Porous aromatic frameworks by highly connected building blocks for hydrogen and methane storage
Abstract
The fabrication of porous materials possessing ultrahigh specific surface areas remains a significant challenge. We report the synthesis of two novel porous aromatic frameworks, PAF-336 and PAF-337, constructed from 6- and 8-connected building blocks with triangular prismatic and cuboid geometries, respectively. PAF-336 demonstrates an ultrahigh specific surface area (~5,210 m2·g-1) and large pore volume (3.5 cm3·g-1). This high porosity translates to high hydrogen storage capacity and state-of-the-art methane storage performance, positioning PAF-336 as a potential material for clean energy storage.
Keywords
INTRODUCTION
Carbon dioxide (CO2) emissions are the main contributor to the critical rise in global temperatures[1-6], primarily caused by the burning of fossil fuels, such as coal and gasoline[7]. Hydrogen (H2), when used as an energy carrier, produces no CO2 emissions upon combustion, whereas methane (CH4) emits significantly less CO2 than traditional fossil fuels, thereby potentially mitigating the greenhouse effect[8,9]. The main challenge limiting the use of H2 or CH4 as energy lies in the storage methods. Currently, the predominant techniques for storing combustible gases involve liquefaction and compression, both of which require costly storage systems and high pressures (typically 250 bar for CH4 and 700 bar for H2)[10-12]. In contrast to alternative approaches, porous materials with high specific surface areas and designable topological structure show exceptional promise for H2 and CH4 storage[3,13-19].
