The effects of prenatal alkylphenol exposure on offspring neurodevelopment: an evidence-based risk assessment using the TRAEC strategy
Abstract
The neurodevelopmental effects of early pregnancy exposure to alkylphenols (APs) have been extensively studied, yet findings remain inconsistent. To address these discrepancies, 28 published studies were integrated with an original epidemiological dataset, and the Targeted Risk Assessment of Environmental Chemicals (TRAEC) strategy (version 1.1) was employed. Analysis of the primary early pregnancy cohort data revealed consistent inverse associations between maternal exposure to nonylphenol (NP) [β = -3.032; 95% confidence interval (95%CI): -6.059, -0.005], 4-nonyl-nonylphenol (4-N-NP) (β = -4.280; 95%CI: -7.402, -1.159), and 4-tert-octylphenol (4-T-OP) (β = -5.835; 95%CI: -10.879, -0.792) and children’s Ages and Stages Questionnaire (ASQ) scores, as indicated by generalized linear models (GLMs). These findings suggest that early pregnancy exposure to APs may disrupt neurodevelopmental trajectories in children, with sex-specific vulnerabilities. To provide a thorough assessment throughout gestation, the neurodevelopmental risks of prenatal AP exposure were systematically evaluated by incorporating reliability, weight of concentration, risk intensity, and correlation. Risk rankings for the nine APs evaluated were as follows: 4-para-nonylphenol (4-P-NP) > 4-N-NP > para-nonylphenol (P-NP) > 4-octylphenol (4-OP) > 4-T-OP > nonylphenol (NP) > octylphenol (OP) > 4-nonyl-octylphenol (4-N-OP) > 4-nonylphenol (4-NP). The final comprehensive risk score of 5.91 indicated a moderate level of neurotoxicity risk. Additionally, the TRAEC assessment demonstrated strong alignment with four established frameworks - Toxicological data reliability assessment tool (ToxRTool), Science in Risk Assessment and Policy (SciRAP), Office of Health Assessment and Translation (OHAT), and Integrated Risk Information System (IRIS) - highlighting its methodological robustness. This study successfully applies the TRAEC framework, advancing the understanding of neurodevelopmental risks following prenatal AP exposure.
Keywords
INTRODUCTION
Alkylphenols (APs), a diverse class of environmental endocrine-disrupting chemicals (EDCs) comprising various structural isomers, are both widespread and persistent in the environment[1]. Among these, nonylphenol (NP) is classified as a high production volume (HPV) chemical, with annual production reaching 1.7 million tons in the United States and 100,000 tons in the European Union, acting as both a potent endocrine disruptor and a severe ecotoxicant[2]. Notably, 4-tert-octylphenol (4-T-OP) and 4-nonylphenol (4-NP) dominate industrial and domestic applications, representing over 80% of AP usage in the commercial manufacturing of textiles, paper, and food packaging[3]. Consequently, human exposure to APs is both constant and widespread, primarily through the consumption of contaminated water and food, contact with consumer products, and inhalation of airborne residues. Due to their chemical persistence and lipophilicity, APs resist environmental degradation, accumulate along the food chain, and result in significant human body burdens[1]. This exposure is further evidenced by detectable AP concentrations in maternal urine, breast milk, and umbilical cord blood. Importantly, APs can cross both the placental and blood-brain barriers[4-7], where they may disrupt neuroendocrine balance, induce neurotoxicity, and impair cognitive function. The fetal brain is especially susceptible to such insults during critical windows of neurodevelopment[8]. However, the precise effects of prenatal AP exposure on offspring neurodevelopment remain unclear.
The embryonic stage is a period of heightened vulnerability for neural development[9,10]. Adverse exposures during gestation can have profound and lasting effects on central nervous system (CNS) maturation, often with greater consequences than postnatal exposures[11]. Numerous in vivo studies have shown that prenatal exposure to NP and related APs can impair learning and memory in offspring[12]. Multiple biological mechanisms are involved, including disruption of thyroid hormone balance[13-15], induction of oxidative stress and inflammation, activation of apoptotic pathways[16,17], interference with neuronal DNA synthesis[18], dysregulation of neurotransmitters, and impairment of synaptic plasticity[19]. These mechanisms contribute to neurobehavioral and cognitive dysfunction during early brain development[5]. Additionally, given their strong estrogenic and potential anti-androgenic effects, APs may disrupt fetal neural differentiation through hormone-mediated neurotoxic processes[20]. Nevertheless, current evidence has yet to establish a statistically significant relationship between AP exposure and neurodevelopmental outcomes in children.
Despite extensive experimental and epidemiological evidence supporting the neurotoxic potential of APs[18], findings remain inconsistent. This discrepancy is largely attributed to the absence of a standardized, multidimensional risk assessment framework. First, although commercial APs consist of complex mixtures of structural isomers with varying toxicological potencies, data that systematically differentiate their specific neurodevelopmental impacts are scarce[21]. Second, the dose-response relationship for EDCs is often poorly defined. Some animal studies have demonstrated neurotoxic effects at exceptionally low doses[22], challenging the conventional assumption of linear dose-effect relationships and highlighting the relevance of non-monotonic dose-response (NMDR) models[23]. Consequently, current risk assessment models, such as the European Food Safety Authority (EFSA) linear model, are inadequate for addressing the complexities associated with low-dose exposure and isomeric variations characteristic of APs.
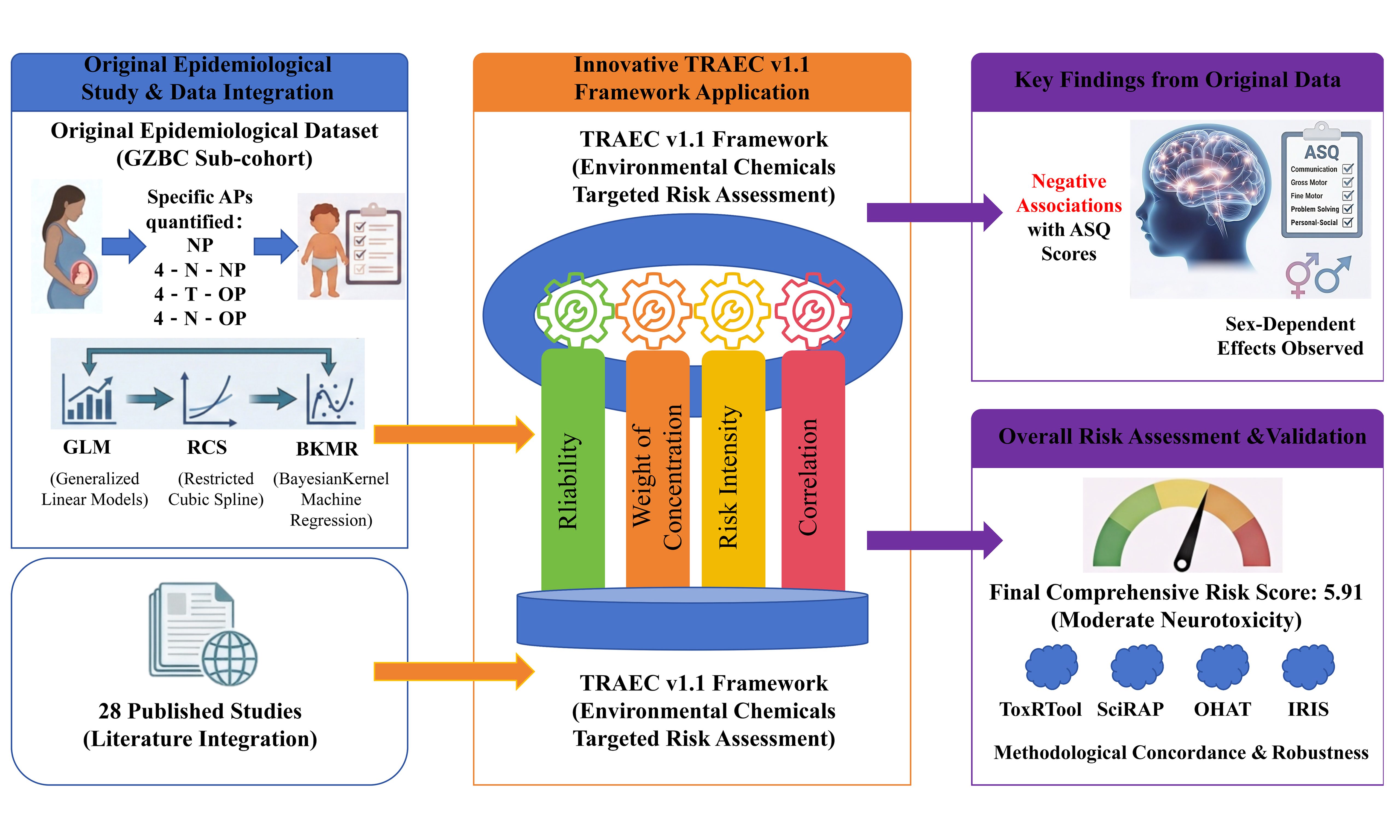
To address these limitations and overcome the constraints of traditional linear paradigms, the present study offers a comprehensive evaluation of AP-induced CNS effects, with a focus on offspring neurodevelopment. Specifically, the Targeted Risk Assessment of Environmental Chemicals (TRAEC) framework was employed to integrate an original early-pregnancy epidemiological dataset with 28 published studies. This systematic approach synthesized multidimensional data - assessing reliability, weight of concentration, risk intensity, and correlation - enabling a standardized risk assessment of prenatal AP exposure. Additionally, the neurotoxic risks of distinct AP isomers were evaluated, and the robustness of the TRAEC framework was compared to established models, including Toxicological data reliability assessment tool (ToxRTool), Science in Risk Assessment and Policy (SciRAP), Office of Health Assessment and Translation (OHAT), and Integrated Risk Information System (IRIS). In summary, this study introduces a novel, multidimensional scoring system that transcends traditional risk assessment paradigms, aiming to enhance both methodological accuracy and public health relevance in evaluating EDC risks.
EXPERIMENTAL
Risk assessment of offspring neurodevelopment by APs based on the TRAEC strategy
This study utilized the TRAEC strategy (version 1.1)[24] to evaluate the neurodevelopmental toxicity of APs, following the TRAEC framework through the following steps:
(1) Problem formulation: The research was focused on the scientific question, “How is early pregnancy AP exposure related to offspring neurodevelopment?” The rationale for this focus is that AP exposure has been known to induce reproductive and neurodevelopmental toxicity, and NP has been detected in maternal plasma, amniotic fluid, and breast milk; however, conclusions from existing studies on the neurodevelopmental effects of APs remain inconsistent.
(2) Evidence collection: A systematic literature search was conducted, including investigator-independent epidemiological studies, to gather targeted evidence. Multi-source data were then integrated to comprehensively compile the existing body of evidence.
