Gut microbial dysbiosis in Gaucher disease: implications for associated complications
Abstract
The human gut microbiome is a highly dynamic and exceptionally diverse microbial ecosystem. Gut dysbiosis, or an imbalance in gut microbiota, can lead to various metabolic conditions associated with immune activation and systemic inflammation. Gaucher disease (GD) is a rare lysosomal disorder caused by variations in the GBA1 (Glucosylceramidase Beta 1) gene, leading to the accumulation of glucocerebrosides in lysosomes, predominantly in macrophages. This leads to multi-organ complications in patients, such as hepatosplenomegaly, hematological and skeletal abnormalities. GBA1 variants also predispose individuals to neurodegenerative disorders such as Parkinson's disease (PD). Emerging preclinical evidence suggests that gut dysbiosis may contribute to the pathophysiology of GD and related complications, although causal relationships have not been established. Recent studies have reported gut microbial alterations in preclinical models harboring GBA1 variants, descriptively linking dysbiosis with chronic immune activation and PD-related phenotypes. Additional research is warranted to define the role of gut dysbiosis in the pathogenesis of GD and to explore potential adjunctive strategies in a hypothesis-generating context. This review summarizes current evidence regarding gut dysbiosis in GD and discusses its conceptual relevance to immunological, metabolic, neurological, and skeletal manifestations in a hypothesis-generating framework. Probiotics, prebiotics, synbiotics, dietary supplements, and lifestyle modifications are discussed as exploratory adjunctive approaches that warrant further investigation in parallel with established GD therapies.
Keywords
INTRODUCTION
The gut microbiome plays a crucial role in maintaining human health and contributing to disease pathogenesis[1]. It is a highly diverse and dynamic microbial community of bacteria, viruses, fungi, and archaea that populate the gastrointestinal (GI) tract[2,3]. The microbes regulate a variety of physiological processes, such as immunological regulation, metabolism, and digestion, that maintain overall homeostasis[4,5]. Metabolites derived from the gut regulate behavior, emotion, and cognition, and are central regulators of immunological responses, systemic inflammation, metabolic disorders, and neurological function[6,7]. There are a variety of factors that influence gut microbiota composition, such as age, sex, diet, environment, and genetics[8].
Dysbiosis is a disruption of the gut microbiota that leads to metabolic disease and has also been implicated in immune activation and systemic inflammation[9]. In human and animal models, gut dysbiosis research has investigated immunological dysregulation, brain protein aggregation abnormalities, and reduced neuronal and synaptic activity, which are key drivers of neurodegenerative diseases[10]. Advances in gut microbiome research have unveiled the broad-ranging influence of gut microbiota on immuno-metabolism and brain health, and unearthed sophisticated interactions between microbial communities and host physiology[11-13].
Alterations in gut microbial composition and the metabolites they produce are involved in regulating metabolism and controlling liver and brain function[14,15]. The gut-liver barrier regulates the gut-liver interaction; upon passage, bacteria and their metabolites become accessible to the liver and contribute to a variety of hepatic ailments[16]. In addition, stimulation of vagal and spinal afferent neurons by intestinal-derived peptides and hormones released in response to eating affects neuronal signaling, modulating gut and liver function through parasympathetic regulation[17]. The gut-liver-brain axis is a multifaceted communication pathway that involves all three organs, regulating immunological, hormonal, metabolic, and neurological signals[18,19]. The intricacy of microbial communities, which collectively have 100-fold more genes than the human genome, and their interactions with human physiology have been an active area of research in recent years[20].
Gaucher disease (GD) is a rare autosomal recessive disorder characterized by reduced lysosomal β-glucocerebrosidase (GCase) activity resulting from mutations in the GBA1 (Glucosylceramidase Beta 1) gene on chromosome 1 (1q21). Ninety percent of disease-causing alleles in GBA1 gene are attributed to four most common variants: c.1226A>G (N409S or N370S), c.84dupG (84GG), c.1448T>C (L483P or L444P), and c.115+1G>A (IVS2+1)[21,22]. Deficiency of GCase leads to excessive storage of glucocerebrosides in lysosomes, predominantly in the macrophages, described as lipid-rich macrophages, known as Gaucher cells[23,24]. The most common clinical manifestations of GD include hepatosplenomegaly, anemia, thrombocytopenia, bone pain, and skeletal abnormalities due to the accumulation of Gaucher cells in organs, causing abdominal distension and tenderness, asthenia, bleeding tendencies, and immune dysfunction[25,26]. Bone complications in GD, such as pain, osteopenia, fractures, and avascular necrosis, are severe and significantly affect the quality of life of patients with GD[27].
