Organic radicals for selective micropollutant removal in complex water
Abstract
Water treatment efficiency is often limited by complex water matrices that inhibit micropollutant degradation. Organic radicals are emerging as a promising alternative to conventional hydroxyl radical-based oxidation due to their higher selectivity toward electron-rich micropollutants (up to 107-109 M-1·s-1) and lower susceptibility to matrix interference. They can be generated under mild conditions via activation of organic peroxides or natural redox mediators and participate in electron transfer, hydrogen abstraction, addition, and polymerization reactions, enabling both micropollutant transformation and potential valorization. However, concerns about toxic by-products from radical precursors have driven strategies such as precursor immobilization and in situ radical generation. Despite these advances, critical knowledge gaps remain in reaction selectivity and the development of sustainable application frameworks. Future research should emphasize data-driven kinetic modeling, utilization of matrix-derived organic radicals, and the design of stable materials to advance practical implementation of organic radical-based water treatment technologies.
Keywords
INTRODUCTION
Advanced oxidation processes have the capability to generate abundant inorganic radicals with high one-electron reduction potentials, such as hydroxyl radical (HO•, 1.9-2.7 V) and sulfate radical (SO4•-,
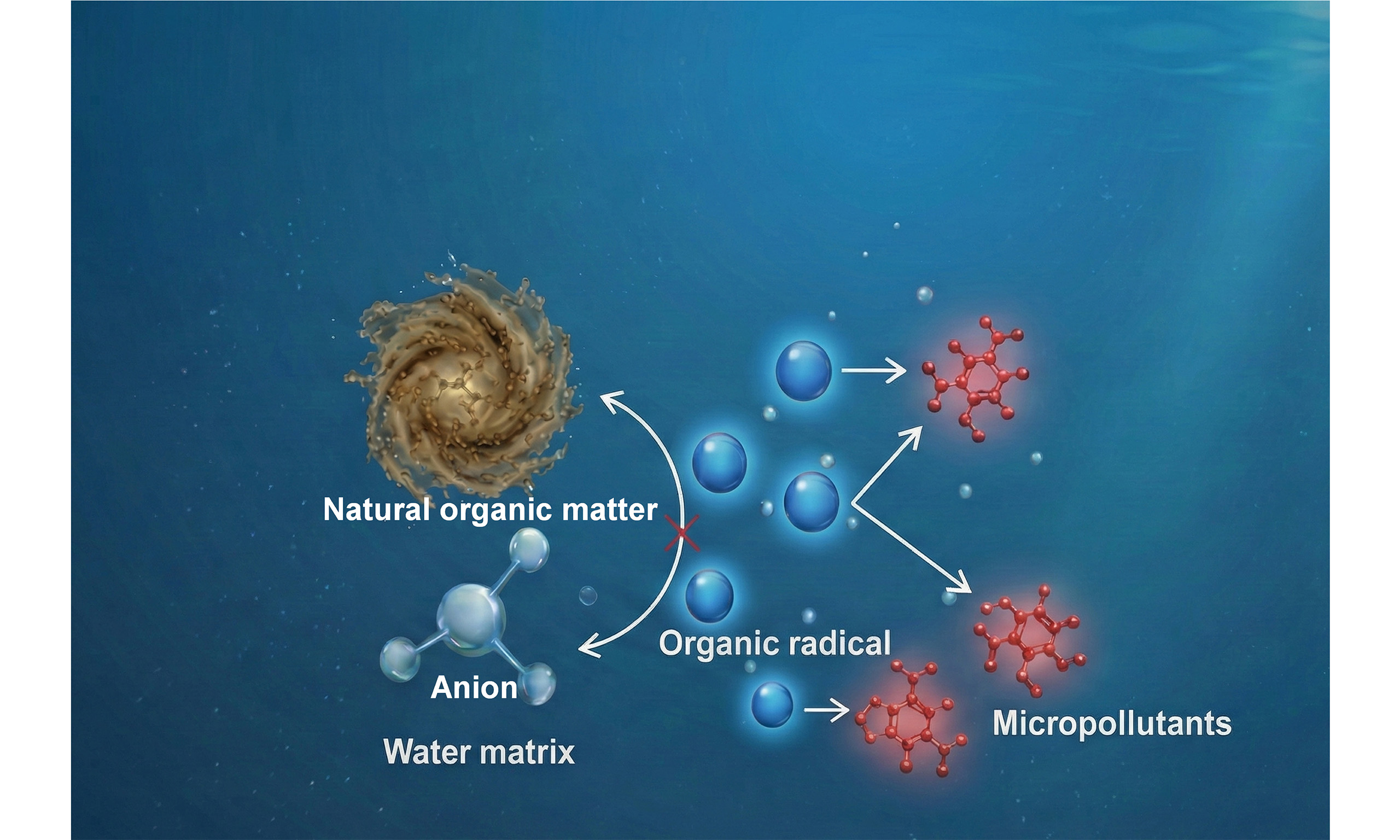
Organic radicals have recently emerged as promising alternatives or complements to conventional inorganic radicals in water treatment, primarily comprising O-centered, N-centered, C-centered, and S-centered radicals[4,9,10]. Compared with inorganic radicals, organic radicals can be generated under milder oxidative conditions, and many exhibit high selectivity toward electron-rich micropollutants with improved tolerance to matrix effects, thereby offering intrinsic anti-interference advantages[11,12]. For example, peracetic acid activation can be activated under ultraviolet (UV) irradiation or metal catalysis to generate acetyloxyl radical [CH3C(O)O•] and peroxyacetyl radical [CH3C(O)OO•], which are capable of selectively degrading micropollutants in water with reduced susceptibility to inorganic anions[13]. In addition, various natural micromolecular redox mediators (a class of organic radical precursors), such as syringaldehyde, acetosyringone, violuric acid, 4-hydroxybenzoic acid, and catechol, can be transformed into