The pursuit of ultra-high porosity has always been the primary challenge in the design and synthesis of porous materials[20]. Frameworks connected by robust covalent bonds[21-27] are stable and have been demonstrated to be suitable for H2 and CH4 storage. Nevertheless, the covalently connected frameworks with ultra-high specific surface area are still quite rare. Notably, diamond structure mimetic porous aromatic framework PAF-1 achieves an ultrahigh Brunauer-Emmett-Teller (BET) surface area (5,600 m2·g-1) with extraordinary chemical-thermal stability[28]. Subsequent frameworks with ultra-high porosity using tetrahedron-related building blocks were also reported[29-33]. It is well known that the pore size, pore shape and pore environment are related to the H2 and CH4 storage performance[10,34,35]. Therefore, developing topology-guided novel building blocks is an effective way to synthesize porous materials with ultrahigh porosity, which will provide new insights for gas storage.
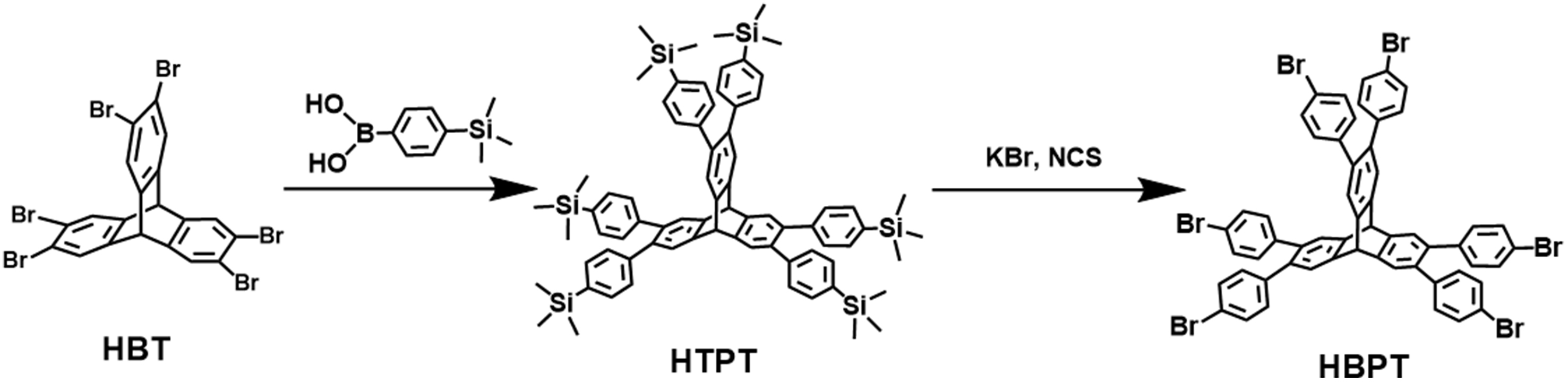
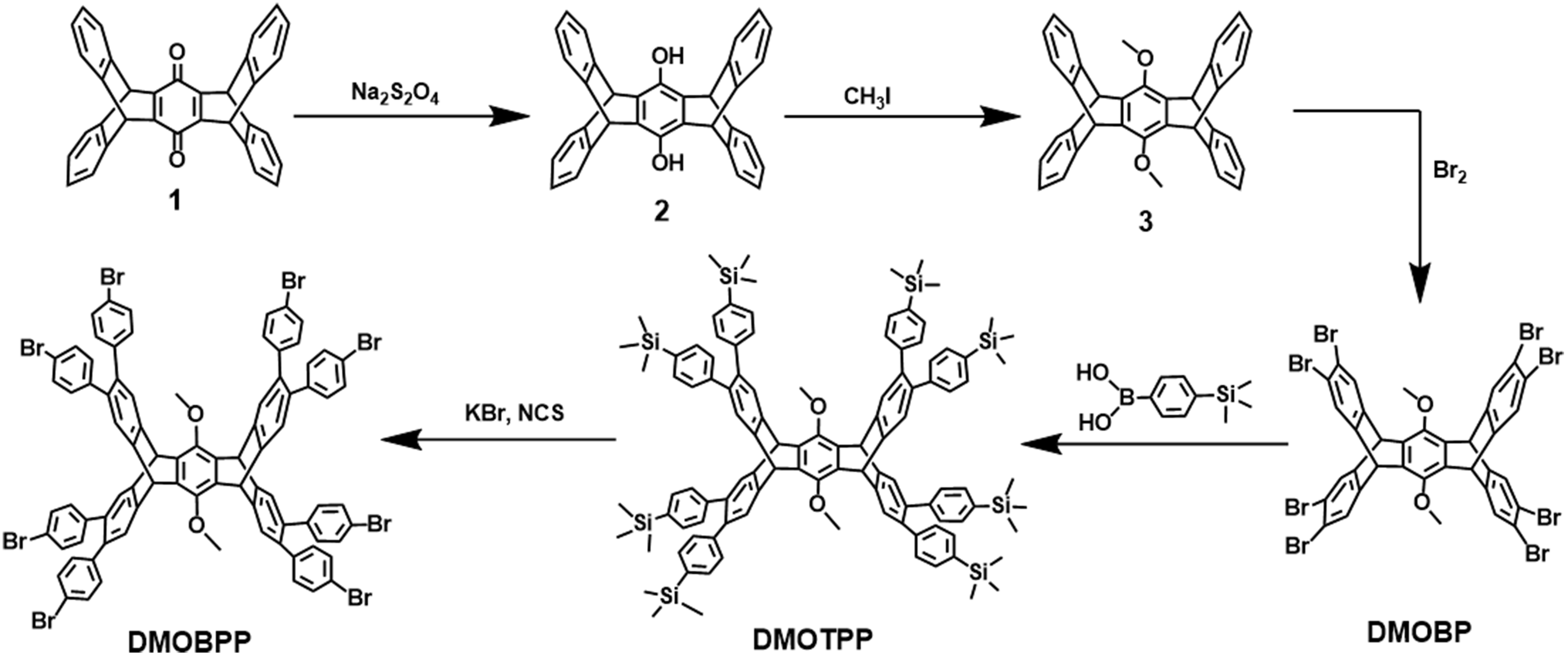
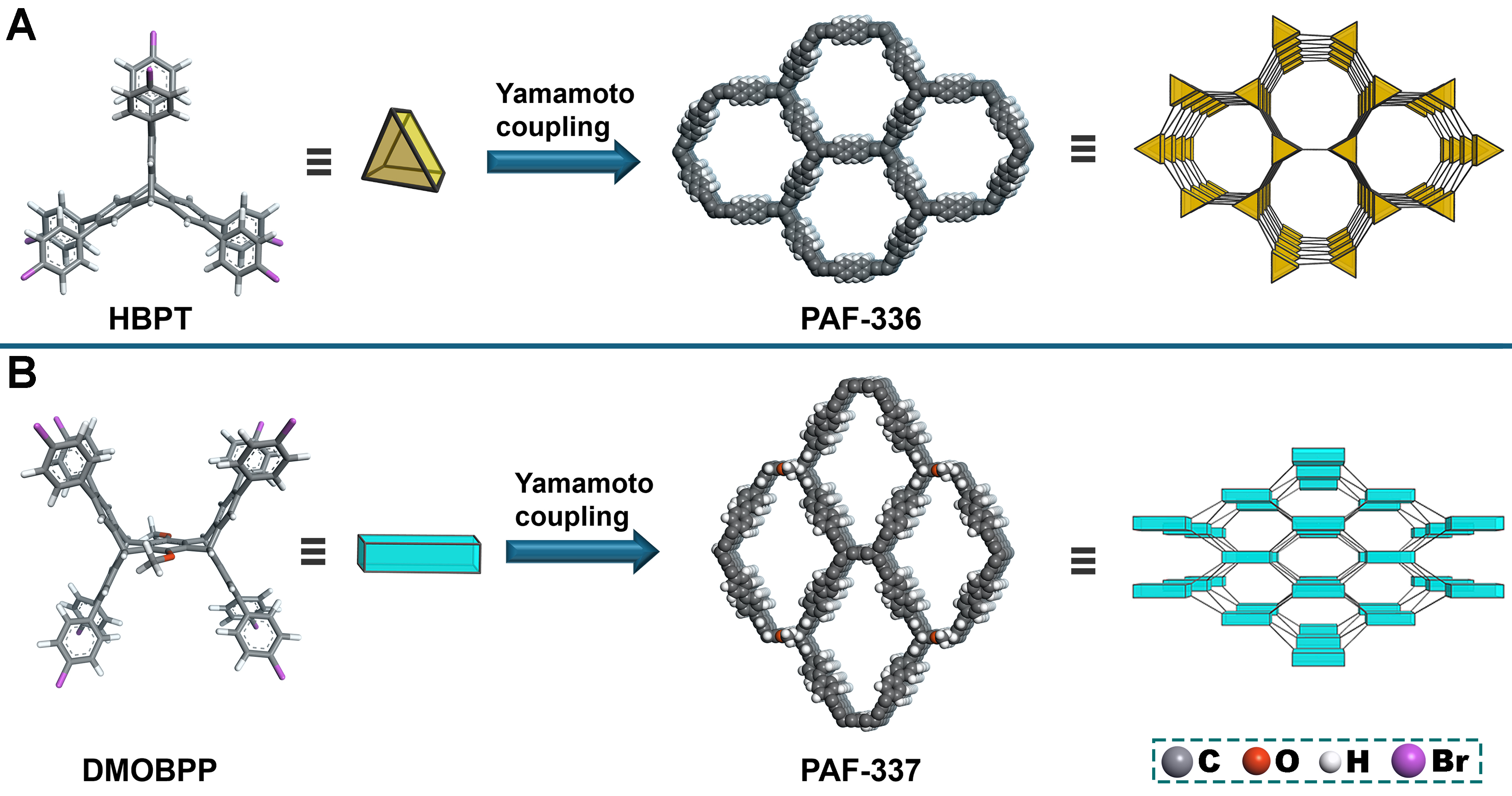
The acs [(3,6)-connected net] and bcu (8-connected body-centered cubic net) topologies, constructed from highly connected trigonal prismatic and cuboidal building blocks, are both edge-transitive topologies widely used in the design of metal-organic frameworks (MOFs) and covalent organic frameworks (COFs)[36-45]. Herein, we synthesized PAF-336 and PAF-337 targeting acs and bcu topologies using unprecedented HBPT [2,3,6,7,14,15-hexa(4′-bromophenyl)triptycene] and DMOBPP [6,13-dimethoxy-2,3,9,10,18,19,24,25-octa(4′-bromophenyl)pentiptycene] as monomers through Yamamoto-type coupling reaction [Schemes 1 and 2]. To be noted, PAF-336 presents an extremely high BET surface area of ~5,210 m2·g-1 and a large pore volume of
Scheme 1. Synthesis route of HBPT. HBPT: 2,3,6,7,14,15-Hexa(4′-bromophenyl)triptycene; HBT: 2,3,6,7,14,15-hexabromotriptycene; HTPT: 2,3,6,7,14,15-hexa(4′-(trimethylsilyl)phenyl)triptycene; NCS: N-chlorosuccinimide.
