will involve clinical testing to evaluate the possible improvements in intraoperative exposure and shortening of the cross-clamp time or of the learning curve. It is already a step forward compared with our previous research on this subject[12], as we evaluated chest dimensions, used a deep-learning approach for image analysis, and compared three different methods to improve the accuracy of our findings. While the present study underscores the promise of DL, it also emphasizes the necessity of prospective validation in real-world clinical settings. Rigorous clinical trials and longitudinal studies will be essential to confirm whether DL-enhanced planning translates into measurable improvements in surgical efficiency, safety, and patient outcomes.
From a future perspective, we plan to obtain a description of other parameters from segmentation, such as the valve perimeter or the anteroposterior diameter. This information is important for the endovascular and robotic approaches for the treatment of mitral valve disease[13].
Our experimental results support the integration of DL into the pre-operative planning of MIMVS. DL represents a transformative opportunity to refine the pre-operative planning of MIMVS. Although technical, clinical, and regulatory challenges remain, the integration of DL into surgical workflows has the potential to standardize decision-making, optimize patient-specific strategies, and ultimately improve the quality of care in mitral valve surgery.
DECLARATIONS
Authors’ contributions
Writing-original draft and conceptualization: Evangelista G
Data curation and formal analysis: Lyu S
Project administration and validation: Simon A
Conceptualization: Castro M, Verhoye JP, Shu H
Conceptualization and methodology: Haigron P
Conceptualization, supervision, writing-review and editing: Anselmi A
Availability of data and materials
The original contributions presented in this study are included in the article. Additional data are available from the corresponding author upon reasonable request. Totalsegmentator is a public dataset that we used to train our Deep-learning model. You can find the dataset following this link: https://zenodo.org/records/10047292.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This study was partially supported by the French National Research Agency (ANR) within the framework of the Investissements d’Avenir Program through Labex CAMI (ANR-11-LABX-0004). This work was also supported in part by the National Key Research and Development Program of China under Grant No. 2022YFE0116700. Lyu S acknowledges support from the China Scholarship Council (CSC Grant No. 202106090281).
Conflicts of interest
Anselmi A is a member of the Junior Editorial Board of Mini-invasive Surgery. Anselmi A was not involved in any part of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision-making. The other authors declare that they have no conflicts of interest.
Ethical approval and consent to participate
This study did not require additional ethics approval. All patient data were extracted from a prospective database registered with the CNIL (Commission Nationale de l’Informatique et des Libertés - National Committee for Informatics and Freedom) under registration number 1207754. All patients undergoing cardiac surgery provided written informed consent for the use of anonymized personal data for research purposes.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Ito T. Minimally invasive mitral valve surgery through right mini-thoracotomy: recommendations for good exposure, stable cardiopulmonary bypass, and secure myocardial protection. Gen Thorac Cardiovasc Surg. 2015;63:371-8.
2. Jung Y, van Kuijk SMJ, Gietema H, Maessen JG, Sardari Nia P. Prediction of poor exposure in endoscopic mitral valve surgery using computed tomography. Eur J Cardiothorac Surg. 2024;65:ezae070.
3. Litjens G, Ciompi F, Wolterink JM, et al. State-of-the-art deep learning in cardiovascular image analysis. JACC Cardiovasc Imaging. 2019;12:1549-65.
4. Wasserthal J, Breit HC, Meyer MT, et al. Totalsegmentator: robust segmentation of 104 anatomic structures in CT images. Radiol Artif Intell. 2023;5:e230024.
5. Isensee F, Jaeger PF, Kohl SAA, Petersen J, Maier-Hein KH. nnU-Net: a self-configuring method for deep learning-based biomedical image segmentation. Nat Methods. 2021;18:203-11.
6. Fedorov A, Beichel R, Kalpathy-Cramer J, et al. 3D Slicer as an image computing platform for the quantitative imaging network. Magn Reson Imaging. 2012;30:1323-41.
7. Finnegan RN, Chin V, Chlap P, et al. Open-source, fully-automated hybrid cardiac substructure segmentation: development and optimisation. Phys Eng Sci Med. 2023;46:377-93.
8. Oktay O, Schlemper J, Folgoc LL, et al. Attention u-net: learning where to look for the pancreas. J Digit Imaging. 2018. Available from: https://www.semanticscholar.org/paper/Attention-U-Net%3A-Learning-Where-to-Look-for-the-Oktay-Schlemper/ae1c89817a3a239e5344293138bdd80293983460. [Last accessed on 23 Mar 2025].
9. Tao Q, Yan W, Wang Y, et al. Deep learning-based method for fully automatic quantification of left ventricle function from cine MR images: a multivendor, multicenter study. Radiology. 2019;290:81-8.
10. Heuts S, Maessen JG, Sardari Nia P. Preoperative planning of left-sided valve surgery with 3D computed tomography reconstruction models: sternotomy or a minimally invasive approach? Interact Cardiovasc Thorac Surg. 2016;22:587-93.
11. Miles TJ, Ghanta RK. Machine learning in cardiac surgery: a narrative review. J Thorac Dis. 2024;16:2644-53.
12. Di Perna D, Castro M, Gasc Y, Haigron P, Verhoye JP, Anselmi A. Patient-specific access planning in minimally invasive mitral valve surgery. Med Hypotheses. 2020;136:109475.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.