organic radicals under redox conditions, including UV irradiation, enzyme-catalyzed oxidation, and metal redox processes. These transformations generate radicals such as phenoxy radicals (PhO•) or semiquinone radicals (SQ•-), which enable micropollutant degradation through mediated electron-transfer or radical-addition pathways[11,12,14-16]. Notably, although macromolecular DOM has traditionally been regarded as a radical scavenger, it can also serve as a precursor of organic radicals (e.g., PhO• and ROO•) under photochemical or oxidative conditions, generating oxidative organic radicals that further degrade micropollutants[4,17]. These findings open new possibilities for harnessing naturally abundant components in water matrices for micropollutant degradation.
Despite growing interest, the reactions between organic radicals and micropollutants remain insufficiently understood, with limited knowledge of their reaction selectivity and the potential application in water treatment. A deeper and systematic investigation is therefore required to fully elucidate their advantages and to enable their rational application in water treatment. This perspective highlights the unique advantages and potential applications of organic radicals in treating micropollutants in water, with a particular focus on their reaction with micropollutants and their compatibility with green and low-carbon treatment strategies.
SELECTIVE REACTIONS OF ORGANIC RADICALS AND MICROPOLLUTANTS
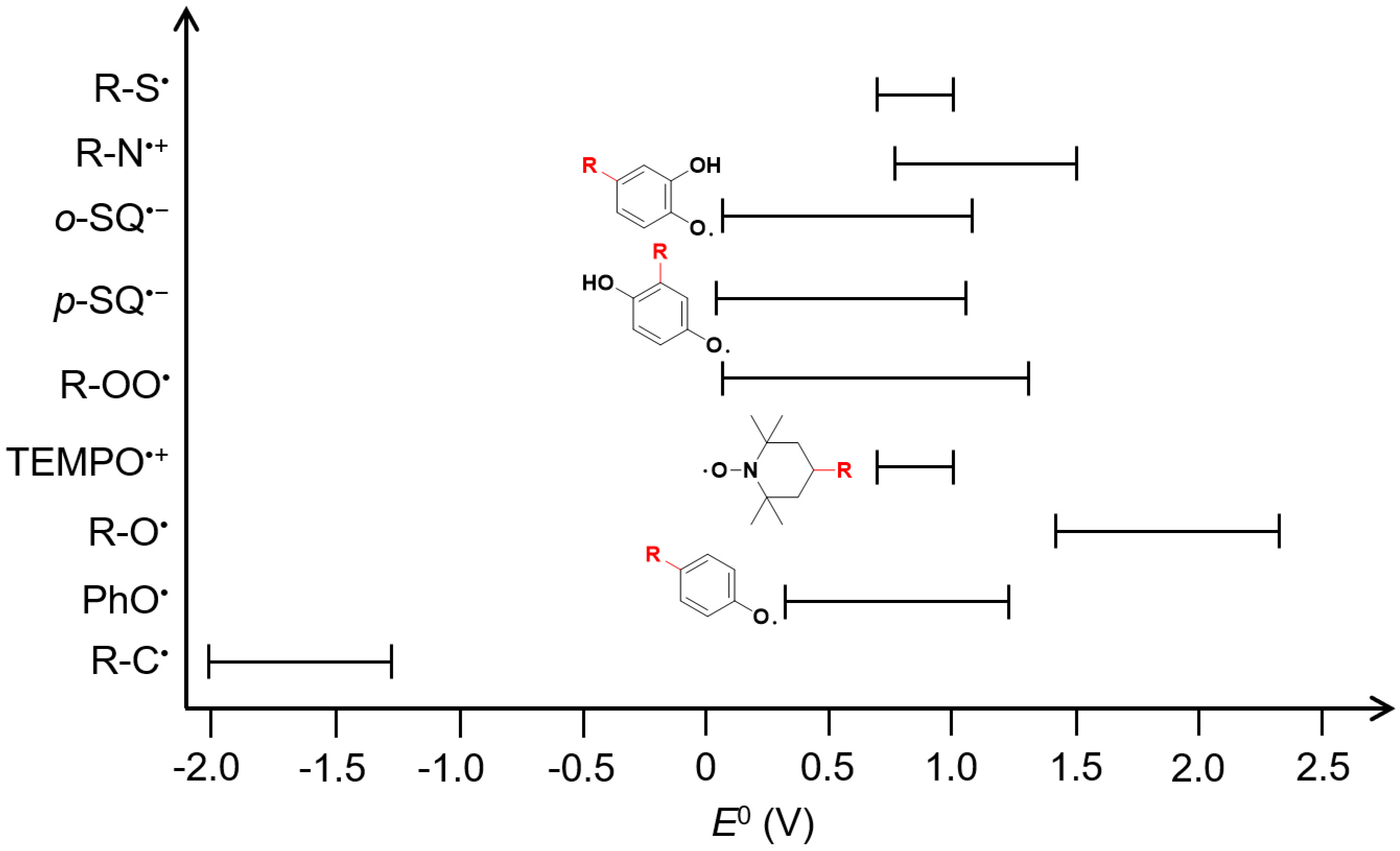
Compared with inorganic radicals such as HO• and SO4•-, organic radicals generally exhibit moderate redox potentials (E0, Table 1). With the exception of certain alkoxy radicals, the E0 of most organic radicals is lower than the upper potential limit of oxide anions (approximately 1.6 V), thereby precluding the oxidation of other anions in aqueous systems by organic radicals [Figure 1][30]. In addition, our previous study shows that in systems containing SQ•-, anions such as Cl-, CO32-, and Br- do not affect its reaction with anilines, further confirming the resistance of organic radicals to anion quenching[11]. Inorganic cations such as Na+, K+, Ca2+ and Mg2+ present in effluent at mg·L-1 levels show negligible reactivity toward radicals because they neither readily gain nor donate electrons[31]. Although SQ•- can react with transition metal cations such as Fe2+ and Cu2+ at rates of 105-108 M-1·s-1, and ROO• reacts with Fe2+ at ~106 M-1·s-1 under acidic conditions, the relatively low concentrations of transition metal cations in effluent (approximately μg·L-1) limit their quenching effect on organic radicals[32-34]. For DOM, the second-order rate constants of it with ROO• are around 105-