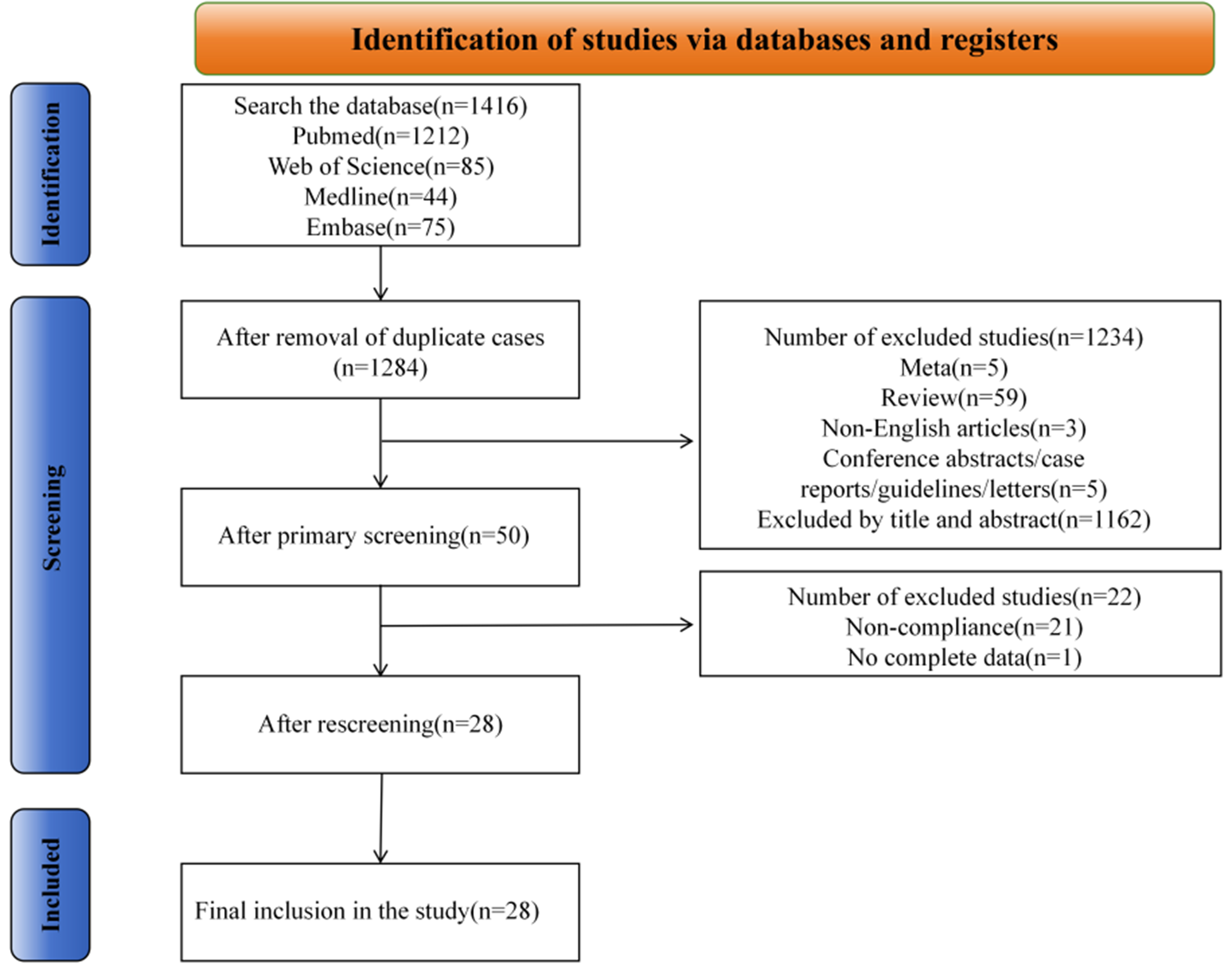
(3) Evidence scoring and conclusion: Scoring was performed using the TRAEC web platform, a free online tool for TRAEC promotion and calculation (https://traec.njmu.edu.cn/). Based on the TRAEC scale and risk matrix, the risk of the target chemicals was graded as follows: (0,4] low risk; (4,8] medium risk; (8,10] high risk. The grading was based on factors including the reliability of study design, the correlation between exposure and health outcomes, weight of concentration, and risk intensity. Conversely, protective effects were defined as follows: (0, -4] low protection; (-4, -8] medium protection; (-8, -10] high protection. The final strength-of-evidence score and risk rating for neurodevelopmental toxicity from prenatal AP exposure were then derived. The literature search covered databases including PubMed, Web of Science, Medline, and Embase up to September 30, 2025. The search terms were: “(# alkylphenol exposure AND # neurological outcome) AND [(# timenode prenatal AND # timenode offspring) OR # in vitro studies]”. Epidemiological, in vivo, and in vitro studies were considered for inclusion. During the screening process, duplicates were first excluded. Titles and abstracts were reviewed to remove irrelevant, non-compliant, review, or non-peer-reviewed studies. Full-text assessments were then carried out to exclude studies lacking sufficient data, not meeting the inclusion criteria, or containing non-extractable data. The detailed search strategy, including specific keywords, is provided in the Supplementary Table 1.
The formula for scoring the evidence is as follows:
ω: Reliability score, ε: weight of concentration, φ: risk intensity. R: correlation score; C: lowest concentration of effects, n: number of evidence items for a single component, i: total number of evidence items.
Epidemiological study design
Quasi-experimental research design
This study utilized data from a sub-cohort of the Guangxi Zhuang Birth Cohort (GZBC), which enrolled participants between September 2014 and December 2019, to investigate the effects of prenatal environmental exposures on maternal and infant health outcomes[25]. Detailed information on the GZBC design, including the inclusion and exclusion criteria, has been provided elsewhere[26]. Briefly, eligible mother-infant pairs were those who delivered at the study hospital and had complete obstetric records. Baseline inclusion criteria required pregnant women to be ≤ 13 weeks of gestation, not to have conceived via assisted reproductive technology, to be of Zhuang ethnicity, and to have either the mother or her partner planning to deliver at the study hospital, as well as willingness to provide peripheral venous blood samples during early pregnancy. Women with incomplete epidemiological data or insufficient blood samples for laboratory analysis were excluded. Upon enrollment, participants provided written informed consent, completed a baseline self-administered questionnaire, and provided fasting peripheral venous blood samples (≤ 13 weeks of gestation). Starting in 2016, eligible children were enrolled in follow-up visits, during which comprehensive questionnaire data and medical records were collected and maintained. The study adhered to the Declaration of Helsinki and was approved by the Ethics Committee of Guangxi Medical University (No. 20140305-001). A flowchart of participant selection is available in Supplementary Figure 1.
To accommodate the inherent heterogeneity across the included literature, which encompasses varying exposure windows ranging from early pregnancy to cord blood matrices, as well as diverse neurodevelopmental assessment instruments such as the Ages and Stages Questionnaires (ASQ), Wechsler Preschool and Primary Scale of Intelligence-Fourth Edition (WPPSI-IV), and Bayley Scales of Infant and Toddler Development, Third Edition (Bayley-III), the TRAEC framework applies a standardized weighting approach. Rather than relying on absolute raw scores, studies were systematically grouped and weighted based on the reliability of study design, sample size, and the statistical strength of the exposure-outcome correlations. This structured methodology ensures that the integration of various biological matrices and clinical assessment instruments remains scientifically valid and reproducible.
Blood sample collection
Fasting venous blood samples from each pregnant woman were collected into non-anticoagulated vacutainers during the first trimester. The samples were kept at 4 °C, centrifuged at 3,500 rpm for 10 min, and the serum was collected and stored in polyethylene tubes at -20 °C on the same day. The serum samples were periodically shipped on dry ice to the laboratory and stored at -80 °C until further analysis.
Serum NPs and octylphenol analysis
The serum samples were thawed at 4 °C the day before analysis. After thorough mixing, 500 μL of serum was transferred into a glass centrifuge tube. To this, 500 μL of 1 M sodium dihydrogen phosphate dihydrate buffer (pH = 5.4), 5 μL of a 1 μg/mL isotope-labeled internal standard mixture containing 4-N-NPC13 (Cambridge Isotope Lab, Inc.), 4-T-OPC13 (Cambridge Isotope Lab, Inc.), and 4-N-OPd17 (ISOREAG, Inc.), and 50 μL of β-glucuronidase/sulfatase (2,000 units/mL, Sigma, St. Louis, MO, USA) were added. The mixture was vortexed for about 10 s and then enzymatically incubated overnight at 37 °C in a water bath. Following incubation, 2 mL of a n-hexane/acetone (7:3, v/v) mixture was added. The sample was vortexed for approximately 2.5 min, sonicated for 15 min, and centrifuged at 4,000 rpm for 10 min. The supernatant (approximately 1.6 mL) was transferred to a new glass centrifuge tube. Subsequently, 2 mL of methyl tert-butyl ether was added, and the extraction process was repeated using the same method. The combined supernatants were evaporated to dryness in a vacuum oven at 40 °C. The residue was reconstituted in 100 μL of a methanol/0.1% ammonia solution (50:50, v/v) and filtered for instrumental analysis.
NP and octylphenol (OP) concentrations in maternal serum were determined using ultra-performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS; Xevo TQD, Waters, USA). Separation was performed on an Acquity UPLC BEH C18 column (1.7 μm, 2.1 mm × 50 mm; Waters, USA). The mass spectrometer operated in negative-ion electrospray ionization mode with multiple reaction monitoring (MRM). The optimal parameters were as follows: capillary voltage, 2.9 kV; desolvation temperature, 550 °C; desolvation gas flow, 50 L/h; and cone gas flow, 1,000 L/h. The limits of detection (LODs) for NP, 4-nonyl-nonylphenol (4-N-NP), 4-T-OP, and 4-nonyl-octylphenol (4-N-OP) in serum were 0.135, 1.718, 0.439, and 0.787 ng/mL, respectively.
Quality assurance and quality control
Due to the widespread presence of APs as environmental contaminants, stringent quality assurance and quality control (QA/QC) procedures were implemented. Sample preparation was carefully conducted to avoid plastic contamination, and each glass tube was thoroughly washed with methanol and ultrapure water before use. To monitor background contamination, field and procedural blanks were included in each analytical batch. The target analytes in all blank samples were either not detected or present at concentrations well below the LODs, indicating negligible contamination. Matrix spike recoveries were assessed at various concentration levels, yielding acceptable recoveries ranging from 86.8% to 101.8%, with relative standard deviations (RSDs) between 8.0% and 12.9%. Calibration curves demonstrated excellent linearity (R2 > 0.990) across the target concentration ranges. All concentrations were quantified using stable isotope-labeled internal standards to correct for matrix effects and analytical losses.
ASQ assessment in children
The ASQ is an internationally recognized developmental screening tool designed for children from birth to five years of age. It provides a rapid assessment of developmental performance across five key domains: communication, gross motor, fine motor, problem-solving, and personal-social. In this study, the raw data were analyzed by an independent researcher blinded to the study conditions. Scores for each domain, as well as the total ASQ score, were calculated and presented as standardized scores, with a maximum possible score of 60.
Covariates
Sociodemographic information, including pre-pregnancy weight, height, maternal education level, alcohol consumption, passive smoking, folic acid supplementation, parity, family income, and medical history, was collected via a structured self-administered questionnaire during the first prenatal visit. Reproductive and delivery data, including gestational hypertension, gestational diabetes, gestational age, maternal age at delivery, infant sex, birth weight, and birth length, were obtained from electronic medical records. Maternal pre-pregnancy body mass index (BMI, kg/m2) was calculated as weight divided by height squared.
Based on previous studies[26,27], the models included the following confounding factors: family income, maternal education level, maternal occupation, breastfeeding duration, folic acid supplementation, use of nutritional supplements, passive smoking, maternal age at delivery, and infant sex.
Statistical analysis
Independent samples t-tests, Wilcoxon rank-sum tests, and chi-square tests were used to assess baseline characteristics. Normally distributed continuous variables are presented as mean ± standard deviation (SD), non-normally distributed variables as median (interquartile range, IQR), and categorical variables as number (percentage). The relationships among APs were analyzed using Spearman correlation, while the internal consistency of the ASQ scores was evaluated using Pearson correlation. Due to skewed distributions, maternal serum AP concentrations were log10-transformed and included in the models as continuous variables.
Generalized linear models (GLMs) were employed to estimate the associations between prenatal AP exposure and child neurodevelopment, with results presented as β coefficients and 95% confidence intervals (95%CIs). AP concentrations were analyzed both as log10-transformed continuous variables and as ordinal categorical variables (quantiles, Q1-Q3), with integer scores assigned to test for linear trends. A dose-response relationship was assessed using restricted cubic spline (RCS) models with three knots at the 10th, 50th, and 90th percentiles of the log10-transformed AP concentrations, with the 50th percentile as the reference. Additionally, Bayesian Kernel Machine Regression (BKMR) was applied to analyze the independent effects of individual APs, their interactions within the mixture, and the overall mixture effect[26,28]. The BKMR model, with 50,000 iterations, was implemented using a Markov chain Monte Carlo algorithm.
Sensitivity analyses included: (1) stratification by infant sex; (2) repeating the GLM after excluding mothers who took folic acid supplements; and (3) validating the reliability of the TRAEC strategy using four conventional risk assessment tools, including ToxRTool, SciRAP, OHAT, and IRIS[29,30].
All statistical analyses were performed using R software (version 4.5.1). The BKMR and RCS models were implemented using the bkmr and rms packages, respectively. To control for potential Type I error inflation due to multiple comparisons in the primary GLM analyses, p-values were adjusted using the false discovery rate (FDR) method based on the Benjamini-Hochberg procedure. An FDR-adjusted P-value (q-value) < 0.05 was considered statistically significant. Figures were generated using GraphPad Prism (version 9.2).
RESULTS AND DISCUSSION
Literature search and study characteristics
A total of 1,416 studies were initially identified through database searches. Based on predefined inclusion and exclusion criteria, 1,366 studies were excluded, leaving 50 articles for preliminary inclusion. After full-text review, 22 studies were further removed, and a final set of 28 articles met the eligibility criteria for TRAEC risk assessment [Figure 1]. Since some articles contributed multiple lines of evidence, a total of 44 evidence entries were included, consisting of 13 epidemiological studies, 21 in vivo experiments, and 10 in vitro assays [Tables 1 and 2, Supplementary Table 2]. Among the 28 publications, 16 were published between 2018 and 2025, while the remaining 12 were published before 2017. All studies examined the impact of prenatal AP exposure on offspring neurodevelopment, with risk assessments focusing primarily on nine AP compounds: NP, 4-N-NP, 4-T-OP, 4-N-OP, 4-NP, 4-octylphenol (4-OP), 4-para-nonylphenol (4-P-NP), para-nonylphenol (P-NP), and OP. The flow diagram of the literature selection process is presented in Figure 1.
