The road ahead for hydrogel electrolytes in aqueous zinc metal batteries
Abstract
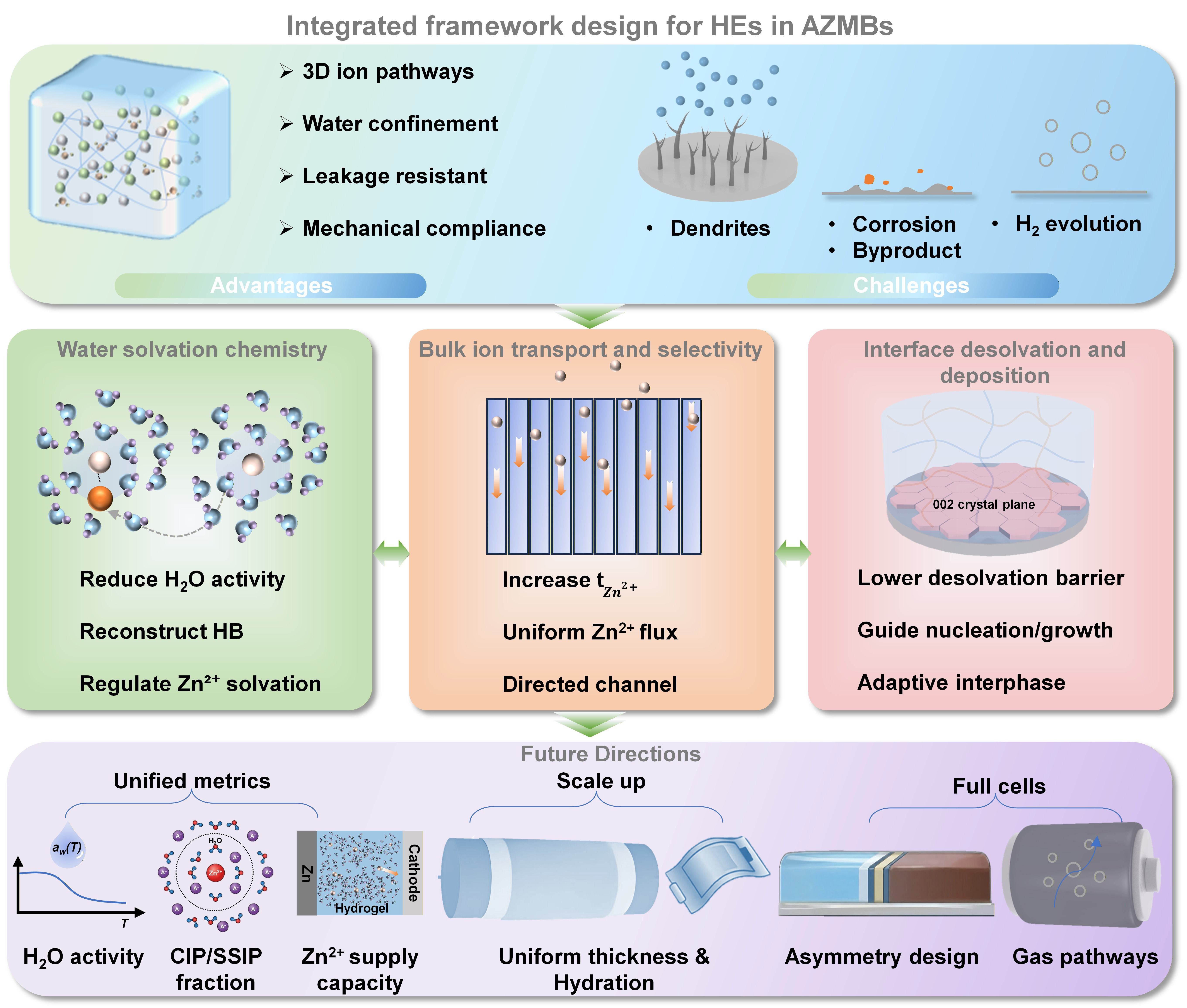
Hydrogel electrolytes, enabled by tunable polymer networks, mechanical robustness, and water confinement, provide a promising platform for stabilizing Zn metal anodes in aqueous zinc metal batteries. Progress can be broadly viewed through three coupled modules: microenvironment control to regulate water activity and Zn2+ solvation, bulk transport engineering to sustain selective Zn2+ delivery, and interfacial design to tune desolvation, nucleation, and growth. Our perspective is that the central challenge is not the independent optimization of these modules, but the rate mismatch that can emerge among them under practical conditions: suppressing water activity alone may not sustain Zn2+ supply, improving bulk transport alone may not resolve interfacial kinetic barriers, and interfacial stabilization alone may remain ineffective if local ion delivery is insufficient. As a result, the practical operating window is often governed by how well these three processes are temporally and spatially coordinated, especially at high current densities and areal capacities. Additionally, scalable fabrication, full-cell architectures that accommodate electrode asymmetry, and sealed-cell management of gas evolution and pressure are needed.