The redox potentials (E0) of selected organic radicals
| Organic radical | E0 (V) | Organic radical | E0 (V) | References |
| Tyrosyl•+ | 0.89 | CCl3O• | 2.30 | [18-20] |
| 2-aminopurine•+ | 1.50 | (CH3)2C(O-)O• | 2.19 | [19,20] |
| guanosine•+ | 1.29 | (CH3)2C(OH)O• | 1.45 | [19,20] |
| DPD•+ | 0.76 | CH3CH(OH)O• | 2.27 | [19,21] |
| ABTS•+ | 0.81 | CH3CH(O-)O• | 2.17 | [19,22] |
| PhO• | 0.97 | CH3CH(OH)O• | 1.47 | [19,23] |
| p-CO2--PhO• | 1.04 | (CH3)3CHO• | 1.70 | [19,23] |
| p-(CH3)2N-PhO• | 0.36 | (CH3)3CH2CO• | 1.55 | [19,23] |
| p-NH2-PhO• | 0.42 | (CH3)2C(OH)O• | 2.31 | [19,23] |
| p-OCH3-PhO• | 0.72 | CH3CH2OO• | 0.74 | [19,23,24] |
| p-F-PhO• | 0.93 | (CH3)2CHOO• | 0.64 | [19,23,24] |
| p-Cl-PhO• | 0.94 | CH3OO• | 0.13-0.89 | [23,24] |
| p-Br-PhO• | 0.96 | CCl3OO• | 1.13-1.19 | [23,24] |
| p-I-PhO• | 0.96 | CH3C(O)OO• | 1.14 | [23-25] |
| p-COCH3-PhO• | 1.06 | CH2ClOO• | 1.00 | [23-25] |
| p-CN-PhO• | 1.17 | CH3CHClOO• | 0.99 | [23-25] |
| p-NO2-PhO• | 1.23 | CF3CHClOO• | 1.10 | [23-25] |
| CH3-p-SQ•- | 0.46 | CHCl2OO• | 1.08 | [24-26] |
| p-SQ•- | 0.02-1.04 | CH3CCl2OO• | 1.09 | [24-26] |
| o-SQ•- | 0.04-1.06 | CCl2(CN)OO• | 1.10 | [24-26] |
| COOCH3-o-SQ•- | 0.12 | CCl3CCl2OO• | 1.08 | [24-26] |
| CH2COOH-o-SQ•- | 0.02 | CH2CHCH2OO• | 0.75 | [24-26] |
| C2H5-p-SQ•- | 0.40 | C6H5CH2OO• | 0.78 | [24-26] |
| 2-Cl-p-SQ•- | 0.62 | (CH3)3COO• | 0.43 | [24-26] |
| 4-Cl-p-SQ•- | 0.65 | 4-C6H5-C6H4OO• | 1.22 | [24-26] |
| TEMPO•+ | 0.74 | 4-CN-C6H4OO• | 1.15 | [24,25,27] |
| p-NH2-TEMPO•+ | 0.93 | 9-C14H9OO• | 1.26 | [24,25,27] |
| p-OH-TEMPO•+ | 0.85 | CO2•- | -1.85 | [27,28] |
| p-NHCOCH3-TEMPO•+ | 0.86 | CH3• | -2.0 | [27,28] |
| 4-H-Ph-S• | 0.69 | CH2CH3• | -1.9 | [18,20,29] |
| Ph-COS• | 1.21 | •CH2OH | -1.18 | [18,20,29] |
| Cysteine• | 0.73-1.10 | •CH3CHOH | -1.25 | [18,20,29] |
The reaction kinetics between organic radicals and micropollutants differ markedly from those involving inorganic radicals. While HO• typically reacts with various micropollutants (e.g., phenols and alcohols) at near diffusion-controlled rates (108-1010 M-1·s-1, pH 1-9), organic radicals tend to exhibit compound-specific and structure-dependent rate constants[40]. For example, for certain micropollutants containing electron-donating structures such as phenols, amines, and thiols, the second-order reaction rate constants of PhO• with them can