EXPERIMENTAL
Materials
The chemicals used in this study include: 2,3,6,7,14,15-hexabromotriptycene (HBT, Alfa, > 97%), (4-(trimethylsilyl)phenyl)boronic acid (Macklin, 98%), N-chlorosuccinimide (NCS, Energy Chemical, 98%), tris(dibenzylideneacetone)dipalladium(0) [Pd2(dba)3, Energy Chemical, 98%], tri-tert-butylphosphine tetrafluoroborate [HP(tBu)3BF4, Energy Chemical, 98%], sodium dithionite [Na2S2O4, Energy Chemical, analytical reagent (AR) grade, ≥ 88.0%], tetra-chloro-1,4-benzoquinone (Energy Chemical, 97%), anthracene (Innochem, 99%), iodomethane (Innochem, 99%), 18-Crown-6 (Macklin, 99%), p-benzoquinone (Macklin, 99%), 1,5-cyclooctadiene (COD, Innochem, 98%), bis(1,5-cyclooctadiene)nickel(0) [Ni(COD)2, Laajoo, 97%], 2,2′-bipyridyl (Aldrich, 99%), N,N-dimethylformamide [DMF, Macklin, 99.8%, with molecular sieves, Water ≤ 50 parts per million (ppm)], and tetrahydrofuran (THF, Macklin, 99.5%, with molecular sieves, Water
Synthesis method
Synthesis of HBPT
Synthesis of 2,3,6,7,14,15-hexa(4′-(trimethylsilyl)phenyl)triptycene (HTPT)
A combination of HBT (250 mg, 0.34 mmol) and (4-(trimethylsilyl)phenyl)boronic acid (1.00 g, 5.2 mmol) was added to a mixed solvent of anhydrous THF (7 mL) and degassed 1 M aqueous (aq.) K2CO3-solution
Synthesis of HBPT
A mixture of HTPT (200 mg, 0.18 mmol) and potassium bromide (KBr, 226 mg, 1.90 mmol) was dissolved in a mixed solvent of CH3COOH (30 mL) and CH3OH (4 mL). The solution was heated at 60 °C for
Synthesis of DMOBPP
Synthesis of Compound 1
A mixture of anthracene (3.02 g, 17 mmol), p-benzoquinone (919 mg, 8.5 mmol) and tetra-chloro-1,4-benzoquinone (4.18 g, 17 mmol) was dissolved in glacial acetic acid (AcOH, 250 mL) and refluxed at 120 °C with magnetic stirring at 500 rpm for 24 h. After cooling to room temperature, the resulting precipitate was collected by filtration. The residue was washed several times with diethyl ether and dried under reduced pressure at room temperature to afford Compound 1 as a yellow solid (3.5 g, 90% yield). The structure was confirmed using 1H NMR
Synthesis of Compound 2
Compound 1 (414 mg, 0.90 mmol) and sodium bicarbonate (1.00 g, 12.0 mmol) were dissolved in anhydrous DMF (9 mL), followed by the addition of Na2S2O4 (1.00 g, 6 mmol). The mixture was stirred at 500 rpm and 100 °C for
Synthesis of Compound 3
A mixture of Compound 2 (790 mg, 1.70 mmol), freshly dehydrated K2CO3 (1.20 g, 8.70 mmol) and 18-crown-6 (5 mg) was suspended in anhydrous acetone (75 mL), followed by the addition of iodomethane (1.1 mL, 18 mmol). The mixture was stirred at 500 rpm and 70 °C for 14 h and then concentrated under reduced pressure using a rotary evaporator. The resulting residue was dissolved in chloroform (CHCl3) and washed with saturated brine. The organic layer was dried over anhydrous MgSO4, filtered, and concentrated under vacuum. The crude material was purified by column chromatography (gradient elution: 25% to 50% CH2Cl2 in hexanes) and dried under vacuum at 60 °C for 24 h to afford Compound 3 as a white powder
Synthesis of 6,13-dimethoxy-2,3,9,10,18,19,24,25-octabromopentiptycene (DMOBP)
Compound 3 (490 mg, 1 mmol) and a catalytic amount of iron filings (50 mg) were added in anhydrous CH2Cl2 (56 mL) in a flame-dried 100 mL Schlenk flask. The reaction flask was deoxygenated via three freeze-pump-thaw cycles. After stirring at 500 rpm for 5 min to ensure the complete dissolution of Compound 3, bromine (Br2, 0.44 mL, 8.4 mmol) was added. The flask was then subjected to one cycle of evacuation and nitrogen backfilling. After refluxing at 60 °C for 2 h, the reaction mixture was cooled and slowly added into ethanol (40 mL). The resulting yellow precipitate was collected by filtration and purified by recrystallization from a mixture of CHCl3 and ethanol to afford DMOBP (867 mg, 78% yield). The 1H NMR spectrum (CDCl3) is shown in Supplementary Figure 9. 1H NMR (500 MHz,
Synthesis of 6,13-dimethoxy-2,3,9,10,18,19,24,25-octa(4′-(trimethylsilyl)phenyl)pentiptycene (DMOTPP)
A mixture of 6,13-dimethoxy-2,3,9,10,18,19,24,25-octabromopentiptycene (DMOBP, 200 mg, 0.180 mmol) and (4-(trimethylsilyl)phenyl)boronic acid (1.38 g, 7.13 mmol) was dissolved in a mixture of anhydrous THF (18 mL) and degassed 1 M aq. K2CO3-solution (24 mL). The resulting mixture was deoxygenated via three freeze-pump-thaw cycles. Under Ar atmosphere, Pd2(dba)3 (52.2 mg, 0.057 mmol) and HP(tBu)3BF4
Synthesis of DMOBPP
A mixture of 6,13-dimethoxy-2,3,9,10,18,19,24,25-octa(4′-(trimethylsilyl)phenyl)pentiptycene (DMOTPP, 200 mg, 0.12 mmol) and KBr (307 mg, 2.58 mmol) was dissolved in a solvent mixture of CH3COOH
Synthesis of PAF-336 and PAF-337
Ni(COD)2 (1.96 mmol, 538 mg), 2,2′-bipyridyl (1.96 mmol, 306 mg), and COD (2.04 mmol, 0.251 mL) were successively added to dehydrated DMF (20 mL) under a nitrogen atmosphere. The mixture was heated at
Characterizations
The manufacturer, model, and country of origin of the experimental instruments have been provided in the Supplementary Materials.
RESULTS AND DISCUSSION
Simulation and characterization of PAFs
Computational modeling can be employed to predict pore aperture and evaluate the structural feasibility of porous materials. Therefore, the anticipatory frameworks for PAF-336 (based on acs topology) and PAF-337 (based on bcu topology) were constructed and validated using Material Studio software [Scheme 3]. Computational simulations revealed distinct pore volumes and pore geometries between the two topological frameworks. PAF-336 exhibited a pore volume of 3.5 cm3·g-1 and featured hexagonal pores approximately
Scheme 3. Topology-guided structural design and simulations of (A) PAF-336 and (B) PAF-337. HBPT: 2,3,6,7,14,15-Hexa(4′-bromophenyl)triptycene; DMOBPP: 6,13-dimethoxy-2,3,9,10,18,19,24,25-octa(4′-bromophenyl)pentiptycene.