Keywords
INTRODUCTION
Aqueous zinc metal batteries (AZMBs) have attracted widespread attention in various energy storage applications due to their intrinsic high safety, low cost, and the high theoretical gravimetric capacity of zinc metal (820 mAh g-1)[1-3]. However, zinc anodes in aqueous environments undergo coupled failure pathways, including dendritic growth, hydrogen evolution, and corrosion. These processes continuously consume electrolyte and increase polarization, which sharply limits the stable operating range in terms of both current density and areal capacity[4]. For deformable and wearable devices, conventional separators and liquid electrolytes also suffer from interfacial contact degradation, insufficient mechanical stability, and leakage risk[5]. A central challenge is therefore to realize safe, stable, and controllable Zn2+ transport and deposition under aqueous conditions.
Hydrogel electrolytes (HEs) provide a platform to reshape electrochemical behavior in AZMBs through structural design and chemical tunability[6-7]. Their three-dimensional polymer networks enable quasi-solid operation while simultaneously serving as both separator and electrolyte, maintaining continuous ionic pathways and stable interfacial contact at the device scale[8]. Functional groups and additives in the network can regulate water coordination and the local Zn2+ solvation structure, making water chemistry a design variable rather than a passive outcome of the bulk electrolyte[9-11].
Under practical conditions of high current density and high areal capacity, however, intrinsic constraints remain. Compared with liquid electrolytes, effective Zn2+ transport in HEs is highly sensitive to pore connectivity, tortuosity, and the swelling state[12]. Sustained flux demands can therefore lead to stronger concentration polarization, interfacial Zn2+ depletion, and accelerated water-driven side reactions[10,13]. Microstructural and chemical heterogeneity in the network can further produce local flux variations and destabilize deposition morphology. Residual free water can still participate in hydrogen evolution, corrosion, and byproduct formation, thereby hindering the development of a stable and self-consistent interphase. These observations indicate that failure rarely reflects a single missing property, but instead arises from coupled constraints among water chemistry, transport, and interfacial reactions.
In this perspective, we analyze the interdependence among Zn2+ solvation chemistry, selective bulk transport, and interfacial desolvation and deposition. We discuss how these coupled processes define a sustainable operating window for AZMBs. We then summarize recent progress in three corresponding design dimensions. First, microenvironment regulation aims to reduce free-water activity and reshape the solvation structure. Second, transport regulation seeks to increase the Zn2+ transference number (
PROGRESS IN HYDROGEL ELECTROLYTES
Regulating the water network and solvation structure
In AZMB systems, free water activity, hydrogen-bond (HB) network connectivity, and the number of water molecules in the primary Zn2+ solvation sheath jointly govern the kinetics of hydrogen evolution, corrosion, and byproduct formation. HEs leverage polymer networks and functional components to tune water speciation and the local coordination environment of Zn2+, thereby mitigating water-driven side reactions[14].