reach up to 107-109 M-1·s-1 under neutral and alkaline conditions, whereas reactions with alcohols, olefins, and fatty acid esters are slower, with rate constants of less than 104 M-1·s-1[4,32]. These kinetics are closely related to the electronic properties of target micropollutants, including electron density distribution, ionization state, and substituent effects, which are influenced by adjacent electronegative atoms or the resonance stabilization of PhO•[11]. Importantly, although the intrinsic reaction rates of organic radicals may be lower than those of inorganic radicals, their effective reactivity in real water systems can be comparable or even superior due to reduced competition from inorganic ions and DOM. This feature highlights the importance of evaluating organic radical-driven kinetics under environmentally relevant conditions.
Organic radicals can react with micropollutants through multiple pathways, including electron transfer, hydrogen atom abstraction, radical addition, and coupling or polymerization reactions [Figure 2][11,32,34]. In many cases, electron transfer dominates the initial interaction with electron-rich micropollutants, generating radical cation intermediates that subsequently undergo structural rearrangement or bond cleavage. In contrast, addition and polymerization reactions often proceed with high selectivity (107-109 M-1·s-1, pH 1–14) and improved controllability[11,32,34,41,42]. These characteristics not only facilitate targeted micropollutant transformation but also suggest the possibility of converting micropollutants into high-value products during wastewater treatment. For instance, the rapid polymerization of PhO• can be leveraged to convert phenolic compounds in water into recoverable polymers, providing a promising strategy for transforming pollutants into value-added resources[41,42].
APPLICATIONS OF ORGANIC RADICALS IN WATER TREATMENT
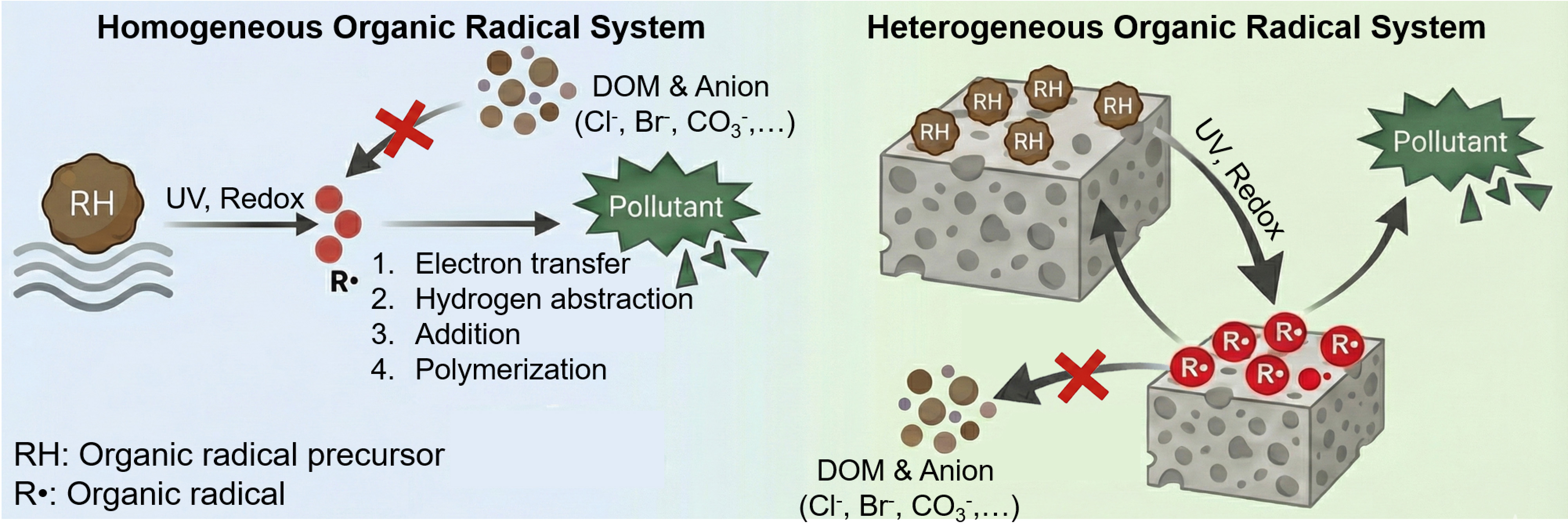
While organic radicals exhibit selective degradation of micropollutants in real water, their inherent solubility in water presents potential toxicity risks, limiting their direct application for micropollutant degradation. For example, peracetic acid can trigger the formation of disinfection by-products in water containing halide ions[43]. Similarly, the addition of redox mediators not only poses the risk of transformation into more toxic by-products, but some of these compounds can also generate colored transformation products, thereby adversely affecting water colorimetry and turbidity[44].