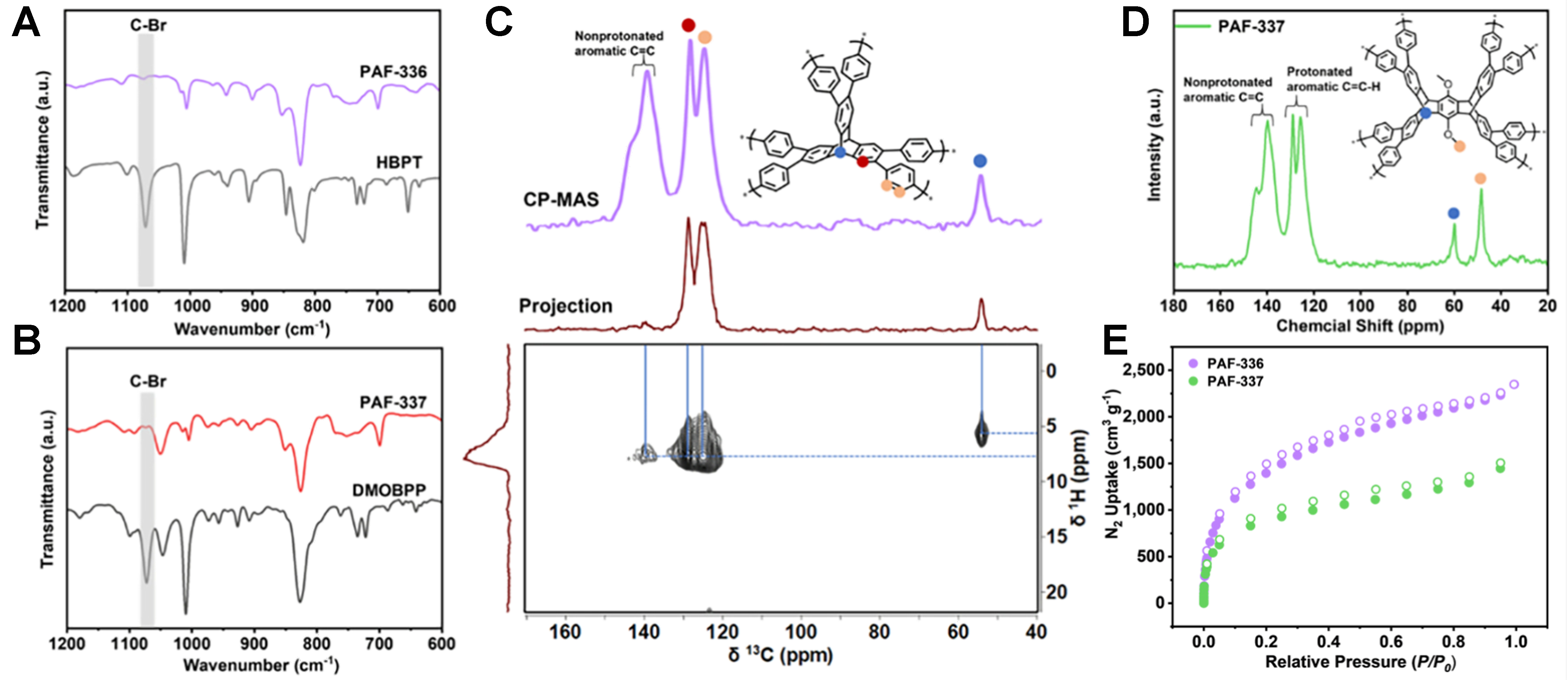
Initially, the monomers of HBPT and DMOBPP were successfully synthesized and structurally characterized [Schemes 1 and 2]. Their chemical identity and purity were rigorously confirmed through 1H NMR and 13C NMR spectroscopy [Supplementary Figures 1-4 and Supplementary Figures 6-12], followed by definitive validation via MALDI mass spectrometry [Supplementary Figures 5 and 13]. The completion of the reaction between the monomers was monitored using Fourier transform-infrared (FT-IR) spectroscopy. As shown in Figure 1A and B [Supplementary Figures 14 and 15], the C-Br peak at 1,070 cm-1 disappeared after the reaction, signaling the successful synthesis of PAF-336 and PAF-337 via a C-C coupling reaction[33]. High-resolution X-ray photoelectron spectroscopy (XPS) analysis of the monomers and resulting PAF-336 and PAF-337 revealed a distinct Br 3d peak in the monomers [Supplementary Figure 16], which disappeared after Ullmann coupling. This absence confirms the bromine removal during polymerization. The chemical structure of PAF-336 and PAF-337 was studied through the solid-state 13C cross-polarization magic-angle spinning (CP-MAS) and 2D 1H-13C heteronuclear correlation spectroscopy (HETCOR) NMR. The solid-state 13C NMR spectra of both PAFs [Figure 1C and D] show two types of carbon atoms: aromatic (125-145 ppm) and aliphatic (54 ppm for PAF-336; 48 and 60 ppm for PAF-337) carbon. Specifically, the 2D 1H-13C HETCOR NMR spectrum of PAF-336 shows different hydrogen atoms at 5.7 and 7.7 ppm, which reveal correlations with the aliphatic carbon atoms and aromatic carbon atoms. Strong correlations are exhibited as blue-, orange-, and red-labeled carbon, suggesting the direct bonding of hydrogen with tertiary carbon and aromatic carbon in the structure of PAF-336. The weak correlations of nonprotonated aromatic carbon atoms with hydrogen reveal long-distance contact. Combined analysis of FT-IR and solid-state NMR unequivocally confirmed the successful synthesis of both PAFs. Thermogravimetric analysis (TGA) under an air atmosphere revealed that PAF-336 exhibited higher thermal stability than PAF-337, with initial decomposition temperatures of ~400 and ~350 °C, respectively
Figure 1. (A) FT-IR spectra of PAF-336 and HBPT; (B) FT-IR spectra of PAF-337 and DMOBPP; (C) Solid-state 13C CP-MAS and 2D
As shown in Figure 1E, the nitrogen physisorption isotherms at 77 K reveal that both PAF-336 and PAF-337 exhibit Type I behavior according to the International Union of Pure and Applied Chemistry (IUPAC) classification. The BET surface areas were calculated to be ~5,210 and ~3,430 m2·g-1, respectively. The lower surface area of PAF-337 relative to PAF-336 is likely attributed to the higher molecular weight of the DMOBPP monomer compared to HBPT, which increases framework density. Additionally, the bcu topology involves denser packing of its constituent units, reducing internal voids and accessible surface area, thus lowering specific surface area. Conversely, the acs topology typically features a more open arrangement, promoting greater porosity and surface accessibility and thereby enhancing specific surface area[46]. To confirm the reproducibility of PAF-336 synthesis, two independent batches showed nearly identical BET surface areas (~5,210 and ~5,180 m2·g-1, Supplementary Figure 23), demonstrating excellent synthetic consistency. Considering the scope of practical applications, PAF-336 must be compressed into gas storage tanks to minimize its volume. Therefore, we measured the nitrogen adsorption isotherms of PAF-336 after compression under a pressure of 10 MPa. As shown in Supplementary Figure 24, the BET surface area
The pore size distribution analysis of PAF-336 and PAF-337 - derived from quenched solid density functional theory (QSDFT) calculations employing a cylindrical pore approximation - exhibited a pronounced peak centered at approximately 2.4 and 2.1 nm, respectively [Supplementary Figure 25]. Quantitative analysis of nitrogen physisorption data at a relative pressure (P/P0) of 0.95 revealed total pore volumes of 3.5 cm3·g-1 for PAF-336 and 2.5 cm3·g-1 for PAF-337. The experimentally calculated pore volume and pore size closely align with the theoretical predictions, with the exception of a slightly larger calculated pore size for PAF-337, which is likely attributable to framework flexibility. This demonstrates that computational simulations are a powerful tool for guiding the fabrication of highly porous polymers. Furthermore, the porosity of both PAFs was compared to other previously reported porous materials, including PAFs, COFs, hyper-cross-linked polymers (HCPs), and hydrogen-bonded organic frameworks (HOFs) [Supplementary Figure 26 and Supplementary Table 1]. Remarkably, the total pore volume of PAF-336 exceeds that of many porous materials with the same specific surface area. This significant observation is attributed to the unique pore structure of PAF-336. The BET surface area (derived from N2 uptake at P/P0 < 0.2) and the total pore volume (measured at P/P0 = 0.95) are related yet distinct physical properties. The high pore volume at a comparable surface area indicates a significant portion of larger pores, likely originating from the framework’s intrinsic architecture and potential defects, which contribute more to volume than to surface area[36]. These features - substantial pore volume together with high specific surface area - constitute critical parameters that significantly influence adsorption capacity and storage efficiency in gas storage applications[29,33]. This structural characteristic improves the adsorption capacity of porous materials by increasing adsorption sites and providing sufficient space. Furthermore, the chemical stability of PAF-336 was systematically assessed under harsh conditions, including exposure to 6 M concentrated HCl and 6 M NaOH for 72 h [Supplementary Figure 27]. The BET surface areas of the acid-treated and base-treated PAF-336 were 5,050 and 5,080 m2·g-1, respectively, corresponding to 97% and 98% retention of the pristine material’s porosity. These results demonstrate the excellent physicochemical stability of PAF-336, indicating its suitability for long-term practical gas storage applications.