A widely adopted strategy is to reconstruct the HB network through multipoint interactions, decreasing the fraction of free water and lowering bulk water activity. Polyhydroxy molecules and other multidentate hydrogen-bond donors can simultaneously engage water molecules and polymer chains. For instance, the introduction of sucrose into polyacrylamide-based hydrogels simultaneously regulates the HB network and the Zn2+ solvation structure, enabling Zn||Zn symmetric cells to operate stably for over 3,970 h at 0.5 mA cm-2 and -10 °C. The corresponding Zn||VO2 full cell also exhibits excellent durability, sustaining 10,000 cycles at 1 A g-1 even at 0 °C[15]. However, it is important to note that excessive immobilization of water or an overly compact polymer network can reduce the concentration of mobile charge carriers, increase viscous resistance, and generate locally starved Zn2+ microenvironments[16]. Moreover, spatially nonuniform microenvironments can induce local flux heterogeneity, exacerbating polarization under high-rate conditions[17].
A second approach targets the Zn2+ solvation sheath directly by introducing functional groups or molecules that compete with water in a controlled manner, partially replacing water molecules within the first solvation shell[18]. Cosolvents and urea-based molecules can coordinate with Zn2+ while simultaneously tuning gel swelling through hydrogen bonding. For instance, tetramethylurea incorporated into polyvinyl alcohol or polyacrylamide gels can lower water activity and adjust solvation composition, contributing to improved corrosion resistance and cycling stability at elevated temperatures[9,19]. In zwitterionic polyelectrolyte gels, increasing salt concentration can reshape solvation through changes in ionic strength and preferential ion–polymer interactions[20].
Overall, microenvironment engineering aims to lower water activity while optimizing Zn2+ solvation. In our view, its effectiveness should be evaluated not only by its ability to suppress water-driven side reactions, but also by whether it preserves sufficient and spatially uniform Zn2+ supply. Otherwise, reducing water activity alone may simply shift the bottleneck from parasitic chemistry to transport limitations under high-rate or high-loading conditions[21]. We argue that microenvironment engineering remains limited in practice by the lack of standardized protocols for quantifying water activity and solvation structure under operating, rather than equilibrium, conditions.
Selective bulk ion transport and decoupling
In HEs, bulk ion transport is not fully captured by ionic conductivity alone. It is also governed by
Hydrogels with polyanionic backbones or anion binding sites immobilize anions through electrostatic interactions, increasing
Enhanced bulk transport, however, does not necessarily translate into interfacial stability[34]. Interfacial reactions and morphology evolution are highly localized and can be amplified by electrode roughness and reaction heterogeneity[35]. Even with an enhanced ion supply, desolvation, nucleation, and growth can still become the dominant lifetime limiting steps. This motivates direct regulation of interfacial processes. In our view, meaningful progress requires a standardized framework that reports
Interfacial desolvation and deposition regulation
After Zn2+ reaches the electrode surface, it undergoes desolvation, nucleation, and growth. Even when bulk transport is improved, a high desolvation barrier, elevated local water activity, or a nonuniform interfacial ion supply can still lead to heterogeneous nucleation and unstable deposition. This is often accompanied by the thickening of byproduct layers and rising polarization[36].
Interfacial regulation generally follows two complementary routes. One is to build structure-guided interfaces that promote uniform nucleation and controlled growth. For example, introducing silk fibroin into hydrogels can yield an ordered, β-sheet-rich network via solvent-induced assembly. Zincophilic polar groups in this network can tune Zn2+ solvation and promote preferential growth along the (002) plane, thereby reducing dendrite formation and side reactions[6].
The second route is to construct dynamic, adaptive interfaces that preserve interfacial contact stability during cycling. For instance, a zinc alginate hydrogel interlayer uses zincophilic carboxylate groups and a negatively charged network to guide Zn2+ transport, repel SO42-, and facilitate interfacial desolvation. Meanwhile, its dynamic flexibility accommodates volume changes and repairs microcracks during cycling, sustaining stable Zn deposition for over 2,200 h without dendrite or byproduct formation[37]. These examples highlight that bulk ion supply and interfacial desolvation kinetics must be co-designed, yet we note that desolvation barriers and their dependence on local water activity remain poorly quantified relative to the field’s focus on dendrite morphology.