Contextual details of included epidemiological studies
| Study | Population (n) | Gender (% male) | Exposure Concentration (mean ± SD, ng/mL or 95%CI) | Detection methods | β/OR/RR/HR (95%CI) | Outcomes of neurodevelopment | Conclusion |
| Lin et al. 2017[25] | 356 | 52.7-56.3 | NP (91.7 ± 83.9) OP (9.0 ± 19.1) | UPLC-MS | NP: 0.1 (-5.2, 5.4) OP: -5.3 (-10.7, 0.0) | Based on the Taiwan Birth Cohort Study, neurodevelopmental assessments were conducted in 2- and 7-year-old children using the CDIIT and WISC-IV, respectively. Multiple linear regression analysis revealed that the detection rates of NP and OP were 77.6% and 68.3%, respectively, with median levels of 72.6 and 3.3 ng/mL, respectively, in 2-year-old children | / |
| Wang et al. 2022[31] | 150 | 50 | NP (91.7 ± 83.9) | HPLC | NP: -11.29(-18.62, -3.96) | Children’s neurodevelopment was assessed using the Bayley-III, WPPSI-IV. Results showed that children aged 4-6 years had higher urinary NP detection rates and concentrations than those aged 2-3 years. Notably, boys in the high-concentration group had significantly lower FSIQ scores than those in the NP reference group (95%CI: -18.62, -3.96, P < 0.05) | + |
| Long et al. 2024[26] | 221 | 56.6 | NP (45.72, 204.73) N-NP (1.32, 1.78) 4-T-OP (12.38, 89.03) 4-N-OP (0.63, 0.95) | UPLC-MS | NP: -2.38 (-4.59, -0.16) | Prenatal exposure to various APs impacts the IQ of preschool children, exhibiting potential non-monotonic and sex-specific effects | + |
| This work | 175 | 54.9 | NP (55.16, 224.04) 4-N-NP (1.22, 1.78) 4-T-OP (19.27, 92.48) 4-N-OP (0.56, 0.96) | UPLC-MS | NP (Communication) -1.71(-3.50, 0.07) | Prenatal exposure to various APs impacts the ASQ scores of preschool children, exhibiting potential non-monotonic and sex-specific effects | + |
Contextual details of the included in vitro studies
| Study | Location | Exposure period | Cell type | Cell source | Type of APs | Exposure concentration | Outcomes of neurodevelopment | Conclusion |
| Xu et al. 2023[60] | Korea | 24 h | CGC | Cerebellum of B. orientalis | 4-OP | 0, 2, 10, 20, 50, 100 mM | In B. orientalis embryos, OP induced oxidative damage and apoptotic cell death, and potentiated EMT, ectopic migration, and defective chondrogenic differentiation of cranial NCCs. The downregulation of sox9 and upregulation of sox10, foxd3, and migration-related genes may underlie OP-induced underdevelopment of cranial cartilages | + |
| Qiu et al. 2019[34] | China | 24 h | BV2 MG | Beijing Cell Research Center | 4-N-NP | 50 μM | Mechanistic investigations reveal that NP exposure increases the production of nitric oxide and prostaglandin E2 by overactivating microglia. The TLR4/MyD88/NF-κB and NRF2 signaling pathways are deeply involved in this microglial activation and the subsequent overproduction of inflammatory mediators, corroborating observations in the offspring hippocampus | + |
| Lee et al. 2024[8] | Korea | 48 h | CGC | Cerebellum of mice | 4-N-NP | 10-11 M, 10-8 M, 10-5 M | NP exposure directly activates microglia and reduces the expression of myelin proteins in oligodendrocytes. These cellular disruptions align with in vivo findings, where maternal NP exposure increases microglia and oligodendrocyte counts in the adult cerebral cortex, ultimately contributing to anxiety- and depression-like behaviors | + |
| Lee et al. 2024[35] | Korea | 24 h | CGC | Cerebellum of mice | 4-T-OP | 10-13 M, 10-11 M, 10-9 M, 10-7 M, 10-5 M, 10-3 M | The neurodevelopmental impact of OP exposure persists into adulthood, driven by microglial dysfunction mediated by the ER signaling pathway in the offspring brain | + |
| Yao et al. 2024[33] | China | 24 h | NE-4C | Stem Cell Bank of Chinese Academy of Sciences | NP | 1, 3, 5 μM | Exposure to NP reduces cell proliferation by inhibiting the Shh signaling in NE-4C cells. Furthermore, it suppresses the proliferation of NPCs in the subgranular zone of the dentate gyrus, leading to depletion of the NPC pool | + |
| Kim et al. 2006[42] | Korea | 24 h | hESC | MEF-CM | NP, OP | 12.5-200 μM | NP and OP can trigger apoptosis in hESCs through pathways dependent on caspase activation and Fas-FasL interactions. Notably, hESC-derived NPCs are more sensitive to these toxic substances than undifferentiated hESCs, indicating that the toxic stress response varies significantly across developmental stages | + |
| Tran et al. 2020[43] | Korea | 24 h | CGC | Cerebellum of rat | 4-T-OP | 10-8 M, 10-6 M | Perinatal OP exposure induces significant neurogenesis defects and behavioral abnormalities. It not only impairs neurogenesis and neurite outgrowth but also damages memory and social behaviors, exacerbating anxiety-like phenotypes in offspring | + |
| Xu et al. 2021[45] | Korea | 24 h | CGC | Cerebellum of B. orientalis | 4-OP | 0, 0.1, 1, 5, 10, 25, 50 mM | In the Frog Embryo Teratogenesis Assay-Xenopus model, OP affected embryonic growth, increased mortality, and induced tadpole malformations. At low concentrations, OP increased lipid peroxidation and endoplasmic reticulum stress, while high concentrations induced apoptosis. OP also affected embryonic growth and the development of skin melanocytes and head chondrocytes via disruption of NCC differentiation | + |
| Litwa et al. 2014[44] | Poland | 24 h | CD1 | Cerebellum of mice | 4-P-NP | 1, 5, 10 mM | Overall, studies have demonstrated the apoptotic and toxic effects of NP on neuronal cells, suggesting that NP-induced apoptosis involves both intrinsic (caspase-9) and extrinsic (caspase-8) apoptotic pathways. Evidence also indicates that NP impairs ERs and stimulates retinoid X receptor intracellular signaling in neuronal cells during early developmental stages | + |
Epidemiological associations between AP exposure and neurodevelopment
Using the TRAEC strategic framework, this study integrated original epidemiological data to investigate the association between prenatal AP exposure and ASQ scores in preschool children. The key findings are summarized as follows.
Baseline characteristics of the mother-child pairs
The characteristics of the mother–child pairs are shown in Table 3. The mean maternal age at delivery was 29.05 ± 5.62 years, with a pre-pregnancy BMI of 20.32 ± 2.78 kg/m2. Among the mothers, 43.3% were exposed to passive smoking during pregnancy, and 61.7% had at least a high school education. Approximately 49.7% were primiparous, and 70.9% reported taking folic acid supplements during pregnancy. In 44.9% of households, the annual family income was below 100,000 yuan.
Characteristics of the mother-child pairs (n = 175)
| Characteristics | Mean ± SD/number (%) |
| Children’s characteristics | |
| Gender | |
| Boys | 96 (54.9) |
| Girls | 79 (45.1) |
| Age (years) | 4.56 ± 0.64 |
| Residential area | |
| Urban | 132 (75.4) |
| Rural | 43 (24.6) |
| Annual family income | |
| Less than 29,900 yuan | 29 (16.6) |
| 30,000 to 99,900 yuan | 67 (38.3) |
| More than 100,000 yuan | 79 (45.1) |
| Children’s height (cm) | 103.20 ± 5.90 |
| Children’s weight (kg) | 15.73 ± 2.54 |
| Birth weight (g) | 3,119.89 ± 438.32 |
| Birth length (cm) | 49.73 ± 1.94 |
| Neonatal BMI (kg/m2) | 12.55 ± 1.09 |
| Sitting height (cm) | 61.42 ± 11.36 |
| Head circumference (cm) | 49.53 ± 1.66 |
| Maternal characteristics | |
| Pre-pregnancy BMI (kg/m2) | 20.32 ± 2.78 |
| Maternal educational level | |
| Primary and junior high schools | 67 (38.3) |
| Senior high school | 36 (20.6) |
| Above junior college level | 72 (41.1) |
| Maternal age at delivery | |
| < 35 years | 140 (80%) |
| ≥ 35 years | 35 (20%) |
| Gestational age (weeks) | 38.66 ± 1.14 |
| Duration of breastfeeding | |
| < 6 m | 19 (10.9) |
| ≥ 6 m | 156 (89.1) |
| Nutritional supplementation | |
| Yes | 163 (93.1) |
| No | 12 (6.9) |
| Folic acid supplementation | |
| None | 51 (29.1) |
| Started before pregnancy | 32 (18.3) |
| Started in early pregnancy | 92 (52.6) |
| Maternal smoking exposure | |
| Yes | 76 (43.3) |
| No | 99 (56.6) |
| Alcohol consumption during pregnancy | |
| Yes | 10 (5.7) |
| No | 165 (94.3) |
| Parity | |
| Primiparity | 87 (49.7) |
| Multiparity | 88 (50.3) |
| Children’s ASQ scores | |
| Communication | 52.40 ± 8.07 |
| Gross motor | 55.03 ± 8.03 |
| Fine motor | 42.00 ± 14.55 |
| Problem-solving | 51.69 ± 8.42 |
| Personal-social | 54.03 ± 7.86 |
| Total score | 255.14 ± 30.62 |
Of the 175 children included, 96 (54.9%) were boys, and 79 (45.1%) were girls. The mean gestational age was 38.66 ± 1.14 weeks, with a mean birth weight of 3,119.89 ± 438.32 g. The majority (89.1%) of children were breastfed for at least six months. At the time of assessment, the mean age of the children was 4.56 ± 0.64 years.
ASQ scores were as follows: Communication (52.40 ± 8.07), Gross Motor (55.03 ± 8.03), Fine Motor (42.00 ± 14.55), Problem-Solving (51.69 ± 8.42), and Personal-Social (54.03 ± 7.86). The total ASQ score was 255.14 ± 30.62.