A feasible strategy to circumvent these limitations is the immobilization of organic radical precursors, such as 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonate), (2,2,6,6-tetramethylpiperidin-1-yl) oxyl and 9-azabicyclo [3,3,1] nonan-3-one-9-oxyl (ABNO), onto engineered nanosystems such as chitosan, cellulose, silica, and ferrite nanoparticles[45,46]. Immobilization can reduce direct exposure of soluble mediators to the aquatic environment, mitigate leakage-related toxicity, and enable more controllable organic radical generation at solid-water interfaces [Figure 2]. For example, Zhang et al. successfully covalently immobilized ABNO onto the surface of silica. Upon the addition of permanganate, the immobilized ABNO is rapidly activated to generate oxygen-centered ABNO radicals, capable of efficiently degrading micropollutants[45]. Nevertheless, the deployment of immobilized redox mediators faces challenges related to recyclability. Repeated activation cycles can weaken the active sites of the mediators through chemical transformation or surface passivation, resulting in decreased re-generability of redox mediators and gradual material deactivation.
Constructing regenerable organic radical precursors offers an alternative strategy to overcome these limitations. Under solar irradiation, quinone compounds can be efficiently converted into hydroquinone and SQ•-[47]. Hydroquinone derivatives exhibit strong reducing ability and can be readily oxidized by molecular oxygen, generating SQ•- and quinone species[47]. Ye et al. synthesized donor-acceptor ligand structures containing anthraquinone derivatives[48,49]. Specifically, upon light irradiation, the material undergoes charge-carrier separation, generating long-lived oxygen-centered organic radicals (SQ-like radical). These organic radicals subsequently react with micropollutants, converting into anthrahydroquinone-type species. The anthrahydroquinone moieties are then oxidized by molecular oxygen, regenerating the original structure. This closed photoredox cycle demonstrates high stability and reusability (≥ 50 cycles) under light irradiation while effectively suppressing secondary by-product accumulation, thus enhancing the environmental compatibility and operational safety of organic radical-based water treatment technologies.
SUMMARY AND PERSPECTIVE
In summary, organic radicals hold significant potential for degrading micropollutants in complex water bodies. However, several aspects still require further investigation.
The reaction kinetics of organic radicals with micropollutants remain poorly understood, and the transient spectrum is currently the primary experimental technique used to quantify these reactions[11]. However, due to the substantial structural diversity of organic radicals, even subtle variations in functional groups can lead to significant changes in reactivity. Consequently, experimentally determining reaction kinetics is a complex, labor-intensive, and potentially unending process. This can be achieved by constructing comprehensive and standardized kinetic databases, systematically integrating molecular descriptors derived from quantum chemical calculations with structural features, and developing graph neural network models coupled with physics-informed or hybrid modeling frameworks to capture both molecular structure and underlying reaction mechanisms[50]. In addition, rigorous cross-validation and independent experimental validation are necessary to ensure model reliability and generalizability. Such efforts would facilitate the rational design of more selective and efficient organic radical-based processes for water treatment applications.
In addition, DOM is an important source of organic radicals in real water matrices, and the targeted utilization of DOM-derived organic radicals holds great promise for water treatment applications. Previous studies have shown that SQ•- in DOM can complex with MnO2 or Zn2+, enhancing the stability and persistence of SQ•-, which in turn improves the degradation of sulfonamide micropollutants[51,52]. Future research should prioritize optimizing targeted strategies to enhance the generation of DOM-derived organic radicals while suppressing their self-decay. Specifically, controlled photoactivation approaches and catalytic mineral interfaces can be engineered to promote the selective formation of organic radicals[17,51]. Surface complexation strategies may be employed to stabilize organic radicals and prolong their lifetimes[51]. In addition, systematic optimization of solution chemistry parameters, such as pH and temperature, can further regulate radical concentrations.