Hydrogen storage of PAF-336
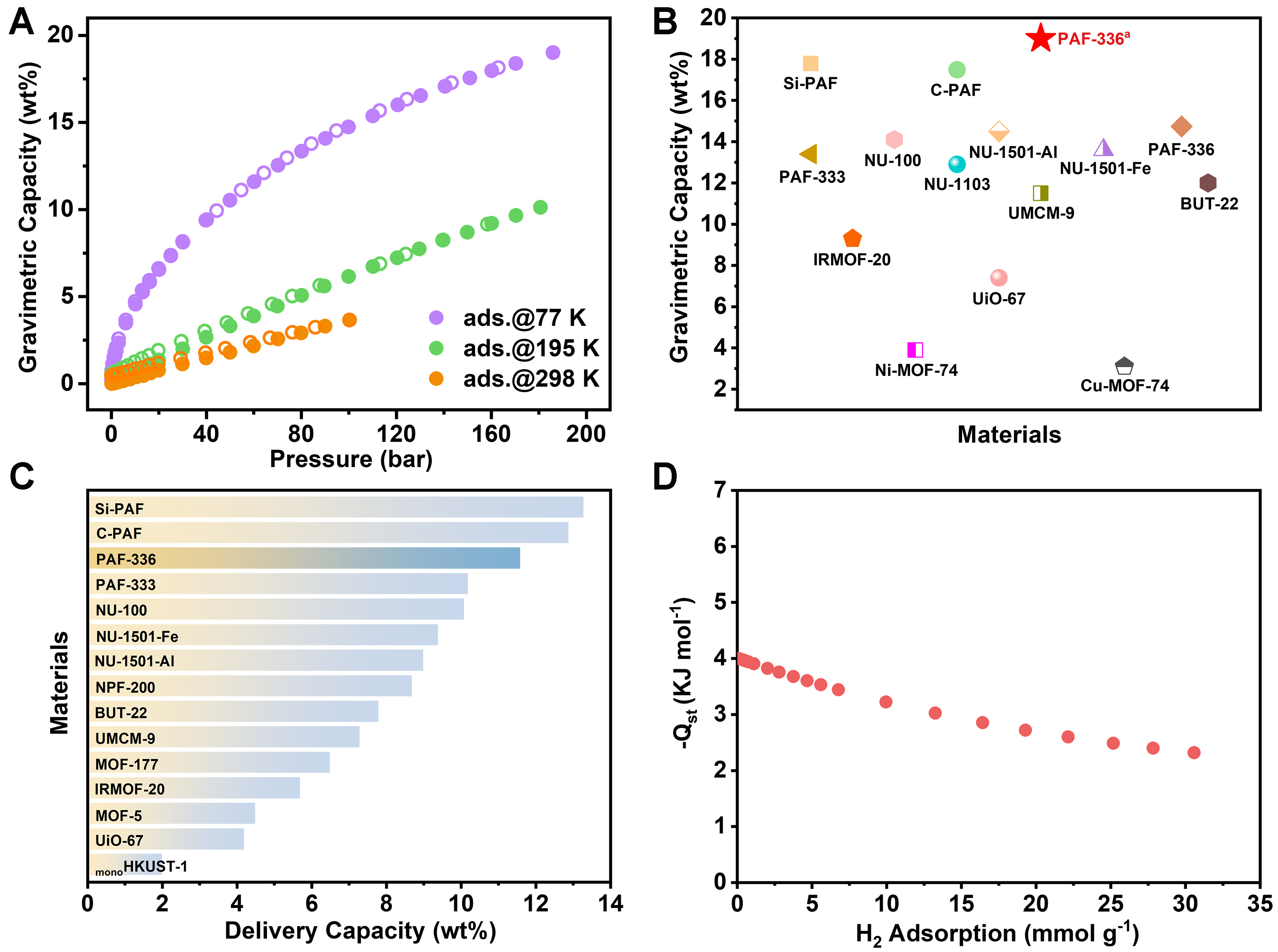
Building on the analysis of pore architectures and H2 storage performance criteria[34], PAF-336 emerges as a highly competitive candidate for H2 storage applications, owing to its exceptionally high specific surface area, structural robustness, and facile synthetic route. In contrast, PAF-337 exhibits inherent synthetic limitations characterized by multistep synthesis, which impose potential challenges for practical application. Therefore, PAF-336 was selected for subsequent property investigation. High-pressure hydrogen adsorption measurements were conducted on PAF-336 at 77, 195, and 298 K to evaluate its H2 storage performance. The total adsorption capacity and heat of adsorption were calculated according to the calculation methods section in the supporting information. The excess and total H2 uptake values, as shown in Figure 2A, Supplementary Figure 28 and 29, reveal a total capacity of 14.7 wt% (35 g·L-1) at 77 K and 100 bar, placing PAF-336 among the top-performing materials [Figure 2B]. Upon increasing the pressure to 185 bar at 77 K, the uptake reaches 19.0 wt%. This exceptional performance is attributed to the material’s high specific surface area and large pore volume. Gravimetric and volumetric capacity are key metrics for vehicle applications, yet balancing them is difficult. Materials with high volumetric capacity often have low gravimetric capacity due to density-volume trade-offs[47]. As shown in Supplementary Table 2 and literature[33], MOFs exceed 40 g·L-1 in volumetric capacity but face stability issues and lower gravimetric performance compared to porous organic polymers. Importantly, our material approaches the U.S. Department of Energy (DOE) volumetric target while maintaining high gravimetric capacity, achieving a balanced adsorption performance that directly contributes to vehicle lightweighting and reduced energy consumption[33].