OUTLOOK
Based on the above discussion, we suggest that the next stage of HE design should move beyond the isolated optimization of individual properties. A key challenge is the emergence of rate mismatches among water regulation, bulk Zn2+ delivery, and interfacial reaction kinetics, particularly under practically relevant current densities and areal capacities. The resulting concentration polarization can elevate local overpotential and may locally increase water activity, potentially accelerating hydrogen evolution, corrosion, and byproduct accumulation. Approaches that mainly immobilize water or boost apparent conductivity may therefore be counterproductive if they reduce the density of mobile charge carriers, lower
Progress will benefit from a unified, quantitative evaluation framework that links chemistry, transport, and interfaces. At a minimum, studies should report water activity, descriptors of Zn2+ solvation and ion pairing, and the effective Zn2+ flux under practical loads. Ion association can be quantified as the fraction of contact ion pairs (CIPs) versus solvent-separated ion pairs (SSIPs), extracted using a defined protocol [e.g., Raman or infrared (IR) spectral deconvolution, or molecular dynamics (MD) statistics]. Flux should be benchmarked together with concentration polarization and interfacial Zn2+ depletion, rather than inferred from conductivity alone. Interfacial assessment should capture desolvation kinetics, nucleation uniformity, and the evolution of impedance and byproduct layers during cycling. Such reporting can identify the rate-limiting process for a given design and guide rate matching across the three coupled domains.
Translation to practical applications also introduces constraints that remain underdiscussed. Scalable manufacturing will likely require spatially uniform thickness, hydration, and crosslink density across large areas. Full cells impose anode-cathode asymmetry, so hydrogel designs should be validated under realistic electrode loadings and capacity ratios, where cathode compatibility, interfacial contact, and water balance may dominate failure. Sealed devices further convert water and gas dynamics into engineering boundary conditions. Long-term stability is expected to depend on providing controlled pathways and buffering volume for gas, and on monitoring or limiting pressure buildup. If these engineering factors are overlooked, it may lead to significant spatial performance variations across devices, premature battery failure under actual electrode loadings, and unsafe pressure buildup caused by uncontrolled gas evolution. All these issues will hinder the reliable translation of laboratory achievements into practical AZMBs. Addressing these coupled materials and engineering challenges within a shared quantitative framework will be essential for advancing HEs from laboratory demonstrations toward robust, deployable AZMBs.
DECLARATIONS
Authors’ contributions
Conceptualization and writing: Wang, Z.; Liu, S.
Supervision: Zeng, X.; Liu, S.
Critical revision of the manuscript: Zhang, Y.; Yang, B.; Li, W.; Zhu, H.
Availability of data and materials
Not applicable.
AI and AI-assisted tools Statement
Not applicable.
Financial support and sponsorship
This work was supported by grants from the Australian Research Council (IE240100186) and the SafeREnergy Hub (IH200100035). The authors also acknowledge support from the Humboldt Foundation awarded to Zeng, X.
Conflicts of interest
Liu, S. is an Editorial Board Member of the journal Iontronics. Liu, S. was not involved in any steps of the editorial process, notably including reviewer selection, manuscript handling, or decision making. The other authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Dai, Y.; Lu, R.; Zhang, C.; et al. Zn2+-mediated catalysis for fast-charging aqueous Zn-ion batteries. Nat. Catal. 2024, 7, 776-84.
2. Mackanic, D. G.; Chang, T. H.; Huang, Z.; Cui, Y.; Bao, Z. Stretchable electrochemical energy storage devices. Chem. Soc. Rev. 2020, 49, 4466-95.
3. Li, Y.; Ma, X.; Zhang, X.; et al. High Zn(002)-preferential orientation enabled by a proton additive for dendrite-free zinc anodes. Energy. Environ. Sci. 2024, 17, 9205-14.