Finally, although immobilized organic radical precursors or radical-generating materials show clear potential for application in packed-bed reactors and catalytic membrane systems, their practical performance is governed by a critical trade-off between radical reactivity and material stability[45]. In immobilized organic radical systems, reactions mainly occur at the solid-liquid interface, requiring micropollutants to diffuse to the material surface before degradation. Under high-flow or high-throughput conditions, mass transfer limitations can markedly reduce treatment efficiency, particularly in packed-bed or fixed-bed reactors. Addressing this issue requires rational material and reactor design to enhance mass transport while maintaining radical reactivity and structural stability. Effective strategies include constructing hierarchical micro-meso-macroporous architectures to facilitate micropollutant diffusion, increasing specific surface area without compromising mechanical strength, and optimizing the spatial distribution of active sites to minimize radical self-quenching[53,54]. In addition, flow-through catalytic membranes can reduce boundary layer resistance, and fluidized-bed or structured monolith reactors can improve hydrodynamic performance[55].
DECLARATIONS
Authors’ contributions
Made substantial contributions to conception: Zhou, Y.; Yang, X.
Performed data acquisition: Zhou, Y.
Provided administrative, technical, and material support: Leung, K. S. Y.; Lee, Y.; Ouyang, G; Yang, X.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Some elements in the Graphical Abstract are created by Gemini 3.0, released 2025-11-18.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Nos. 22506221, 22561160125 and 22425607), Guangdong Provincial Science and Technology Planning Project (2025A0505020031), and the Hong Kong Scholarship Program (XJ2024045).
Conflicts of interest
Yang, X. is an Associate Editor of Journal Greenverse Science. She had no involvement in the review or editorial process of this manuscript, including but not limited to reviewer selection, evaluation, or the final decision, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Lee, J.; von Gunten, U.; Kim, J. H. Persulfate-based advanced oxidation: critical assessment of opportunities and roadblocks. Environ. Sci. Technol. 2020, 54, 3064-81.
2. Schwarzenbach, R. P.; Escher, B. I.; Fenner, K.; et al. The challenge of micropollutants in aquatic systems. Science 2006, 313, 1072-7.
3. Miklos, D. B.; Remy, C.; Jekel, M.; Linden, K. G.; Drewes, J. E.; Hübner, U. Evaluation of advanced oxidation processes for water and wastewater treatment - a critical review. Water. Res. 2018, 139, 118-31.
4. Yang, X.; Rosario-Ortiz, F. L.; Lei, Y.; Pan, Y.; Lei, X.; Westerhoff, P. Multiple roles of dissolved organic matter in advanced oxidation processes. Environ. Sci. Technol. 2022, 56, 11111-31.
5. Leenheer, J. A.; Croué, J. Peer reviewed: characterizing aquatic dissolved organic matter. Environ. Sci. Technol. 2003, 37, 18A-26A.
6. Lian, L.; Yao, B.; Hou, S.; Fang, J.; Yan, S.; Song, W. Kinetic study of hydroxyl and sulfate radical-mediated oxidation of pharmaceuticals in wastewater effluents. Environ. Sci. Technol. 2017, 51, 2954-62.
7. Westerhoff, P.; Mezyk, S. P.; Cooper, W. J.; Minakata, D. Electron pulse radiolysis determination of hydroxyl radical rate constants with Suwannee River fulvic acid and other dissolved organic matter isolates. Environ. Sci. Technol. 2007, 41, 4640-6.
8. Yang, Z.; Qian, J.; Shan, C.; Li, H.; Yin, Y.; Pan, B. Toward selective oxidation of contaminants in aqueous systems. Environ. Sci. Technol. 2021, 55, 14494-514.
9. Yang, S.; Sun, S.; Xie, Z.; et al. Comprehensive insight into the common organic radicals in advanced oxidation processes for water decontamination. Environ. Sci. Technol. 2024, 58, 19571-83.
10. Zhang, N.; Samanta, S. R.; Rosen, B. M.; Percec, V. Single electron transfer in radical ion and radical-mediated organic, materials and polymer synthesis. Chem. Rev. 2014, 114, 5848-958.
11. Zhou, Y.; Lei, Y.; Kong, Q.; et al. o-Semiquinone radical and o-benzoquinone selectively degrade aniline contaminants in the periodate-mediated advanced oxidation process. Environ. Sci. Technol. 2024, 58, 2123-32.
12. Song, Y.; Jiang, J.; Qin, W.; Li, J.; Zhou, Y.; Gao, Y. Enhanced transformation of organic pollutants by mild oxidants in the presence of synthetic or natural redox mediators: a review. Water. Res. 2021, 189, 116667.