Figure 2. (A) Gravimetric hydrogen capacity of PAF-336 at various temperatures; (B) Comparison of gravimetric hydrogen capacity with several high-performing adsorbents [Supplementary Table 2]. PAF-336a used the data tested at 185 bar, while PAF-336 used the data tested at 100 bar; (C) Comparison of gravimetric delivery capacities of hydrogen under PSA conditions (100 → 5 bar, 77 K)
The delivery capacity of H2 storage materials is crucial for practical applications. In light-duty fuel cell vehicles, the minimum pressure needed to source hydrogen to the fuel cell module is 5 bar[33]. Therefore, we evaluated the hydrogen delivery capacity of PAF-336 by measuring its storage performance under 100 and 5 bar conditions [Supplementary Figure 30]. The delivery capacity under pressure-swing adsorption (PSA) conditions (100 → 5 bar, 77 K) was determined to be 11.6 wt% for PAF-336, ranking among the top reported porous materials and surpassed only by carbon-based PAF (C-PAF[33], 12.9 wt%) and silicon-based PAF (Si-PAF[33], 13.3 wt%) in gravimetric delivery capacity [Figure 2C]. For a more realistic approximation of practical conditions, the volumetric and deliverable capacities of PAF-336 were determined based on its tapped density to be 28.2 g·L-1 (77 K, 100 bar) and 22.3 g·L-1 (100 → 5 bar, 77 K), respectively
The heat of adsorption is crucial for hydrogen adsorption and desorption because excessively high adsorption heat can be detrimental to hydrogen desorption[49]. The isosteric enthalpy of adsorption (Qst) for hydrogen of PAF-336 was calculated using a virial equation to be -4.0 kJ·mol-1 at low pressure
Methane storage of PAF-336
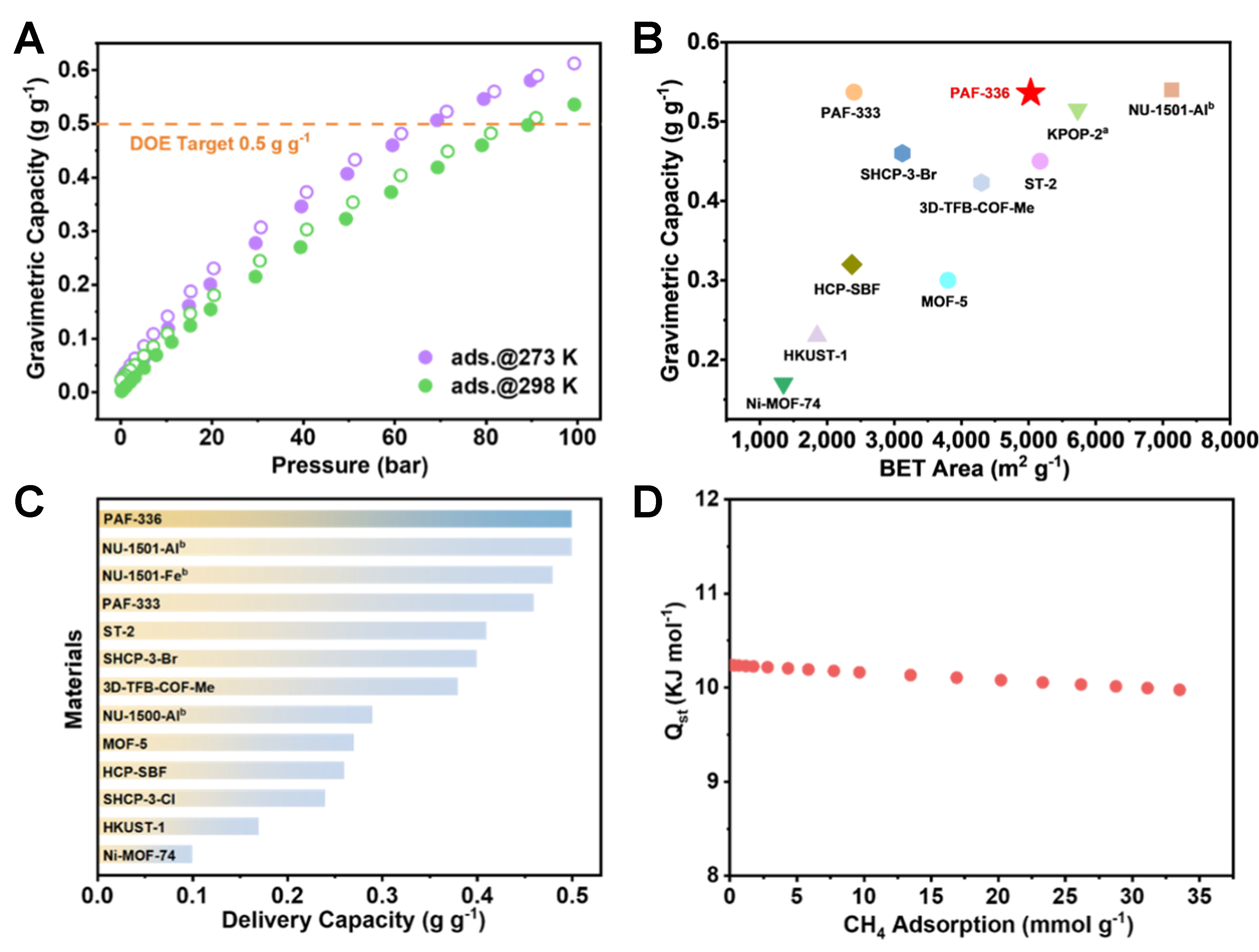
Methane has become an important transitional fuel for achieving carbon offset energy systems due to its large reserves, low cost, and low carbon-to-hydrogen (C/H) ratio[13,51]. However, storage tanks and multi-stage compression are still the majority of CH4 storage technology at present, which is very energy-consuming and unsafe. Porous materials with ultra-high specific surface area provide new strategies for efficient and safe storage of methane. Here, high-pressure methane adsorption was tested to evaluate the adsorption capacity of PAF-336 [Figure 3A, Supplementary Figures 33 and 34]. At 100 bar, the total CH4 uptake of PAF-336 was determined to be
Figure 3. (A) The total methane capacity of PAF-336 at 298 and 273 K; (B) Comparison of the total methane capacity and BET surface areas with previously reported highly porous materials [Supplementary Table 3]; (C) Comparison of the gravimetric delivery capacity of methane with some of the high performing adsorbents (100 → 5 bar, 298 K, Supplementary Table 3); (D) The methane Qst for PAF-336. BET: Brunauer-Emmett-Teller; DOE: U.S. Department of Energy.
The delivery capacity and storage capacity of methane determine the actual driving distance of adsorbed natural gas (ANG) vehicles, as the minimum inlet pressure of the engine requires 5 bar[18]. As shown in Figure 3C, the delivery capacity (100 → 5 bar) calculated for PAF-336 was 0.50 g·g-1 at 298 K, ranking among the top-performing reported porous organic materials, level with NU-1501-Al[47]. For a more realistic approximation of practical conditions, the volumetric and deliverable capacities of PAF-336 were determined based on its tapped density to be 103 g·L-1 (298 K, 100 bar) and 96 g·L-1 (100 → 5 bar, 298 K), respectively [Supplementary Figure 36]. Moreover, the deliverable methane capacity of PAF-336 was evaluated at 298 K between 5 and 80 bar. As shown in Supplementary Table 3, PAF-336 exhibits a high deliverable capacity of 0.42 g·g-1 under these conditions. Although slightly lower than that of NU-1501-Al (0.44 g·g-1) under similar conditions, this value remains highly competitive among advanced porous materials, demonstrating the promising potential of PAF-336 for CH4 storage applications. Cyclical stability is crucial for industrial CH4 storage applications. The material demonstrates robust performance, as evidenced by CH4 capacity over three adsorption-desorption cycles [Supplementary Figure 37]. To further investigate the methane-adsorbent interactions, the Qst was calculated from adsorption isotherms of methane measured at 273 and 298 K, employing the virial equation [Supplementary Figure 38]. Figure 3D shows that PAF-336 has a low Qst value (10.2 kJ·mol-1) at minimal loading, lower than most benchmark porous materials under similar conditions, which will reduce the CH4 uptake at low pressure (5 bar)[18,47,49,52]. As evidenced by the preceding investigation, the superior working capacity of PAF-336 arises from the synergistic interplay between its high pore volume and low adsorption heat. High pore volume enhances methane adsorption capacity by providing ample storage space[53-55], while low adsorption heat facilitates rapid desorption by overcoming the small energy barrier[18].