Distribution of APs and ASQ internal consistency
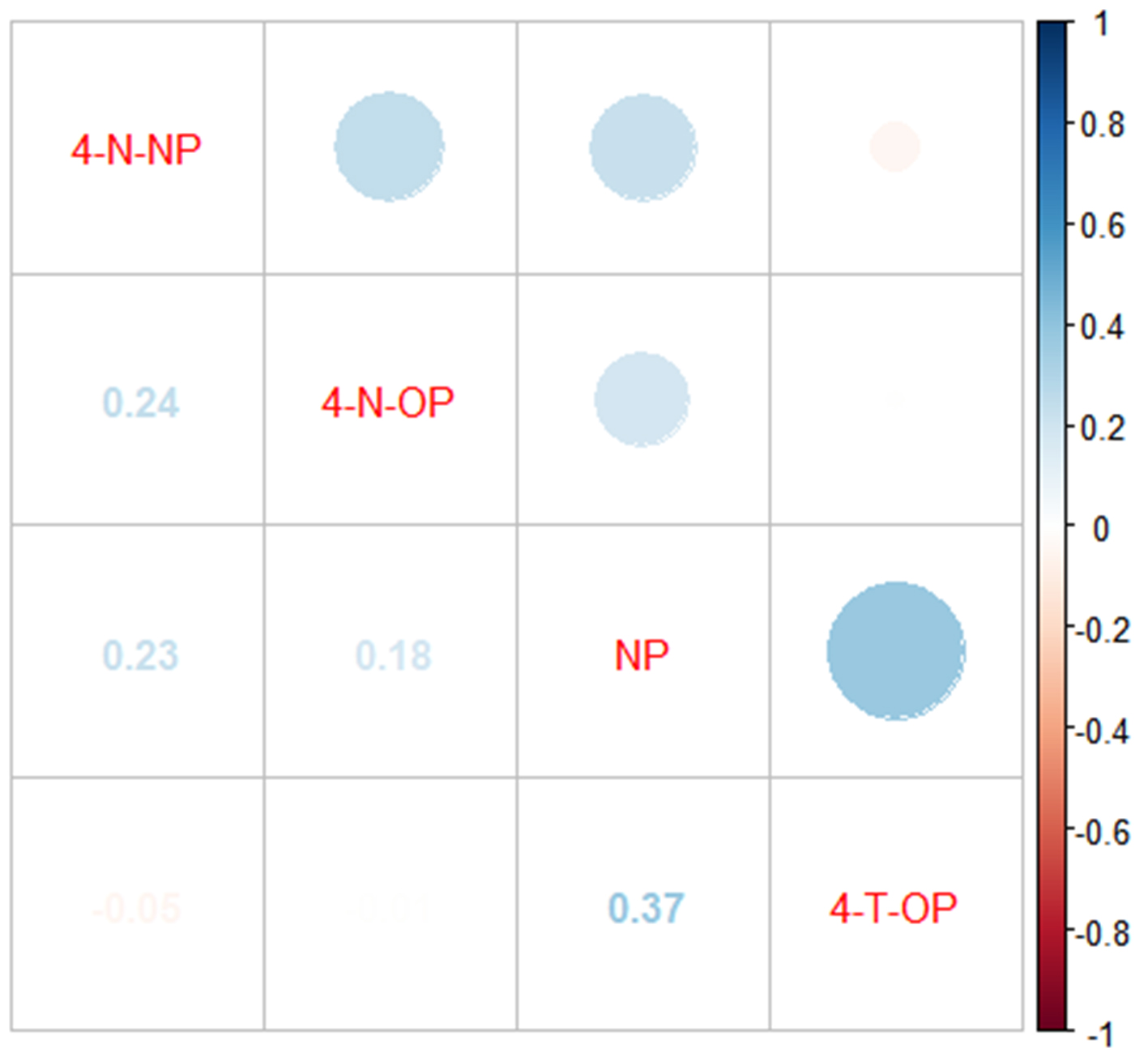
The distribution of APs in maternal serum is summarized in Supplementary Tables 3 and 4. All four APs were detected in the serum samples, with NP exhibiting the highest detection frequency (97.71%) and 4-N-NP the lowest (60.0%) [Supplementary Table 3]. Among the 175 maternal serum samples analyzed, the median concentrations were 110.84 ng/mL for NP, 1.74 ng/mL for 4-N-NP, 48.15 ng/mL for 4-T-OP, and 0.85 ng/mL for 4-N-OP [Supplementary Table 4]. No significant differences were observed in serum AP concentrations between mothers of boys and girls (P > 0.05; Supplementary Table 4). As shown in Figure 2, Spearman correlation analysis revealed predominantly weak to moderate associations among the APs, with correlation coefficients ranging from -0.048 to 0.374. The internal consistency of each domain of the ASQ is presented in Supplementary Table 5. Significant positive correlations were observed across the ASQ domains, with correlation coefficients ranging from 0.18 to 0.37 (all P < 0.05).
Single-pollutant associations with neurodevelopmental outcomes
The associations between maternal log10-transformed AP concentrations and ASQ scores in children are presented in Table 4. After adjustment for covariates, moderate AP exposure was significantly linked to reduced developmental scores among children whose mothers received folic acid supplementation during pregnancy. Specifically, NP exposure was associated with lower gross motor scores (β = -3.032, 95%CI: -6.059, -0.005), while 4-N-NP concentrations were related to decreased communication scores (β = -4.28, 95%CI: -7.402, -1.159). Higher exposure to 4-T-OP was significantly correlated with lower fine motor scores (β = -5.835, 95%CI: -10.879, -0.792). Sex-stratified analyses revealed stronger associations in boys. Maternal exposure to higher NP levels during pregnancy was associated with reductions in both communication (β = -4.65, 95%CI: -8.953, -0.348) and personal-social (β = -4.463, 95%CI: -8.872, -0.054) scores in boys. Elevated 4-N-NP exposure also correlated with lower communication scores in boys (β = -5.369, 95%CI: -10.562, -0.177). Notably, after adjusting for multiple comparisons using the FDR method, these associations did not retain statistical significance (all FDR-adjusted P > 0.05). However, the overall inverse trends and effect directions remained consistent.
Association between maternal serum AP concentration (ng/mL; tertiles) and children’s ASQ scores (n = 175)
| APs | Communication β (95%CI) | Gross motor β (95%CI) | Fine motor β (95%CI) | Problem-solving β (95%CI) | Personal-social β (95%CI) |
| Total | |||||
| NP | -1.714 (-3.496, 0.067) | -1.253 (-3.079, 0.572) | -0.724 (-3.828, 2.381) | -0.274 (-2.145, 1.598) | -0.977 (-2.666, 0.713) |
| P value | 0.065 | 0.186 | 0.584 | 0.749 | 0.263 |
| q-value | 0.554 | 0.910 | 0.910 | 0.966 | 0.910 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -1.084 (-4.060, 1.891) | -3.032 (-6.059, -0.005) | -2.164 (-7.299, 2.970) | 1.189 (-1.932, 4.311) | -1.697 (-4.505, 1.112) |
| P value | 0.421 | 0.063 | 0.442 | 0.547 | 0.219 |
| q-value | 0.910 | 0.554 | 0.910 | 0.910 | 0.910 |
| Tertile3 | -2.873 (-5.863, 0.117) | -2.336 (-5.377, 0.706) | -4.887 (-10.047, 0.272) | 0.978 (-2.159, 4.114) | -2.367 (-5.189, 0.456) |
| P value | 0.061 | 0.150 | 0.055 | 0.601 | 0.100 |
| q-value | 0.554 | 0.899 | 0.554 | 0.910 | 0.670 |
| 4-N-NP | 0.164 (-5.419, 5.747) | 1.388 (-4.298, 7.074) | -1.454 (-11.081, 8.172) | 0.156 (-5.646, 5.958) | 1.864 (-3.385, 7.114) |
| P value | 0.983 | 0.594 | 0.848 | 0.987 | 0.505 |
| q-value | 0.987 | 0.910 | 0.978 | 0.987 | 0.910 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -4.280 (-7.402, -1.159) | -1.645 (-4.885, 1.595) | -4.025 (-9.494, 1.444) | -1.869 (-5.171, 1.432) | -1.229 (-4.230, 1.771) |
| P value | 0.006 | 0.287 | 0.226 | 0.297 | 0.401 |
| q-value | 0.364 | 0.910 | 0.910 | 0.910 | 0.910 |
| Tertile3 | -1.598 (-4.454, 1.258) | 0.135 (-2.829, 3.099) | -0.919 (-5.922, 4.084) | 0.005 (-3.016, 3.025) | -0.724 (-3.468, 2.021) |
| P value | 0.242 | 0.957 | 0.915 | 0.928 | 0.579 |
| q-value | 0.910 | 0.987 | 0.987 | 0.987 | 0.910 |
| 4-T-OP | -0.403 (-1.724, 0.918) | -0.351 (-1.698, 0.996) | -0.793 (-3.071, 1.484) | -0.533 (-1.905, 0.839) | -0.548 (-1.791, 0.694) |
| P value | 0.602 | 0.661 | 0.377 | 0.396 | 0.409 |
| q-value | 0.910 | 0.925 | 0.910 | 0.910 | 0.910 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | 0.669 (-2.307, 3.645) | -0.578 (-3.614, 2.457) | -5.835 (-10.879, -0.792) | 0.243 (-2.854, 3.339) | -0.443 (-3.249, 2.363) |
| P value | 0.607 | 0.759 | 0.015 | 0.943 | 0.784 |
| q-value | 0.910 | 0.966 | 0.451 | 0.987 | 0.966 |
| Tertile3 | -0.480 (-3.498, 2.538) | 0.273 (-2.805, 3.352) | -0.620 (-5.734, 4.494) | 0.731 (-2.409, 3.871) | -0.677 (-3.522, 2.169) |
| P value | 0.792 | 0.821 | 0.713 | 0.686 | 0.659 |
| q-value | 0.966 | 0.966 | 0.950 | 0.936 | 0.925 |
| 4-N-OP | 1.985 (-3.348, 7.317) | 1.737 (-3.700, 7.174) | 3.581 (-5.615, 12.777) | 2.174 (-3.367, 7.715) | 1.083 (-3.945, 6.110) |
| P value | 0.456 | 0.422 | 0.568 | 0.579 | 0.663 |
| q-value | 0.910 | 0.910 | 0.910 | 0.910 | 0.925 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -0.152 (-3.138, 2.834) | -1.521 (-4.486, 1.443) | -4.207 (-9.315, 0.902) | 1.723 (-1.350, 4.797) | -1.305 (-4.110, 1.499) |
| P value | 0.912 | 0.357 | 0.096 | 0.319 | 0.359 |
| q-value | 0.987 | 0.910 | 0.670 | 0.910 | 0.910 |
| Tertile3 | 0.251 (-2.823, 3.326) | 3.065 (0.041, 6.089) | -2.694 (-7.954, 2.565) | -1.018 (-4.183, 2.146) | -0.342 (-3.230, 2.545) |
| P value | 0.886 | 0.049 | 0.288 | 0.439 | 0.811 |
| q-value | 0.987 | 0.554 | 0.910 | 0.910 | 0.966 |
| Boys | |||||
| NP | -2.411 (-4.902, 0.080) | -1.240 (-4.025, 1.545) | -2.239 (-6.695, 2.216) | 0.162 (-2.326, 2.649) | -1.511 (-4.082, 1.060) |
| P value | 0.071 | 0.378 | 0.276 | 0.894 | 0.257 |
| q-value | 0.711 | 0.881 | 0.881 | 0.958 | 0.881 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -1.005 (-5.376, 3.366) | -3.454 (-8.309, 1.402) | -2.693 (-10.494, 5.108) | 3.688 (-0.642, 8.018) | -1.864 (-6.343, 2.615) |
| P value | 0.547 | 0.190 | 0.581 | 0.136 | 0.412 |
| q-value | 0.894 | 0.859 | 0.894 | 0.859 | 0.881 |
| Tertile3 | -4.650 (-8.953, -0.348) | -4.224 (-9.003, 0.556) | -6.541 (-14.219, 1.137) | 1.720 (-2.542, 5.982) | -4.463 (-8.872, -0.054) |
| P value | 0.034 | 0.085 | 0.096 | 0.440 | 0.048 |
| q-value | 0.711 | 0.723 | 0.723 | 0.881 | 0.711 |
| 4-N-NP | 0.335 (-7.411, 8.080) | -1.278 (-9.788, 7.232) | -6.958 (-20.512, 6.597) | -1.182 (-8.746, 6.382) | 1.958 (-5.917, 9.833) |
| P value | 0.968 | 0.816 | 0.365 | 0.641 | 0.627 |
| q-value | 0.968 | 0.927 | 0.881 | 0.897 | 0.897 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -5.369 (-10.562, -0.177) | -1.088 (-6.926, 4.749) | -6.550 (-15.802, 2.702) | -3.035 (-8.196, 2.126) | -0.882 (-6.299, 4.534) |
| P value | 0.035 | 0.725 | 0.198 | 0.223 | 0.735 |
| q-value | 0.711 | 0.927 | 0.859 | 0.859 | 0.927 |
| Tertile3 | -2.313 (-6.445, 1.820) | -1.598 (-6.244, 3.047) | -4.065 (-11.427, 3.298) | -1.188 (-5.295, 2.919) | -0.847 (-5.157, 3.464) |
| P value | 0.229 | 0.506 | 0.332 | 0.533 | 0.684 |
| q-value | 0.859 | 0.894 | 0.881 | 0.894 | 0.911 |