4. Zhao, L.; Zhao, S.; Zhang, N.; et al. Construction of stable Zn metal anode by inorganic functional protective layer toward long-life aqueous Zn-ion battery. Energy. Storage. Mater. 2024, 71, 103628.
5. Xu, M.; Chen, J.; Zhang, Y.; Raza, B.; Lai, C.; Wang, J. Electrolyte design strategies towards long-term Zn metal anode for rechargeable batteries. J. Energy. Chem. 2022, 73, 575-87.
6. Shen, P.; Xu, S.; Li, D.; et al. A bioinspired flexible hydrogel electrolyte with β-sheet–directed interphase for dendrite-free Zn metal batteries. Energy. Storage. Mater. 2025, 81, 104464.
7. Luo, D.; Yang, L.; Geng, Z.; et al. Underlying principles and practical design strategies of hydrogel electrolytes for long-term stable zinc batteries. Adv. Energy. Mater. 2025, 16, e04151.
8. Li, X.; Wang, D.; Ran, F. Key approaches and challenges in fabricating advanced flexible zinc-ion batteries with functional hydrogel electrolytes. Energy. Storage. Mater. 2023, 56, 351-93.
9. Tian, H.; Yao, M.; Guo, Y.; et al. Hydrogel electrolyte with regulated water activity and hydrogen bond network for ultra-stable zinc electrode. Adv. Energy. Mater. 2024, 15, 2403683.
10. Luo, X.; Nian, Q.; Dong, Q.; et al. Anti-swelling supramolecule-crosslinked hydrogel interphase for stable Zn metal anodes. Adv. Energy. Mater. 2024, 15, 2403187.
11. Xia, H.; Xu, G.; Cao, X.; et al. Single-ion-conducting hydrogel electrolytes based on slide-ring pseudo-polyrotaxane for ultralong-cycling flexible zinc-ion batteries. Adv. Mater. 2023, 35, e2301996.
12. Yang, Y.; Lyu, H.; Wang, Q.; et al. Impact of hydrogel microstructure and mechanics on the growth of zinc dendrites towards long-life flexible batteries. J. Mater. Chem. A. 2023, 11, 12373-83.
13. Peng, H.; Wang, D.; Zhang, F.; et al. Improvements and challenges of hydrogel polymer electrolytes for advanced zinc anodes in aqueous zinc-ion batteries. ACS. Nano. 2024, 18, 21779-803.
14. Guan, J.; Mu, Y.; Wei, X.; et al. Ternary gel electrolyte enabling wide-temperature and high-rate performance in aqueous zinc-ion batteries. Adv. Funct. Mater. 2025, 35, 2508306.
15. An, Y.; Shu, C.; Liu, Y.; et al. Modulating the hydrogen bond for a stable zinc anode with a wide temperature range via the sucrose and polyacrylamide synergistic effect. ACS. Nano. 2025, 19, 11146-63.
16. Qiu, Q.; Fan, W.; Wang, X.; et al. Ordered hierarchical hydrogel electrolyte for long-life zinc-ion batteries. Small 2026, 22, e10607.
17. Zhou, J.; Huang, Q.; Lv, Y.; Song, Z.; Gan, L.; Liu, M. Highly zincophilic-hydrophobic polyzwitterionic hydrogel electrolyte with strong electronegative sulfobetaine-carboxyl motifs for ultrastable zinc-ion batteries. Chem. Sci. 2026, 17, 906-15.
18. Cao, L.; Li, D.; Hu, E.; et al. Solvation structure design for aqueous Zn metal batteries. J. Am. Chem. Soc. 2020, 142, 21404-9.
19. Wang, Y.; Tian, H.; Wu, S.; et al. A Co‐solvent hydrogel electrolyte for high‐temperature and anti-corrosion zinc-ion batteries. Adv. Funct. Mater. 2025, e27198.
20. Lee, S.; Han, I. K.; Jeon, N. G.; et al. Promoting homogeneous zinc-ion transfer through preferential ion coordination effect in gel electrolyte for stable zinc metal batteries. Adv. Sci. (Weinh). 2023, 10, e2304915.