13. Ao, X. W.; Eloranta, J.; Huang, C. H.; et al. Peracetic acid-based advanced oxidation processes for decontamination and disinfection of water: a review. Water. Res. 2021, 188, 116479.
14. Chan, J. C.; Paice, M.; Zhang, X. Enzymatic oxidation of lignin: challenges and barriers toward practical applications. ChemCatChem 2019, 12, 401-25.
15. Jiang, C.; Garg, S.; Waite, T. D. Hydroquinone-mediated redox cycling of iron and concomitant oxidation of hydroquinone in oxic waters under acidic conditions: comparison with iron-natural organic matter interactions. Environ. Sci. Technol. 2015, 49, 14076-84.
16. Yuan, X.; Pham, A. N.; Miller, C. J.; Waite, T. D. Copper-catalyzed hydroquinone oxidation and associated redox cycling of copper under conditions typical of natural saline waters. Environ. Sci. Technol. 2013, 47, 8355-64.
17. Remke, S. C.; von Gunten, U.; Canonica, S. Enhanced transformation of aquatic organic compounds by long-lived photooxidants (LLPO) produced from dissolved organic matter. Water. Res. 2021, 190, 116707.
18. Wardman, P. Reduction potentials of one-electron couples involving free radicals in aqueous solution. J. Phys. Chem. Ref. Data. 1989, 18, 1637-755.
19. Merényi, G.; Lind, J.; Goldstein, S. Thermochemical properties of α-hydroxy-Alkoxyl radicals in aqueous solution. J. Phys. Chem. A. 2002, 106, 11127-9.
20. Jovanovic, S. V.; Jankovic, I.; Josimovic, L. Electron-transfer reactions of alkylperoxy radicals. J. Am. Chem. Soc. 2002, 114, 9018-21.
21. Laszakovits, J. R.; Patterson, A.; Hipsher, C.; MacKay, A. A. Diethyl phenylene diamine (DPD) oxidation to measure low concentration permanganate in environmental systems. Water. Res. 2019, 151, 403-12.
22. Song, Y.; Jiang, J.; Ma, J.; et al. ABTS as an electron shuttle to enhance the oxidation kinetics of substituted phenols by aqueous permanganate. Environ. Sci. Technol. 2015, 49, 11764-71.
23. Lind, J.; Shen, X.; Eriksen, T. E.; Merenyi, G. ChemInform abstract: the one-electron reduction potential of 4-substituted phenoxyl radicals in water. ChemInform 2016, 21, chin.199017091.
24. Merényi, G.; Lind, J.; Engman, L. One- and two-electron reduction potentials of peroxyl radicals and related species. J. Chem. Soc., Perkin Trans. 2 1994, 2551-3.
25. Jonsson, M. Thermochemical properties of peroxides and peroxyl radicals. J. Phys. Chem. 1996, 100, 6814-8.
26. Song, Y.; Buettner, G. R. Thermodynamic and kinetic considerations for the reaction of semiquinone radicals to form superoxide and hydrogen peroxide. Free. Radical. Biol. Med. 2010, 49, 919-62.
27. Zhang, H.; Shi, Z.; Ma, J.; Cui, F.; Zhang, J.; Strathmann, T. J. Abatement of organic contaminants by Mn(VII)/TEMPOs: effects of TEMPOs structure, organic contaminant speciation, and active oxidizing species. Environ. Sci. Technol. 2022, 56, 10361-71.
28. Koppenol, W. H. Oxyradical reactions: from bond-dissociation energies to reduction potentials. FEBS. Lett. 1990, 264, 165-7.
29. Schmidt Am Busch, M.; Knapp, E. W. One-electron reduction potential for oxygen- and sulfur-centered organic radicals in protic and aprotic solvents. J. Am. Chem. Soc. 2005, 127, 15730-7.
30. Ren, W.; Cheng, C.; Shao, P.; et al. Origins of electron-transfer regime in persulfate-based nonradical oxidation processes. Environ. Sci. Technol. 2022, 56, 78-97.
31. Al Hadidi, N.; Al Hadidi, M. Suitability of reclaimed wastewater effluent from decentralized wastewater plant for irrigation. Appl. Water. Sci. 2021, 11, 173.
32. Neta, P.; Grodkowski, J. Rate constants for reactions of phenoxyl radicals in solution. J. Phys. Chem. Ref. Data. 2005, 34, 109-99.
33. Choubert, J. M.; Pomiès, M.; Ruel, S. M.; Coquery, M. Influent concentrations and removal performances of metals through municipal wastewater treatment processes. Water. Sci. Technol. 2011, 63, 1967-73.
34. Neta, P.; Huie, R. E.; Ross, A. B. Rate Constants for reactions of peroxyl radicals in fluid solutions. J. Phys. Chem. Ref. Data. 1990, 19, 413-513.