| 4-T-OP | 0.838 (-1.255, 2.932) | 0.238 (-2.072, 2.548) | -1.227 (-4.919, 2.464) | -0.597 (-2.647, 1.453) | 0.457 (-1.680, 2.595) |
| P value | 0.388 | 0.792 | 0.428 | 0.479 | 0.643 |
| q-value | 0.881 | 0.927 | 0.881 | 0.894 | 0.897 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -1.192 (-5.785, 3.402) | -0.198 (-5.284, 4.889) | -7.016 (-15.004, 0.972) | -1.629 (-6.114, 2.856) | 1.963 (-2.725, 6.651) |
| P value | 0.664 | 0.966 | 0.059 | 0.327 | 0.376 |
| q-value | 0.906 | 0.968 | 0.711 | 0.881 | 0.881 |
| Tertile3 | 1.255 (-3.370, 5.881) | -0.037 (-5.159, 5.085) | -2.042 (-10.086, 6.002) | 0.841 (-3.676, 5.357) | 1.833 (-2.887, 6.554) |
| P value | 0.575 | 0.944 | 0.562 | 0.842 | 0.424 |
| q-value | 0.894 | 0.968 | 0.894 | 0.927 | 0.881 |
| 4-N-OP | 3.741 (-4.151, 11.632) | 1.739 (-6.974, 10.452) | 1.570 (-12.396, 15.536) | 0.262 (-7.490, 8.014) | 0.281 (-7.797, 8.359) |
| P value | 0.375 | 0.600 | 0.850 | 0.838 | 0.920 |
| q-value | 0.881 | 0.897 | 0.927 | 0.927 | 0.968 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | 1.218 (-3.439, 5.876) | -2.525 (-7.492, 2.443) | -7.177 (-15.217, 0.864) | 1.584 (-2.870, 6.038) | -0.602 (-5.350, 4.146) |
| P value | 0.576 | 0.346 | 0.068 | 0.547 | 0.822 |
| q-value | 0.894 | 0.881 | 0.711 | 0.894 | 0.927 |
| Tertile3 | 0.873 (-3.640, 5.386) | 2.872 (-1.942, 7.685) | -6.027 (-13.818, 1.764) | -2.488 (-6.804, 1.828) | -0.614 (-5.214, 3.987) |
| P value | 0.768 | 0.219 | 0.150 | 0.207 | 0.786 |
| q-value | 0.927 | 0.859 | 0.859 | 0.859 | 0.927 |
| Girls | |||||
| NP | -0.570 (-3.366, 2.225) | -1.342 (-3.822, 1.137) | 2.897 (-1.882, 7.675) | -0.090 (-3.143, 2.962) | 0.255 (-1.721, 2.231) |
| P value | 0.653 | 0.335 | 0.242 | 0.877 | 0.995 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.995 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -0.635 (-5.073, 3.802) | -2.379 (-6.303, 1.545) | -1.197 (-8.853, 6.459) | 0.560 (-4.281, 5.400) | -0.750 (-3.847, 2.348) |
| P value | 0.745 | 0.263 | 0.702 | 0.888 | 0.535 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile3 | -0.040 (-4.517, 4.438) | -0.822 (-4.781, 3.137) | -1.316 (-9.040, 6.409) | 0.498 (-4.386, 5.382) | 1.293 (-1.832, 4.419) |
| P value | 0.925 | 0.808 | 0.657 | 0.973 | 0.590 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| 4-N-NP | -1.654 (-10.634, 7.326) | 3.755 (-4.226, 11.735) | 7.798 (-7.598, 23.193) | -1.011 (-10.811, 8.789) | -0.039 (-6.388, 6.309) |
| P value | 0.725 | 0.356 | 0.294 | 0.860 | 0.969 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -4.190 (-8.411, 0.031) | -1.682 (-5.463, 2.099) | -1.481 (-8.885, 5.922) | 0.244 (-4.495, 4.984) | -0.835 (-3.899, 2.229) |
| P value | 0.062 | 0.316 | 0.792 | 0.818 | 0.678 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile3 | -1.527 (-5.881, 2.828) | 2.096 (-1.805, 5.996) | 3.607 (-4.031, 11.244) | 0.772 (-4.117, 5.661) | -0.642 (-3.803, 2.519) |
| P value | 0.569 | 0.381 | 0.266 | 0.603 | 0.908 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| 4-T-OP | -1.203 (-3.015, 0.608) | -0.759 (-2.389, 0.871) | 0.394 (-2.772, 3.561) | 0.064 (-1.937, 2.066) | -0.578 (-1.866, 0.711) |
| P value | 0.173 | 0.406 | 0.946 | 0.950 | 0.337 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | 2.857 (-1.278, 6.993) | -1.369 (-5.186, 2.448) | -2.346 (-9.576, 4.885) | 2.516 (-2.137, 7.169) | -1.962 (-4.963, 1.039) |
| P value | 0.172 | 0.454 | 0.420 | 0.300 | 0.255 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile3 | -2.003 (-6.546, 2.540) | 0.529 (-3.664, 4.722) | 4.731 (-3.212, 12.675) | 1.852 (-3.259, 6.964) | -0.912 (-4.208, 2.385) |
| P value | 0.361 | 0.766 | 0.310 | 0.531 | 0.580 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| 4-N-OP | 0.072 (-7.932, 8.077) | 1.606 (-5.537, 8.748) | 5.466 (-8.284, 19.215) | 1.453 (-7.269, 10.175) | 0.708 (-4.942, 6.359) |
| P value | 0.961 | 0.575 | 0.528 | 0.852 | 0.926 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile1 | Ref | Ref | Ref | Ref | Ref |
| Tertile2 | -1.282 (-5.422, 2.857) | -0.741 (-4.376, 2.895) | -1.683 (-8.835, 5.469) | 1.383 (-3.125, 5.892) | -0.910 (-3.830, 2.011) |
| P value | 0.529 | 0.724 | 0.658 | 0.566 | 0.433 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
| Tertile3 | -0.604 (-5.155, 3.947) | 2.498 (-1.500, 6.495) | -0.233 (-8.096, 7.630) | -0.259 (-5.216, 4.698) | 0.114 (-3.096, 3.325) |
| P value | 0.733 | 0.179 | 0.825 | 0.804 | 0.913 |
| q-value | 0.989 | 0.989 | 0.989 | 0.989 | 0.989 |
Sensitivity analyses excluding maternal folic acid supplementation confirmed that these associations remained stable [Supplementary Table 6], supporting the robustness of the findings. In summary, prenatal exposure to moderate or high concentrations of NP, 4-N-NP, and 4-T-OP was associated with lower neurodevelopmental scores across multiple domains in children aged 3-6 years, with stronger effects observed in boys. No significant associations were found between 4-N-OP exposure and any ASQ domain. Although statistical significance was attenuated after FDR correction, the results remained robust in sensitivity analyses and showed consistent sex-specific patterns, reinforcing the reliability of the observed trends.
Non-linear and mixture effects of APs
RCS models were used to examine potential nonlinear relationships between prenatal AP exposures and neurodevelopmental domain scores. Overall, no significant associations were observed between AP mixtures and ASQ scores (P-overall > 0.5). Evidence of nonlinearity was observed for 4-N-NP in the communication domain (P-nonlinear = 0.021), and for NP and 4-N-OP in the fine motor domain (P-nonlinear = 0.036 and 0.042, respectively) [Supplementary Figures 2-4]. However, these findings should be interpreted cautiously because the overall associations were not consistently statistically significant across domains or models. Therefore, the RCS plots are presented for exploratory purposes only. No significant global or nonlinear associations were observed for other domains, including gross motor, personal-social, and problem-solving (all P-overall > 0.05).
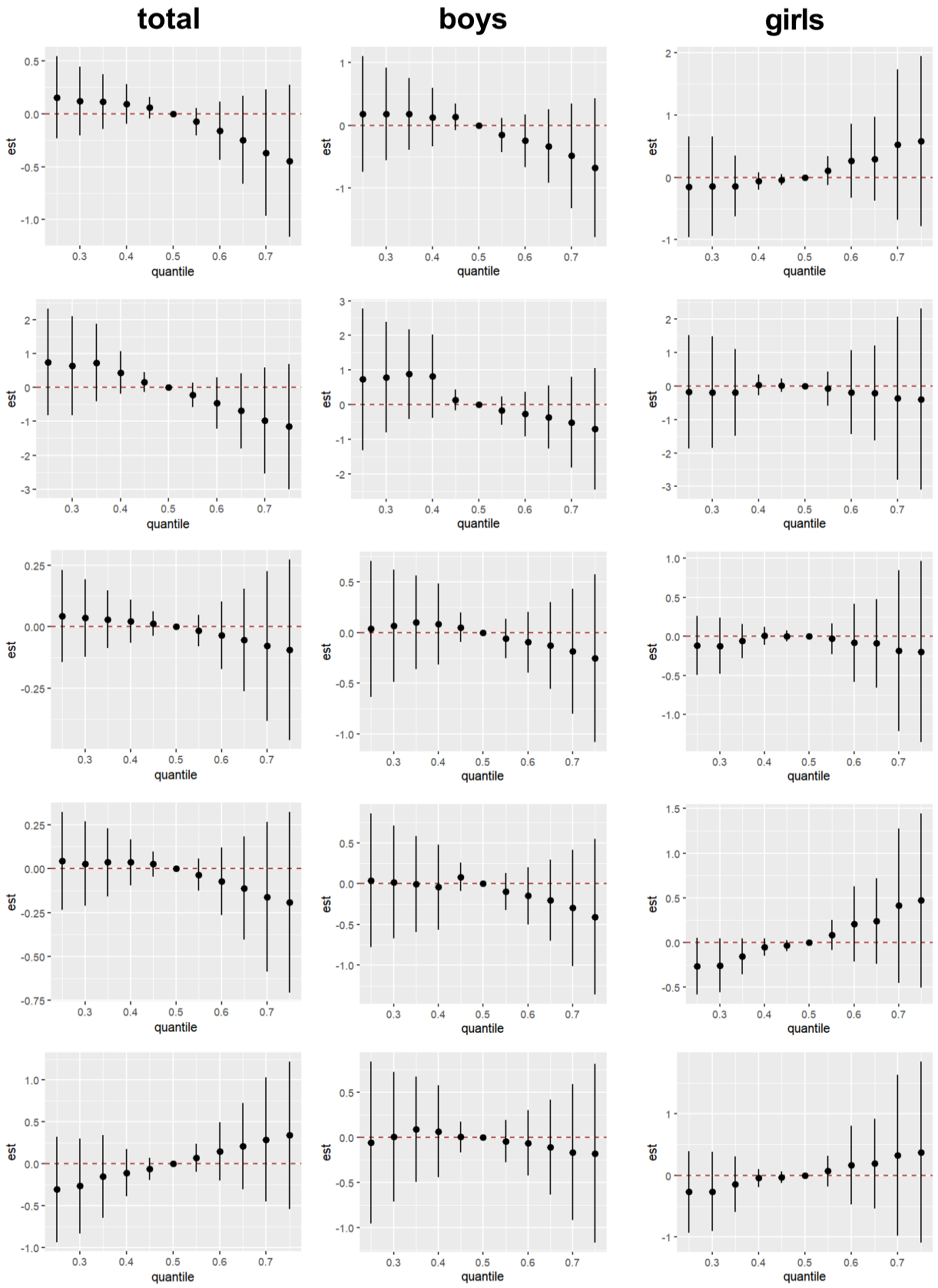
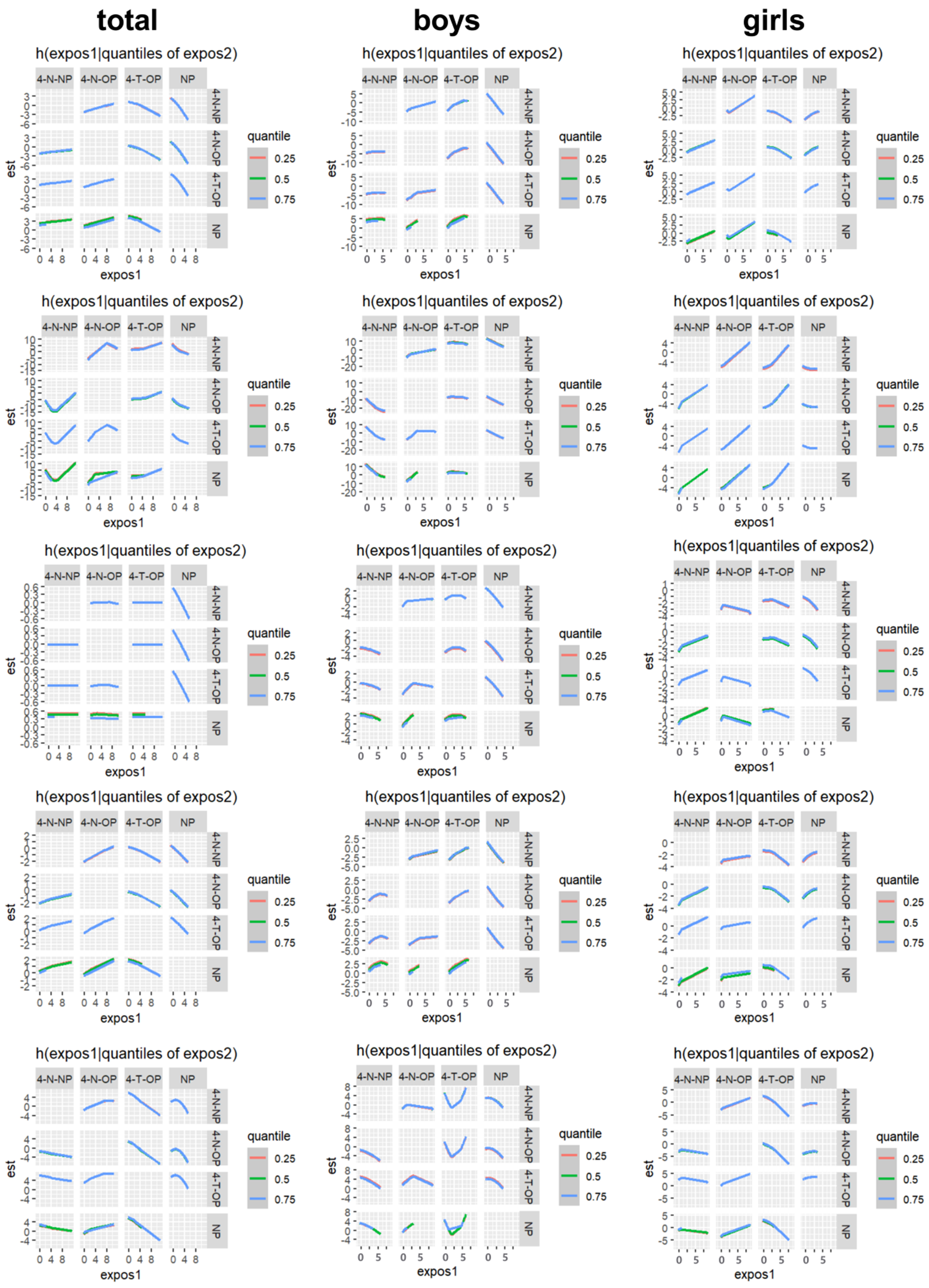
No significant combined effects of AP mixtures on children’s ASQ scores were observed in this study [Figures 3 and 4, Supplementary Figures 5 and 6]. Relative to the 50th percentile of exposure across all four APs, mixtures above this threshold were associated with declining scores in the communication, gross motor, fine motor, and personal-social domains, while an upward trend was noted in problem-solving ability. Sex-stratified analyses revealed more pronounced effects in boys. Among mothers exposed to AP levels above the 50th percentile during pregnancy, boys exhibited declines across all domains - communication, gross motor, fine motor, personal-social, and problem-solving. In contrast, among mothers with similar exposure levels, girls showed reductions in gross and fine motor scores, while communication, personal-social, and problem-solving domains demonstrated upward trends. To identify the primary drivers of these patterns, posterior inclusion probabilities derived from the BKMR model were assessed [Supplementary Table 7]. These analyses revealed distinct sex-specific mixture effects: NP was the predominant contributor to neurodevelopmental alterations in the total population and in boys, whereas 4-T-OP was the primary driver in girls.