21. Gao, N.; Cui, M.; Xi, K.; et al. Elimination of concentration polarization under ultra-high current density zinc deposition by nanofluid self-driven ion enrichment. Adv. Mater. 2025, 37, e2419034.
22. Lei, Y.; Liu, F.; Chen, L.; et al. Polyanionic hydrogel electrolytes to regulate ion transport behavior in long cycle life zinc-ion batteries. Nano. Energy. 2025, 143, 111284.
23. Ji, G.; Sun, M.; Li, M.; Zheng, J. Sulfonate-modified covalent organic framework integrated hydrogel electrolyte: enhancing AZIBs performances by tailoring microstructures and functional groups. Adv. Funct. Mater. 2025, 35, 2500110.
24. Ling, H.; Kong, X.; Wen, L. Ionic superfluidics: a perspective on emerging frameworks for ion transport in confined channels. Iontronics 2025, 1, 6.
25. Peng, P.; Wang, Z.; Wei, D. Modulating multi-ion dynamics for high-performance iontronic systems. Iontronics 2026, 2, 5.
26. Li, S. Q.; Wang, Z.; Zheng, X.; et al. Biomimetic sandwich-structured tubular ion pump arrays for lithium metal batteries. J. Am. Chem. Soc. 2025, 147, 25883-95.
27. Li, S. Q.; Zhang, L.; Liu, T. T.; et al. A dendrite-free lithium-metal anode enabled by designed ultrathin MgF2 nanosheets encapsulated inside nitrogen-doped graphene-like hollow nanospheres. Adv. Mater. 2022, 34, e2201801.
28. Chan, C. Y.; Wang, Z.; Li, Y.; Yu, H.; Fei, B.; Xin, J. H. Single-ion conducting double-network hydrogel electrolytes for long cycling zinc-ion batteries. ACS. Appl. Mater. Interfaces. 2021, 13, 30594-602.
29. Xu, C.; Wang, H.; Lei, C.; Li, J.; Ma, W.; Liang, X. Fast single metal cation conduction in ion-water aggregated aqueous battery electrolytes. Nat. Commun. 2025, 16, 4574.
30. Sun, M.; Ji, G.; Li, M.; Zheng, J. A robust hydrogel electrolyte with ultrahigh ion transference number derived from zincophilic “chain-gear” network structure for dendrite-free aqueous zinc ion battery. Adv. Funct. Mater. 2024, 34, 2402004.
31. Zhu, X.; Zhang, T.; Zhang, J.; et al. Customizing ion transport by anionphilic nanofiber-polymer electrolyte for stable zinc metal batteries. Adv. Mater. 2026, 38, e19057.
32. Liu, C.; Liu, Y.; Li, Z.; et al. Multi-dimensional ion transport in biohybrid hydrogel electrolytes: porous aromatic framework-mediated dendrite suppression for ultrastable zinc metal anodes. Adv. Funct. Mater. 2025, 35, e08289.
33. Yang, Y.; He, Q.; Hu, C.; et al. Electron-initiated self-growth in situ hydrogel electrolyte with gradient protection interface enables stable zinc metal batteries. ACS. Nano. 2025, 19, 21717-28.
34. Yan, X.; Zhang, J.; Sheng, Y.; et al. Decoupling the trade-off between mechanical properties and ionic conductivity in hydrogel polymer electrolytes by anomalous water-induced microphase separation. ACS. Nano. 2025, 19, 40579-93.
35. Han, J.; Seo, E.; Park, S.; Lee, S. H.; Yoo, J. Aligned nanostructures resolve Zn2+ transport bottlenecks via interfacial kinetics-diffusion coupling in aqueous zinc-ion batteries. Adv. Sci. (Weinh). 2026, 13, e12691.
36. Wang, C.; Gong, Z.; Yuwono, J. A.; et al. Ligand-channel-induced ion liberation in crowded zwitterionic hydrogel electrolyte for efficient zinc metal batteries. Nat. Commun. 2025, 16, 11069.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.