CONCLUSIONS
In summary, we have rationally designed and successfully synthesized two porous aromatic frameworks, PAF-336 and PAF-337, both featuring high specific surface areas and large pore volumes. Remarkably, PAF-336 delivers exceptional gravimetric H2 and CH4 storage performance, ranking among the highest levels ever reported for porous organic materials. This work demonstrates the powerful capability of rational framework engineering in optimizing gas storage properties, and our ongoing efforts are focused on developing innovative building blocks to further advance the gas storage performance of porous organic frameworks.
DECLARATIONS
Authors’ contributions
Conception: Jia, J.; Su, B. L.; Zhu, G.
Planned the study, analyzed the data, and wrote the manuscript: Cui, L.; Zhang, M.; Wang, Q.; Jia, J.; Bian, Z.
Synthesis and analytical discussions: Cui, L.; Zhang, M.; Wang, Q.; Wang, Z.; Liu, J.
NMR and TEM characterization: Feng, D.; Lin, L.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data can be available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
We thank Beishide Instrument Technology (Beijing) Co., Ltd. for providing the high-pressure gas adsorption measurements. Financial support from the National Key R&D Program of China (2022YFB3805902, 2022YFB3805900), the National Natural Science Foundation of China (Grant Nos. 22131004, U21A20330), the “111” project (B18012), and the Fundamental Research Funds for the Central Universities (2412024QD012) is gratefully acknowledged.
Conflicts of interest
Su, B. L. is Editor-in-Chief of the journal Chemical Synthesis. Zhu, G. is Executive Editor-in-Chief of the journal Chemical Synthesis. Su, B. L. and Zhu, G. were not involved in any steps of the editorial process, notably including reviewers’ selection, manuscript handling, or decision-making. Liu, J. is affiliated with Analysis Lab, Beishide Instrument Technology (Beijing) Co., Ltd. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Zhou, Z.; Ma, T.; Zhang, H.; et al. Carbon dioxide capture from open air using covalent organic frameworks. Nature 2024, 635, 96-101.
2. Zhu, Z.; Tsai, H.; Parker, S. T.; et al. High-capacity, cooperative CO2 capture in a diamine-appended metal-organic framework through a combined chemisorptive and physisorptive mechanism. J. Am. Chem. Soc. 2024, 146, 6072-83.
3. Morris, R. E.; Wheatley, P. S. Gas storage in nanoporous materials. Angew. Chem. Int. Ed. Engl. 2008, 47, 4966-81.
4. Wang, Z.; Zhang, Y.; Jiang, L.; et al. Post-modified porous aromatic frameworks for carbon dioxide capture. Chem. Synth. 2024, 4, 40.
5. Wang, X.; Alzayer, M.; Shih, A. J.; et al. Tailoring hydrophobicity and pore environment in physisorbents for improved carbon dioxide capture under high humidity. J. Am. Chem. Soc. 2024, 146, 3943-54.
6. Li, H.; Zhou, Z.; Ma, T.; et al. Bonding of polyethylenimine in covalent organic frameworks for CO2 capture from air. J. Am. Chem. Soc. 2024, 146, 35486-92.
8. Furukawa, H.; Yaghi, O. M. Storage of hydrogen, methane, and carbon dioxide in highly porous covalent organic frameworks for clean energy applications. J. Am. Chem. Soc. 2009, 131, 8875-83.
9. Breunig, H.; Rosner, F.; Saqline, S.; et al. Achieving gigawatt-scale green hydrogen production and seasonal storage at industrial locations across the U.S. Nat. Commun. 2024, 15, 9049.
10. Yin, Y.; Zhang, Y.; Zhou, X. et. al. Ultrahigh-surface area covalent organic frameworks for methane adsorption. Science 2024, 386, 693-96.
11. Hwang, H. T.; Varma, A. Hydrogen storage for fuel cell vehicles. Curr. Opin. Chem. Eng. 2014, 5, 42-8.
12. Zhang, C.; Cao, X.; Bujlo, P.; et al. Review on the safety analysis and protection strategies of fast filling hydrogen storage system for fuel cell vehicle application. J. Energy. Storage. 2022, 45, 103451.
13. Kumar, K. V.; Preuss, K.; Titirici, M. M.; Rodríguez-Reinoso, F. Nanoporous materials for the onboard storage of natural gas. Chem. Rev. 2017, 117, 1796-825.
14. He, Y.; Chen, F.; Li, B.; Qian, G.; Zhou, W.; Chen, B. Porous metal–organic frameworks for fuel storage. Coord. Chem. Rev. 2018, 373, 167-98.
15. Saha, D.; Grappe, H. A.; Chakraborty, A.; Orkoulas, G. Postextraction separation, on-board storage, and catalytic conversion of methane in natural gas: a review. Chem. Rev. 2016, 116, 11436-99.
16. He, Y.; Zhou, W.; Qian, G.; Chen, B. Methane storage in metal-organic frameworks. Chem. Soc. Rev. 2014, 43, 5657-78.
17. Wang, Y.; Xue, Y.; Züttel, A. Nanoscale engineering of solid-state materials for boosting hydrogen storage. Chem. Soc. Rev. 2024, 53, 972-1003.
18. Makal, T. A.; Li, J. R.; Lu, W.; Zhou, H. C. Methane storage in advanced porous materials. Chem. Soc. Rev. 2012, 41, 7761-79.
19. Zhang, R.; Daglar, H.; Tang, C.; et al. Balancing volumetric and gravimetric capacity for hydrogen in supramolecular crystals. Nat. Chem. 2024, 16, 1982-8.
20. Yu, B.; Tao, Y.; Yao, X.; et al. Single-crystalline 3D covalent organic frameworks with exceptionally high specific surface areas and gas storage capacities. J. Am. Chem. Soc. 2024, 146, 28932-40.
22. Luo, D.; Li, M.; Ma, Q.; et al. Porous organic polymers for Li-chemistry-based batteries: functionalities and characterization studies. Chem. Soc. Rev. 2022, 51, 2917-38.
23. Ma, T.; Zhou, Y.; Diercks, C. S.; et al. Catenated covalent organic frameworks constructed from polyhedra. Nat. Synth. 2023, 2, 286-95.