Figure 3. Combined effects of maternal serum APs on ASQ scores in the total population, boys, and girls during childhood. AP concentrations were log-transformed. Black circles represent estimated effects, black vertical lines represent 95%CIs, and red dashed lines represent the null value. From top to bottom, the domains are: Communication, Fine motor, Gross motor, Personal-social, and Problem-solving. APs: Alkylphenols; ASQ: Ages and Stages Questionnaire; 95%CIs: 95% confidence intervals.
Figure 4. Bivariate exposure–response functions between each pair of maternal serum APs and children’s ASQ scores for the total population, boys, and girls, with all other serum AP indicators fixed at the 25th, 50th, and 75th percentiles. From top to bottom, the domains are: Communication, Fine motor, Gross motor, Personal-social, and Problem-solving. APs: Alkylphenols; ASQ: Ages and Stages Questionnaire; 4-N-NP: 4-nonyl-nonylphenol; 4-N-OP: 4-nonyl-octylphenol; 4-T-OP: 4-tert-octylphenol; NP: nonylphenol.
The epidemiological review included three Asian cohort studies and one prior study conducted by the authors. The mean age of pregnant women in these studies ranged from 23.90 to 39 years, with a mean gestational age of ≥ 38 weeks. The outcomes covered behavioral disorders, cognitive dysfunction, language impairment, and other neurodevelopmental delays. Based on the Joanna Briggs Institute (JBI) criteria, one study was rated as high quality and three as moderate quality [Supplementary Tables 8-15]. NP and OP were the most frequently investigated compounds, with evidence suggesting that all four APs (NP, 4-N-NP, 4-T-OP, 4-N-OP) are associated with neurodevelopmental impairments in children. Assessments primarily relied on the WPPSI-IV, Wechsler Intelligence Scale, Bayley-III, and CDIIT.
As summarized in Table 1, Lin et al. examined the associations between umbilical cord blood levels of NP and OP and neurodevelopment at ages 2 and 7 years, with detection rates of 77.6% and 68.3%, respectively[25]. However, no significant associations were observed in CDIIT or WISC-IV evaluations. Wang et al. reported higher urinary NP concentrations in children aged 4-6 years compared to those aged 2-3 years[31]. Boys in the highest exposure quartile had significantly lower full-scale intelligence quotient (FSIQ) scores compared to the reference group (β = -11.29, 95%CI: -18.62, -3.96, P < 0.05). Long et al., a prior investigation within the same project, identified nonmonotonic and sex-specific effects of prenatal AP exposure on IQ in preschool children[26]. In GLMs, the second quartile of NP was negatively associated with FSIQ (β = -2.38, 95%CI: -4.69, -0.07), while the second quartile of 4-T-OP was negatively associated with the working memory index (WMI) (β = -5.24, 95%CI: -9.58, -0.89). In girls, the third quartile of 4-N-NP correlated positively with the fluid reasoning index (β = 4.95, 95%CI: 1.14, 8.77). RCS models indicated a U-shaped relationship between 4-T-OP and WMI among all children and girls (P for nonlinearity < 0.05). BKMR analysis suggested that the overall mixture effect above the 50th percentile was primarily driven by NP and exhibited opposite trends for FSIQ in the overall cohort vs. girls.
Evidence from in vivo and in vitro studies
A total of 19 animal studies were included, comprising 4 mouse studies and 15 rat studies. In all cases, animals were exposed to APs throughout gestation, and offspring neurodevelopment was assessed via behavioral tests [Supplementary Table 2]. Among these studies, eight evaluated neurobehavioral and memory functions using behavioral experiments, while seven reported AP-induced hippocampal neurotoxicity. Fang et al. and Yao et al. observed impaired fear memory in offspring following AP exposure[32,33]. In a more comprehensive approach, Li et al. integrated behavioral assessments with molecular analyses to investigate the underlying neurotoxic mechanisms[19]. Several studies focused on hippocampal involvement: Qiu et al. and Lee et al. demonstrated that APs induce microglial overactivation and promote apoptosis via oxidative stress and inflammatory pathways[34,35]. Cerebellar pathology was also observed. You et al. and Negishi et al. found that APs inhibit dendritic growth of Purkinje cells, disrupt protein kinase C γ (PKCγ) phosphorylation, and interfere with brain-derived neurotrophic factor (BDNF)/tropomyosin receptor kinase B (TrkB) signaling, ultimately impairing motor coordination and learning[12,36]. Fang et al. further showed that APs overactivate the Notch2 pathway, leading to excessive proliferation of granule cell precursors and hindering cerebellar development[32]. Negishi et al. also indicated that perinatal AP exposure disrupts cerebellar myelination via the BMP4 pathway[12]. Additional mechanistic insights were provided by Jie et al., who assessed neuronal development by measuring apoptosis rates[37], and Jie et al., who linked combined AP exposure to cholinergic disruption and sex hormone imbalance, resulting in delayed reflex development and aberrant hippocampal enzyme activity[38]. Furthermore, Takagi et al. and Ferguson et al. explored neurodevelopmental toxicity related to sexual differentiation[39,40]. Notably, Kawaguchi et al. reported that low-dose NP exposure during the perinatal period improved spatial cognition, suggesting that endocrine disruptors may exert non-monotonic, dose-dependent effects and may enhance functionality under certain conditions[41].
Table 2 summarizes ten in vitro studies employing various cellular models. Kim et al. and Tran et al. used amphibian neural crest cells (NCCs) to investigate the roles of oxidative damage and apoptosis in neurodevelopment[42,43]. Qiu et al. and Lee et al. further linked hippocampal microglial proliferation to alterations in protein and gene expression associated with specific signaling pathways and neurobehavioral impairments, thereby elucidating potential molecular mechanisms[8,34,35].
In neuronal culture models, Tran et al. and Litwa et al. observed that APs induced dendritic and axonal malformations, disrupted receptor-mediated signaling, and suppressed neuronal proliferation[43,44]. These structural and functional abnormalities interfered with brain development and led to behavioral alterations in mice. Similarly, Xu et al. reported that APs inhibited the Sonic hedgehog (Shh) signaling pathway in NE-4C cells, reducing cell proliferation and depleting neural progenitor pools[45]. Additionally, Kim et al. assessed the cytotoxic effects of APs in human embryonic stem cells, examining their associations with apoptosis-related proteins and signaling cascades[42]. Overall, these in vitro findings consistently demonstrate the neurodevelopmental toxicity of APs from multiple mechanistic perspectives.
TRAEC-based risk assessment
Under the TRAEC strategy framework, this study integrated epidemiological, in vivo, and in vitro experimental data, along with independent research, to establish a risk assessment model for the impact of prenatal AP exposure on offspring neurodevelopment. The model comprehensively and quantitatively evaluated reliability, weight of concentration, risk intensity, and correlation [Supplementary Figure 7]. Through multidimensional evidence integration, a hierarchical evaluation system based on the TRAEC strategy was developed. A systematic literature search identified 13 epidemiological studies, 21 animal experiments, and 10 in vitro studies. This collection was supplemented with original data from a prospective cohort study conducted by our team, resulting in a comprehensive evidence pool of 44 data entries.
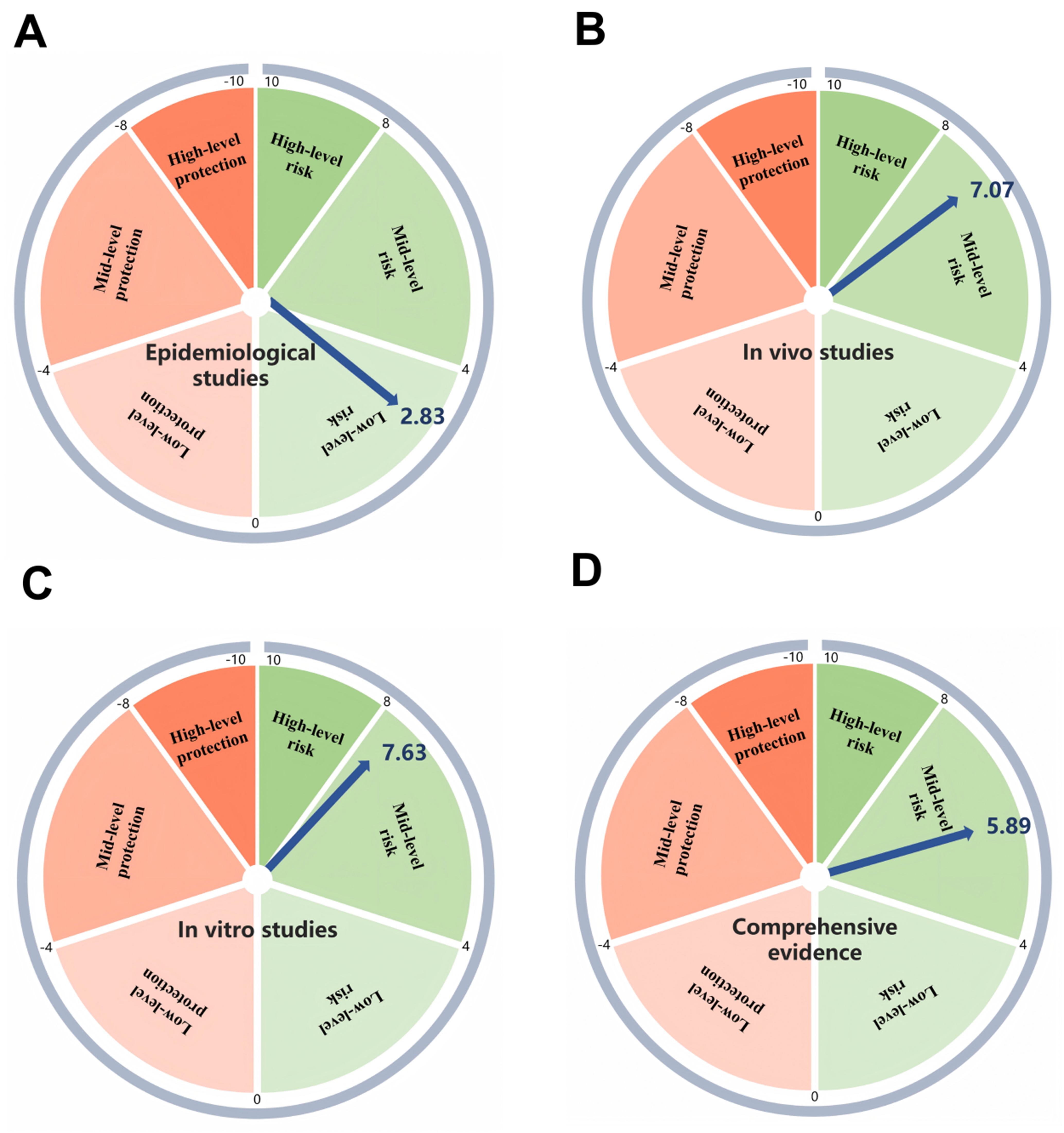
The TRAEC risk matrix[46-49] categorizes health risk into three tiers: low (0-4], medium (4-8], and high (8-10), with protective effects similarly classified as low, medium, or high. As a case study, 44 pieces of evidence concerning 9 APs were evaluated. Among these, NP was the most frequently documented compound (16 instances), followed by 4-N-NP (10 instances), 4-T-OP (7 instances), OP (2 instances), and 4-N-OP (2 instances). The highest risk score (8.5, high risk) was assigned to 4-P-NP, while OP had the lowest (2.5, low risk). One study indicated a moderate protective effect for 4-NP (score: -4.9). Based on the risk classification, two APs were categorized as low risk (4-N-OP, OP), six as medium risk (NP, 4-N-NP, 4-T-OP, 4-OP, P-NP, 4-NP), one as high risk (4-P-NP), and one exhibited a low-level protective effect (4-NP). NP, with a risk score of 6.44, was the most frequently studied compound [Supplementary Figure 8]. The overall mean risk score across all included studies was 5.91, indicating a moderate risk of reproductive neurotoxicity from prenatal AP exposure [Figure 5].