35. Huang, Y.; Zhang, Y.; Zhao, M.; Yan, S.; Song, W. Reaction of the peroxyacetyl radical with dissolved organic matter: kinetics, mechanism, and impacts on contaminant degradation. Environ. Sci. Technol. 2026, 60, 6795-804.
36. Schulte-Frohlinde, D.; Behrens, G.; Onal, A. Lifetime of peroxyl radicals of poly(U), poly(A) and single-and double-stranded DNA and the rate of their reaction with thiols. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1986, 50, 103-10.
37. Svingen, B. A.; Powis, G. Pulse radiolysis studies of antitumor quinones: radical lifetimes, reactivity with oxygen, and one-electron reduction potentials. Arch. Biochem. Biophys. 1981, 209, 119-26.
38. Steenken, S.; Neta, P. Electron transfer rates and equilibriums between substituted phenoxide ions and phenoxyl radicals. J. Phys. Chem. 2002, 83, 1134-7.
39. McKay, G.; Dong, M. M.; Kleinman, J. L.; Mezyk, S. P.; Rosario-Ortiz, F. L. Temperature dependence of the reaction between the hydroxyl radical and organic matter. Environ. Sci. Technol. 2011, 45, 6932-7.
40. Haag, W. R.; Yao, C. C. D. Rate constants for reaction of hydroxyl radicals with several drinking water contaminants. Environ. Sci. Technol. 2002, 26, 1005-13.
41. Zhang, X.; Tang, J.; Wang, L.; et al. Nanoconfinement-triggered oligomerization pathway for efficient removal of phenolic pollutants via a Fenton-like reaction. Nat. Commun. 2024, 15, 917.
42. Liu, H. Z.; Shu, X. X.; Huang, M.; et al. Tailoring d-band center of high-valent metal-oxo species for pollutant removal via complete polymerization. Nat. Commun. 2024, 15, 2327.
43. Wang, J.; Xu, J.; Kim, J.; Huang, C. H. Mechanistic insight for disinfection byproduct formation potential of peracetic acid and performic acid in halide-containing water. Environ. Sci. Technol. 2023, 57, 18898-908.
44. Guo, Q.; Zhou, Y.; Pang, S.; et al. Transformation and detoxification of sulfamethoxazole by permanganate (Mn(VII)) in the presence of phenolic humic constituents. Chem. Eng. J. 2021, 413, 127534.
45. Zhang, H.; Ma, J.; Zhang, J.; Strathmann, T. J. ABNO-functionalized silica as an efficient catalyst for enhancing permanganate oxidation of emerging contaminants. Environ. Sci. Technol. 2023, 57, 635-42.
46. Huang, Y.; Zou, J.; Lin, J.; et al. ABTS as both activator and electron shuttle to activate persulfate for diclofenac degradation: formation and contributions of ABTS•+, SO4•-, and •OH. Environ. Sci. Technol. 2023, 57, 18420-32.
47. Chen, Z.; Jin, J.; Song, X.; Zhang, G.; Zhang, S. Redox conversion of arsenite and nitrate in the UV/quinone systems. Environ. Sci. Technol. 2018, 52, 10011-8.
48. Huang, Y.; Li, X.; Yan, H.; et al. Reusable photocatalytic film for efficient water disinfection under low light intensity. Nat. Water. 2025, 3, 1003-16.
49. Zhang, L.; Huang, Y.; Yan, H.; et al. Oxygen-centered organic radicals-involved unified heterogeneous self-fenton process for stable mineralization of micropollutants in water. Adv. Mater. 2024, 36, e2401162.
50. Zhang, J.; Chen, S.; Ren, H.; Li, Z. Enhancing the predictive kinetics of intramolecular H-migration reactions of ether peroxy radicals by integrating machine learning with quantum chemistry: a comparative study of generic rate rules and machine learning techniques. Ind. Eng. Chem. Res. 2025, 64, 9900-13.
51. Zhong, C.; Cao, H.; Huang, Q.; Xie, Y.; Zhao, H. Degradation of sulfamethoxazole by manganese(IV) oxide in the presence of humic acid: role of stabilized semiquinone radicals. Environ. Sci. Technol. 2023, 57, 13625-34.
52. Ulas, G.; Lemmin, T.; Wu, Y.; Gassner, G. T.; DeGrado, W. F. Designed metalloprotein stabilizes a semiquinone radical. Nature. Chem. 2016, 8, 354-9.
53. Sun, M. H.; Gao, S. S.; Hu, Z. Y.; et al. Boosting molecular diffusion following the generalized Murray’s Law by constructing hierarchical zeolites for maximized catalytic activity. Natl. Sci. Rev. 2022, 9, nwac236.
54. Miao, J.; Jiang, Y.; Wang, X.; et al. Correlating active sites and oxidative species in single-atom catalyzed Fenton-like reactions. Chem. Sci. 2024, 15, 11699-718.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.