24. Lee, J. M.; Cooper, A. I. Advances in conjugated microporous polymers. Chem. Rev. 2020, 120, 2171-214.
25. Tan, L.; Tan, B. Hypercrosslinked porous polymer materials: design, synthesis, and applications. Chem. Soc. Rev. 2017, 46, 3322-56.
26. Asif, M. B.; Kim, S.; Nguyen, T. S.; Mahmood, J.; Yavuz, C. T. Covalent organic framework membranes and water treatment. J. Am. Chem. Soc. 2024, 146, 3567-84.
27. Sun, T.; Liang, Y.; Luo, W.; Zhang, L.; Cao, X.; Xu, Y. A general strategy for kilogram-scale preparation of highly crystal-line covalent triazine frameworks. Angew. Chem. Int. Ed. Engl. 2022, 61, e202203327.
28. Ben, T.; Ren, H.; Ma, S.; et al. Targeted synthesis of a porous aromatic framework with high stability and exceptionally high surface area. Angew. Chem. Int. Ed. Engl. 2009, 48, 9457-60.
29. Yuan, D.; Lu, W.; Zhao, D.; Zhou, H. C. Highly stable porous polymer networks with exceptionally high gas-uptake capacities. Adv. Mater. 2011, 23, 3723-5.
30. Ben, T.; Pei, C.; Zhang, D.; et al. Gas storage in porous aromatic frameworks (PAFs). Energy. Environ. Sci. 2011, 4, 3991.
31. Jia, J.; Chen, Z.; Jiang, H.; et al. Extremely hydrophobic POPs to access highly porous storage media and capturing agent for organic vapors. Chem 2019, 5, 180-91.
32. Li, M.; Ren, H.; Sun, F.; et al. Construction of porous aromatic frameworks with exceptional porosity via building unit engineering. Adv. Mater. 2018, 30, e1804169.
33. Kim, D. W.; Chen, Y.; Kim, H.; et al. High hydrogen storage in trigonal prismatic monomer-based highly porous aromatic frameworks. Adv. Mater. 2024, 36, e2401739.
34. Chen, Z.; Kirlikovali, K. O.; Idrees, K. B.; Wasson, M. C.; Farha, O. K. Porous materials for hydrogen storage. Chem 2022, 8, 693-716.
35. Zhou, S.; Qiu, T.; Wang, H.; et al. Flexible porous organic polymers constructed using C(sp3)-C(sp3) coupling reactions and their high methane-storage capacity. Chem. Sci. 2024, 15, 10830-7.
36. Li, H.; Chen, F.; Guan, X.; et al. Three-dimensional triptycene-based covalent organic frameworks with ceq or acs topology. J. Am. Chem. Soc. 2021, 143, 2654-9.
37. Gong, W.; Gao, P.; Gao, Y.; et al. Modulator-directed counterintuitive catenation control for crafting highly porous and robust metal-organic frameworks with record high SO2 uptake capacity. J. Am. Chem. Soc. 2024, 146, 31807-15.
38. Zhu, Q.; Wang, X.; Clowes, R.; et al. 3D cage COFs: a dynamic three-dimensional covalent organic framework with high-connectivity organic cage nodes. J. Am. Chem. Soc. 2020, 142, 16842-8.
39. Yang, Q.; Chen, K.; Schoedel, A.; Wojtas, L.; Perry Iv, J. J.; Zaworotko, M. J. Network diversity through two-step crystal engineering of a decorated 6-connected primary molecular building block. CrystEngComm 2016, 18, 8578-81.
40. Zhou, J.; Zhang, Z.; Xu, L.; et al. Mixed-ligand strategy affording two 6-connected 3-fold interpenetrated metal-organic frameworks with binuclear CoII2/NiII2 subunits: synthesis, crystal structures and magnetic properties. Inorg. Chem. Commun. 2020, 111, 107624.
41. Li, W.; Probert, M. R.; Kosa, M.; et al. Negative linear compressibility of a metal-organic framework. J. Am. Chem. Soc. 2012, 134, 11940-3.
42. Yin, Y.; Zhang, Y.; Zhou, X.; et al. Single-crystal three-dimensional covalent organic framework constructed from 6-connected triangular prism node. J. Am. Chem. Soc. 2023, 145, 22329-34.
43. Zhao, Y.; Irie, T.; Wen, D.; et al. Highly selective separation of benzene/cyclohexane by three-dimensional covalent organic framework with 8,8-connected bcu net topology. ACS. Materials. Lett. 2024, 6, 3063-70.
44. Alezi, D.; Belmabkhout, Y.; Suyetin, M.; et al. MOF crystal chemistry paving the way to gas storage needs: aluminum-based soc-MOF for CH4, O2, and CO2 storage. J. Am. Chem. Soc. 2015, 137, 13308-18.
45. Liu, W.; Wu, E.; Yu, B.; et al. Reticular synthesis of metal-organic frameworks by 8-connected quadrangular prism ligands for water harvesting. Angew. Chem. Int. Ed. Engl. 2023, 62, e202305144.
46. Chen, F.; Zheng, H.; Yusran, Y.; Li, H.; Qiu, S.; Fang, Q. Exploring high-connectivity three-dimensional covalent organic frameworks: topologies, structures, and emerging applications. Chem. Soc. Rev. 2025, 54, 484-514.
47. Chen, Z.; Li, P.; Anderson, R.; et al. Balancing volumetric and gravimetric uptake in highly porous materials for clean energy. Science 2020, 368, 297-303.
48. Parks, G.; Boyd, R.; Cornish, J.; Remick, R. Hydrogen station compression, storage, and dispensing technical status and costs. 2014. https://docs.nrel.gov/docs/fy14osti/58564.pdf. (accessed 31 Dec 2025).
49. Wang, Z.; Jiang, L.; Jia, J.; Zhu, G. Targeted synthesis of interpenetration-free mesoporous aromatic frameworks by manipulating catalysts as templates. Angew. Chem. Int. Ed. Engl. 2025, 64, e202420746.
50. Gómez-Gualdrón, D. A.; Colón, Y. J.; Zhang, X.; et al. Evaluating topologically diverse metal–organic frameworks for cryo-adsorbed hydrogen storage. Energy. Environ. Sci. 2016, 9, 3279-89.
51. Budzianowski, W. M.; Brodacka, M. Biomethane storage: evaluation of technologies, end uses, business models, and sustainability. Energy. Convers. Manag. 2017, 141, 254-73.
52. Yang, S.; Zhong, Z.; Hu, J.; Wang, X.; Tan, B. Dibromomethane knitted highly porous hyper-cross-linked polymers for efficient high-pressure methane storage. Adv. Mater. 2024, 36, e2307579.
53. Tan, J.; Tao, Y.; Zhang, X.; et al. Control over interpenetration for boosting methane storage capacity in metal–organic frameworks. J. Mater. Chem. A. 2021, 9, 24857-62.
54. Froudas, K. G.; Vassaki, M.; Papadopoulos, K.; et al. Expanding the reticular chemistry building block library toward highly connected nets: ultraporous MOFs based on 18-connected ternary, trigonal prismatic superpolyhedra. J. Am. Chem. Soc. 2024, 146, 8961-70.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.