Figure 5. Risk assessment of all evidence based on the TRAEC strategy. (A) Scores of epidemiological studies; (B) Scores of in vivo studies; (C) Scores of in vitro studies; (D) Comprehensive evidence scores. TRAEC: Targeted Risk Assessment of Environmental Chemicals.
Comparison with conventional validation methods
To validate its robustness, the TRAEC risk assessment was compared with conventional approaches [Supplementary Tables 8-15]. Based on the ToxRTool evaluation, 7 in vivo studies were classified as grade I and 14 as grade II, while among the in vitro experiments, 9 out of 10 were rated grade I and 1 grade II. Using the SciRAP tool, quality scores for in vivo studies ranged from 43.75 to 76.67, and for in vitro assays, from 58.33 to 79.17. According to the OHAT risk-of-bias assessment, 24 items were judged to be of low risk, 4 as medium risk, and 4 as having insufficient information. Additionally, the IRIS evaluation indicated that the epidemiological studies were of moderate risk. Overall, the TRAEC approach demonstrated consistency with traditional methods and exhibited robust performance.
Discussion of potential mechanisms and sex-specific effects
This study employed the TRAEC strategy, a comprehensive and quantitative framework for structured evidence synthesis[24,46-49], to systematically assess the neurodevelopmental risks associated with prenatal AP exposure. By integrating an original epidemiological dataset with 28 published studies, the multidimensional scoring system identified a moderate overall neurotoxic risk (score: 5.91/10). The nine evaluated APs were ranked in descending order of risk (4-P-NP > 4-N-NP > P-NP > 4-OP > 4-T-OP > NP > OP > 4-N-OP > 4-NP), with 4-T-OP, 4-N-NP, and NP supported by the most robust evidence. Importantly, the reliability of the TRAEC-derived conclusions was strongly reinforced by high concordance with four established evaluation tools (ToxRTool, SciRAP, OHAT, and IRIS)[29].
Epidemiological analyses, aligned with the broader evidence synthesis, revealed complex NMDR relationships and sex-specific vulnerabilities. Prenatal exposure to EDCs has long-lasting, sometimes irreversible effects on offspring development[19,50,51], and our findings support the plausibility of a link between gestational AP exposure and altered neurodevelopmental trajectories in children. The synthesis of 28 studies, complemented by original data, highlights the considerable complexity of AP-related neurodevelopmental effects. Most studies reported inverse associations between prenatal AP exposure and neurodevelopmental outcomes, consistent with existing evidence and reinforcing the classification of APs as probable neurodevelopmental toxicants. Notably, one study identified a positive association between low-dose 4-NP exposure and neurodevelopment[41], suggesting that endocrine disruptors may induce non-monotonic responses, highlighting the complexity of their biological mechanisms.
The underlying mechanisms are likely multifaceted. As environmental endocrine disruptors, APs exhibit weak estrogenic activity[52]. Estrogen itself enhances learning and memory, promotes neuroprotection, and supports dendritic spine formation and long-term potentiation - key processes underlying cognitive function. Experimental data show that NP binds to estrogen receptors (ERs), interfering with the development and regulation of the endocrine reproductive system[53,54]. Even low-dose AP exposure may transiently activate neuroprotective or plasticity-related signaling cascades, such as the BDNF/TrkB pathway, and, under specific conditions, trigger stimulatory or functionally promotive responses.
Exposure to EDCs during prenatal and postnatal periods can also influence developmental trajectories across the lifespan[55]. Dynamic interactions between environmental exposures and genetic predispositions may occur through epigenetic modifications, altering neurodevelopmental pathways. Discrepancies across studies may arise from variations in exposure timing, behavioral assessment tools, or animal models, each potentially shaping the observed outcomes. Furthermore, the seemingly “beneficial” cognitive effects at low doses may reflect subtle modulation of the neuroendocrine system by EDCs during sensitive developmental windows. During these periods, minor perturbations to endogenous hormone signaling could transiently enhance specific cognitive functions. However, the reproducibility, persistence, and potential trade-offs of these apparent benefits remain unclear and warrant further investigation. Future studies should prioritize elucidating the mechanisms underlying low-dose EDC effects and their implications for chemical risk assessment.
Our finding of sex-specific neurodevelopmental vulnerabilities, particularly the more pronounced effects in boys, aligns with the well-documented sexually dimorphic actions of EDCs[56]. Biologically, male fetal brain masculinization depends on a critical prenatal androgen surge. However, as potent synthetic xenoestrogens, APs, such as NP, can severely disrupt this delicate steroid environment. Recent evidence indicates that NP induces significant endocrine disruption and oxidative stress by binding to ERs (e.g., upregulating ERα and downregulating ERβ), skewing the hormonal balance toward estrogenic over androgenic activity[57]. This pronounced estrogenic and anti-androgenic interference disrupts the necessary prenatal androgen surge, thereby altering the typical male neurodevelopmental trajectory. Additionally, the generation of reactive oxygen species (ROS) induced by APs exacerbates neurodevelopmental toxicity. In contrast, female brains, which develop largely independent of this early androgen surge, may be relatively protected from these specific hormone-mediated disruptions.
Strengths and limitations
Despite the rigorous framework, several limitations warrant caution. First, exposure misclassification and reduced analytical sensitivity are primary concerns. In our epidemiological cohort, maternal serum was collected at a single time point during early pregnancy, and only the parent AP compounds were quantified, excluding their biotransformation metabolites. As commercial APs undergo rapid in vivo metabolism and clearance, circulating concentrations of the parent compounds in serum are often low. This, combined with single-time-point sampling, reduces measurement sensitivity and may not fully capture the cumulative exposure burden during critical windows of fetal neurodevelopment (including the second and third trimesters)[58,59]. Such temporal misclassification typically biases effect estimates toward the null.
Furthermore, within our systematic review, the TRAEC scoring synthesized data across varying exposure windows (e.g., early pregnancy, late pregnancy, and cord blood). While our standardized weighting approach accounts for the overall study reliability, the lack of strict stratification by specific gestational windows limits the direct comparability across studies and represents a limitation in the current evidence integration. Second, our biomarker scope was restricted. We quantitatively measured only four AP parent compounds (NP, 4-N-NP, 4-T-OP, and 4-N-OP). Technical mixtures of APs in commercial products comprise a highly complex array of branched isomers. Relying on this restricted panel likely underestimates total exposure and limits our ability to evaluate the effects of complex mixtures or synergistic interactions. Third, although the Benjamini-Hochberg procedure was applied to control the FDR, some sex-specific associations lost statistical significance; however, the stable β coefficients suggest that these represent biologically plausible signals rather than stochastic noise. Furthermore, the absence of detailed lifestyle data precludes precise source apportionment, as APs originate from diverse sources, including plastic packaging and personal care products. Finally, while the parent-reported ASQ offers cost-effectiveness for large cohorts, it lacks the diagnostic granularity of clinician-administered tools such as the Bayley Scales, potentially underestimating subtle or subclinical neurodevelopmental shifts induced by AP exposure.
CONCLUSION
This study aimed to evaluate the potential neurodevelopmental risk of prenatal exposure to APs by integrating primary epidemiological data with the published evidence base under the TRAEC strategy (version 1.1). Overall, the integrated evidence indicates that prenatal AP exposure is associated with neurodevelopmental vulnerabilities in offspring. The findings further suggest that risks may differ across individual APs and that susceptibility may vary by sex, underscoring the need for chemical-specific and population-sensitive risk management. By bringing together multiple lines of evidence in a consistent manner, our work provides a robust basis for supporting risk prioritization and strengthening the scientific foundation for environmental health protection. Future studies should validate these findings in larger and more diverse populations and translate the convergent evidence into targeted prevention and regulatory actions to better safeguard maternal and child health.
DECLARATIONS
Acknowledgments
The graphical abstract was created with BioRender (https://www.biorender.com/). The authors sincerely thank the participants for their support, as well as the staff at the collaborating hospitals for their assistance in sample and data collection. The authors also extend sincere thanks to the organizations that supported the laboratory site and provided equipment: the Guangxi Key Laboratory of Environment and Health Research, Guangxi Medical University, and the Guangxi Colleges and Universities Key Laboratory of Prevention and Control of Highly Prevalent Diseases, Guangxi Medical University.
Authors’ contributions
Writing - original draft: Chen, X.; Huang, X.
Writing - review and editing: Ma, X.; Liao, Q.; Pang, L.; Huang, D.; Huang, H.; Liao, T.
Methodology, resources: Ma, X.; Huang, D.
Visualization, formal analysis: Chen, X.; Huang, X.; Wen J.; Li, W.
Investigation, conceptualization: Ma, X.; Liao, Q.; Liang, J.
Data curation: Chen, X.; Huang, X.
Supervision: Liang, J.; Liao, Q.; Huang, H.; Huang, D.
Validation, investigation: Ou, L.; Qin, X.; Li, W.; Huang, Y.; Luo, M.; Tang, L.
Data curation: Chen, X.; Huang, X.
Resources, project administration, funding acquisition, data curation.: Huang, D.
Availability of data and materials
All study data are included in the article.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant number 22366007) and the Guangxi Natural Science Foundation of China (Grant number 2024GXNSFAA010391).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
This study was conducted in accordance with the principles of the Declaration of Helsinki. The study protocol was reviewed and approved by the Ethics Committee of Guangxi Medical University (Approval Number: [No. 20140305-001]). Written informed consent was obtained from all participating mothers prior to enrollment and data collection.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Lee, S. M.; Cheong, D.; Kim, M.; Kim, Y. S. Analysis of endocrine disrupting nonylphenols in foods by gas chromatography-mass spectrometry. Foods 2023, 12, 269.
2. De la Parra-Guerra, A. C.; Acevedo-Barrios, R. Studies of endocrine disruptors: nonylphenol and isomers in biological models. Environ. Toxicol. Chem. 2023, 42, 1439-50.
3. Thi Thu, N.; Thi Quynh Mai, D.; Cam Tu, V.; et al. Occurrence and potential environmental risk assessment of alkylphenols and bisphenols in surface water collected in rivers flowing through Bac Ninh, Vietnam. J. Water. Health. 2024, 22, 1235-47.
4. Gu, W.; Wang, Y.; Qiu, Z.; Dong, J.; Wang, Y.; Chen, J. Maternal exposure to nonylphenol during pregnancy and lactation induces microglial cell activation and pro-inflammatory cytokine production in offspring hippocampus. Sci. Total. Environ. 2018, 634, 525-33.
5. Kazemi, S.; Khalili-Fomeshi, M.; Akbari, A.; Kani, S. N. M.; Ahmadian, S. R.; Ghasemi-Kasman, M. The correlation between nonylphenol concentration in brain regions and resulting behavioral impairments. Brain. Res. Bull. 2018, 139, 190-6.
6. Repková, A.; Mišľanová, C.; Hrabčáková, J.; et al. Relationship between eating habits and 4-nonylphenol concentration in breast milk of women in Slovakia. Life 2023, 13, 2361.
7. Romanelli, A. M.; Montefusco, A.; Sposito, S.; Scafuri, B.; Caputo, I.; Paolella, G. In vitro investigation of biological and toxic effects of 4-octylphenol on human cells. Int. J. Mol. Sci. 2024, 25, 13032.
8. Lee, S. H.; Shin, H. S.; So, Y. H.; et al. Effects of maternal nonylphenol exposure on the proliferation of glial cells in the brain of male offspring mice. Anim. Cells. Syst. 2024, 28, 439-52.
9. Lee, S. H.; Jung, E. M. Adverse effects of early-life stress: focus on the rodent neuroendocrine system. Neural. Regen. Res. 2024, 19, 336-41.
10. Parenti, M.; Slupsky, C. M. Disrupted prenatal metabolism may explain the etiology of suboptimal neurodevelopment: a focus on phthalates and micronutrients and their relationship to autism spectrum disorder. Adv. Nutr. 2024, 15, 100279.
11. Lubrano, C.; Parisi, F.; Cetin, I. Impact of maternal environment and inflammation on fetal neurodevelopment. Antioxidants 2024, 13, 453.
12. Negishi, T.; Kawasaki, K.; Suzaki, S.; et al. Behavioral alterations in response to fear-provoking stimuli and tranylcypromine induced by perinatal exposure to bisphenol A and nonylphenol in male rats. Environ. Health. Perspect. 2004, 112, 1159-64.
13. Wang, L.; Guo, M.; Feng, G.; Wang, P.; Xu, J.; Yu, J. Effects of chronic exposure to nonylphenol at environmental concentration on thyroid function and thyroid hyperplasia disease in male rats. Toxicology 2021, 461, 152918.
14. Yang, Z.; Zhang, J.; Wang, M.; et al. Prenatal endocrine-disrupting chemicals exposure and impact on offspring neurodevelopment: a systematic review and meta-analysis. Neurotoxicology 2024, 103, 335-57.
15. Wang, L.; Miao, J.; Ding, M.; Zhang, W.; Pan, L. Exploring the mechanism of nonylphenol-induced ovarian developmental delay of manila clams, Ruditapes philippinarum: applying RNAi to toxicological analysis. Chemosphere 2024, 356, 141905.
16. Rang, Y.; Liu, H.; Liu, C. The mechanisms of learning and memory impairment caused by nonylphenol: a narrative review based on in vivo and in vitro studies. Environ. Sci. Pollut. Res. Int. 2023, 30, 5530-9.
17. You, M.; Li, S.; Yan, S.; Yao, D.; Wang, T.; Wang, Y. Exposure to nonylphenol in early life causes behavioural deficits related with autism spectrum disorders in rats. Environ. Int. 2023, 180, 108228.
18. Nishimura, Y.; Nagao, T.; Fukushima, N. Long-term pre-exposure of pheochromocytoma PC12 cells to endocrine-disrupting chemicals influences neuronal differentiation. Neurosci. Lett. 2014, 570, 1-4.
19. Li, M.; You, M.; Li, S.; Qiu, Z.; Wang, Y. Effects of maternal exposure to nonylphenol on learning and memory in offspring involve inhibition of BDNF-PI3K/Akt signaling. Brain. Res. Bull. 2019, 146, 270-8.
20. Xu, L. C.; Sun, H.; Chen, J. F.; et al. Evaluation of androgen receptor transcriptional activities of bisphenol A, octylphenol and nonylphenol in vitro. Toxicology 2005, 216, 197-203.
21. Zhang, J.; Liu, L.; Ning, X.; Lin, M.; Lai, X. Isomer-specific analysis of nonylphenol and their transformation products in environment: a review. Sci. Total. Environ. 2023, 901, 165982.
22. Shi, Z.; Xia, M.; Xiao, S.; Zhang, Q. Identification of nonmonotonic concentration-responses in Tox21 high-throughput screening estrogen receptor assays. Toxicol. Appl. Pharmacol. 2022, 452, 116206.
23. Dias, G. R. M.; Giusti, F. C. V.; de Novais, C. O.; et al. Intergenerational and transgenerational effects of endocrine-disrupting chemicals in the offspring brain development and behavior. Front. Endocrinol. 2025, 16, 1571689.
24. Zhu, Y.; Tan, Z.; An, Z.; et al. Effects of per- and polyfluoroalkyl substances on atherosclerosis: an evidence-based risk assessment in the context of the TRAEC strategy. Environ. Res. 2025, 285, 122698.
25. Lin, C. C.; Chien, C. J.; Tsai, M. S.; Hsieh, C. J.; Hsieh, W. S.; Chen, P. C. Prenatal phenolic compounds exposure and neurobehavioral development at 2 and 7years of age. Sci. Total. Environ. 2017, 605-6, 801-10.
26. Long, J.; Liang, J.; Liu, T.; et al. Association between prenatal exposure to alkylphenols and intelligence quotient among preschool children: sex-specific effects. Environ. Health. 2024, 23, 21.
27. van den Dries, M. A.; Guxens, M.; Spaan, S.; et al. Phthalate and bisphenol exposure during pregnancy and offspring nonverbal IQ. Environ. Health. Perspect. 2020, 128, 77009.
28. Bobb, J. F.; Claus Henn, B.; Valeri, L.; Coull, B. A. Statistical software for analyzing the health effects of multiple concurrent exposures via Bayesian kernel machine regression. Environ. Health. 2018, 17, 67.
29. Roth, N.; Zilliacus, J.; Beronius, A. Development of the SciRAP approach for evaluating the reliability and relevance of in vitro toxicity data. Front. Toxicol. 2021, 3, 746430.
30. Couderc, M.; Gandar, A.; Kamari, A.; et al. Neurodevelopmental and behavioral effects of nonylphenol exposure during gestational and breastfeeding period on F1 rats. Neurotoxicology 2014, 44, 237-49.
31. Wang, P. W.; Huang, Y. F.; Wang, C. H.; Fang, L. J.; Chen, M. L. Prenatal to preschool exposure of nonylphenol and bisphenol A exposure and neurodevelopment in young children. Pediatr. Neonatol. 2024, 65, 76-84.
32. Fang, Y.; Che, X.; You, M.; Xu, Y.; Wang, Y. Perinatal exposure to nonylphenol promotes proliferation of granule cell precursors in offspring cerebellum: involvement of the activation of Notch2 signaling. Neurochem. Int. 2020, 140, 104843.
33. Yao, D.; Li, S.; You, M.; et al. Developmental exposure to nonylphenol leads to depletion of the neural precursor cell pool in the hippocampal dentate gyrus. Chem. Biol. Interact. 2024, 401, 111187.
34. Qiu, Z.; Wang, Y.; Chen, J. Perinatal exposure to nonylphenol induces microglia-mediated nitric oxide and prostaglandin E2 production in offspring hippocampus. Toxicol. Lett. 2019, 301, 114-24.
35. Lee, S. H.; Shin, H. S.; So, Y. H.; et al. Maternal exposure to 4-tert-octylphenol causes alterations in the morphology and function of microglia in the offspring mouse brain. J. Hazard. Mater. 2024, 480, 136258.
36. You, M.; Gu, W.; Li, M.; et al. Perinatal exposure to nonylphenol impairs dendritic outgrowth of cerebellar Purkinje cells in progeny. Chemosphere 2018, 211, 758-66.
37. Jie, Y.; Xuefeng, Y.; Mengxue, Y.; et al. Mechanism of nonylphenol-induced neurotoxicity in F1 rats during sexual maturity. Wien. Klin. Wochenschr. 2016, 128, 426-34.
38. Jie, Y.; Fan, Q. Y.; Binli, H.; et al. Joint neurodevelopmental and behavioral effects of nonylphenol and estradiol on F1 male rats. Int. J. Environ. Health. Res. 2013, 23, 321-30.
39. Takagi, H.; Shibutani, M.; Masutomi, N.; et al. Lack of maternal dietary exposure effects of bisphenol A and nonylphenol during the critical period for brain sexual differentiation on the reproductive/endocrine systems in later life. Arch. Toxicol. 2004, 78, 97-105.
40. Ferguson, S. A.; Flynn, K. M.; Delclos, K. B.; Newbold, R. R. Maternal and offspring toxicity but few sexually dimorphic behavioral alterations result from nonylphenol exposure. Neurotoxicol. Teratol. 2000, 22, 583-91.
41. Kawaguchi, S.; Kuwahara, R.; Kohara, Y.; Uchida, Y.; Oku, Y.; Yamashita, K. Perinatal exposure to low-dose nonylphenol specifically improves spatial learning and memory in male rat offspring. Indian. J. Physiol. Pharmacol. 2015, 59, 211-22.
42. Kim, S. K.; Kim, B. K.; Shim, J. H.; Gil, J. E.; Yoon, Y. D.; Kim, J. H. Nonylphenol and octylphenol-induced apoptosis in human embryonic stem cells is related to Fas-Fas ligand pathway. Toxicol. Sci. 2006, 94, 310-21.
43. Tran, D. N.; Jung, E. M.; Yoo, Y. M.; Jeung, E. B. 4-tert-octylphenol exposure disrupts brain development and subsequent motor, cognition, social, and behavioral functions. Oxid. Med. Cell. Longev. 2020, 2020, 8875604.
44. Litwa, E.; Rzemieniec, J.; Wnuk, A.; Lason, W.; Krzeptowski, W.; Kajta, M. Apoptotic and neurotoxic actions of 4-para-nonylphenol are accompanied by activation of retinoid X receptor and impairment of classical estrogen receptor signaling. J. Steroid. Biochem. Mol. Biol. 2014, 144 Pt B, 334-47.
45. Xu, Y.; Jang, J. H.; Gye, M. C. 4-Octylphenol induces developmental abnormalities and interferes the differentiation of neural crest cells in Xenopus laevis embryos. Environ. Pollut. 2021, 274, 116560.
46. Ning, J.; Ding, C.; Xu, H.; et al. Effect of per- and polyfluoroalkyl substances on neurodevelopment: evidence-based risk assessment in the TRAEC strategy context. Environ. Int. 2024, 191, 109003.
47. Sun, X.; Ying, J.; Ma, X.; Zhong, Y.; Huo, R.; Meng, Q. Effects of gossypol exposure on ovarian reserve function: comprehensive risk assessment based on TRAEC strategy. Toxics 2025, 13, 763.
48. Wan, D.; Liu, Y.; Chang, Q.; et al. Micro/nanoplastic exposure on placental health and adverse pregnancy risks: novel assessment system based upon targeted risk assessment environmental chemicals strategy. Toxics 2024, 12, 553.
49. Zhao, S.; Xu, Q.; Su, Y.; et al. Risk assessment of pyridaben exposure on neurodevelopment of offspring by using TRAEC strategy. Sustain. Horiz. 2025, 13, 100135.
50. Li, S.; Jiang, Z.; Chai, W.; Xu, Y.; Wang, Y. Autophagy activation alleviates nonylphenol-induced apoptosis in cultured cortical neurons. Neurochem. Int. 2019, 122, 73-84.
51. Kahn, L. G.; Philippat, C.; Nakayama, S. F.; Slama, R.; Trasande, L. Endocrine-disrupting chemicals: implications for human health. Lancet. Diabetes. Endocrinol. 2020, 8, 703-18.
52. White, R.; Jobling, S.; Hoare, S. A.; Sumpter, J. P.; Parker, M. G. Environmentally persistent alkylphenolic compounds are estrogenic. Endocrinology 1994, 135, 175-82.
53. Park, K.; Kwak, I. S. Gene expression of ribosomal protein mRNA in Chironomus riparius: effects of endocrine disruptor chemicals and antibiotics. Comp. Biochem. Physiol. C. Toxicol. Pharmacol. 2012, 156, 113-20.
54. Roig, B.; Cadiere, A.; Bressieux, S.; Biau, S.; Faure, S.; de Santa Barbara, P. Environmental concentration of nonylphenol alters the development of urogenital and visceral organs in avian model. Environ. Int. 2014, 62, 78-85.
55. Hair, N. L.; Hanson, J. L.; Wolfe, B. L.; Pollak, S. D. Association of child poverty, brain development, and academic achievement. JAMA. Pediatr. 2015, 169, 822-9.
56. Church, J. S.; Tamayo, J. M.; Ashwood, P.; Schwartzer, J. J. Repeated allergic asthma in early versus late pregnancy differentially impacts offspring brain and behavior development. Brain. Behav. Immun. 2021, 93, 66-79.
57. Elewa, H. S.; Salama, D. A.; Hikal, M. S.; et al. Protective effects of resveratrol and naringenin against nonylphenol-induced oxidative stress in rats. AMB. Express. 2025, 15, 7.
58. Amine, I.; Guillien, A.; Bayat, S.; et al. Early-life exposure to mixtures of endocrine-disrupting chemicals and a multi-domain health score in preschool children. Environ. Res. 2025, 272, 121173.
59. Ouidir, M.; Cissé, A. H.; Botton, J.; et al. Fetal and infancy exposure to phenols, parabens, and phthalates and anthropometric measurements up to 36 months, in the longitudinal SEPAGES cohort. Environ. Health. Perspect. 2024, 132, 57002.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.