The commonly recognized phenotypic forms of GD are Type 1 (non-neuronopathic), Type 2 (acute neuronopathic), and Type 3 (chronic neuronopathic) [Table 1]. Type 1 is the most frequent, constituting 95% of all GD, with a median age at diagnosis typically in adulthood. Type 2 appears during infancy with severe neurological impairment and is usually fatal during the initial years of life. Type 3 is chronic, with progressive neurological deterioration, which is slower compared to Type 2[28,29]. The worldwide prevalence of GD1 is 0.9 (95%CI: 0.7-1.1) per 100,000 individuals. Latin America has the lowest prevalence estimates (0.15-0.32), followed by the Middle East (0.20-0.33, excluding Israeli values), Europe (0.11-1.1), and North America (0.60-1.93)[30]. Ashkenazi Jews have a higher risk of GD1, particularly associated with the c.1226A>G and c.84dupG variants. In contrast, individuals carrying the c.1448T>C variant have an increased risk of developing neurological complications, commonly seen in patients with GD2 and GD3[22].
Overview of Gaucher disease: types, clinical characteristics, and prevalence
| GD types | Prevalence/Onset | Neurological involvement | GBA1 genotype | Prognosis | Disease frequency (per 100,000) |
| Type 1 (Non-neuronopathic) | Most frequent form (~95% of all GD); Diagnosis: adulthood | Absent | c.1226A>G, c.84dupG and others | Variable, generally normal life expectancy under treatment | Worldwide: 0.9 (0.7-1.1) |
| Type 2 (Acute neuronopathic) | Less than 5% of all GD; Diagnosis: onset in infancy | Severe, rapid neurodegeneration | c.1448T>C and variants associated with neurological forms | Fatal, 2-3 years of life | Rare (< 0.1 globally) |
| Type 3 (Chronic neuronopathic) | Less than 5% of all GD; Diagnosis: onset in childhood or adolescence | Present, progressive but slower than Type 2 | c.1448T>C, others | Chronic progression with neurodegeneration in later life | Rare (< 0.1 globally) |
GBA1 gene variants lead to specific molecular changes that trigger a series of signaling pathways, including oxidative stress, endoplasmic reticulum (ER) stress, mitochondrial dysfunction, inflammation, immune activation, and defects in autophagy and extracellular vesicle production[31]. Autophagy, a key regulator of cellular homeostasis, is strongly influenced by the gut microbiota. Dysbiosis has been associated with impaired lysosomal function and disruption of the gut and blood-brain barrier (BBB) integrity, triggering neuroinflammation through alterations in microglial and regulatory T-cell activity, ultimately promoting protein misfolding and neurodegeneration. Further, Restorative Microbiota Therapy (RMT), including probiotics, prebiotics, synbiotics, and Fecal Microbiota Transplantation (FMT), has been proposed as a strategy to restore eubiosis, immune balance, and autophagic function in neurodegenerative diseases[32,33], but it requires further validation in animal models and clinical studies.
The heterozygous GBA1 variant is the most common genetic risk factor for PD, implicating lysosomal dysfunction in neurodegeneration[34]. Emerging evidence also suggests that gut microbial dysbiosis may contribute to PD via the gut-brain axis, promoting systemic inflammation, microglial activation, and lysosomal stress[35]. PD is believed to include a prodromal phase characterized by GI dysbiosis, suggesting that microbial changes may precede overt neurological comorbidities. However, it remains challenging to establish whether dysbiosis is a precursor to or a consequence of PD[36].
Traditionally, GI manifestations have not been a major focus in GD management, apart from reports of early satiety due to highly enlarged spleens[37]. Despite the well-characterized systemic manifestations of GD, the role of gut dysbiosis remains under-investigated. However, recent pre-clinical studies [Table 2] of GBA1 gene variants indicate that the gut may play a pivotal role in GD pathophysiology and contribute to the phenotypic heterogeneity observed in GD.
Preclinical evidence of gut dysbiosis in GBA1 variant models
| Experimental model | Key findings | References |
| Gba1b-/-Drosophila | Autophagic dysfunction and gut microbiota dysbiosis caused chronic immune activation via NF-kB/IMD pathway | [85] |
| Gba1b D370S/+, Gba1b L444P/+ Drosophila | Lysosomal abnormalities were seen in the gut region | [86] |
| A53T and A53T-L444P Mice | Depletion of Lactobacillus spp. in the gut composition was observed and linked to impaired cognitive function | [87] |
| Gba1 L444P/WT Mice | Longitudinal studies showed gut microbial imbalances are associated with increased systemic inflammation and altered metabolism | [88] |
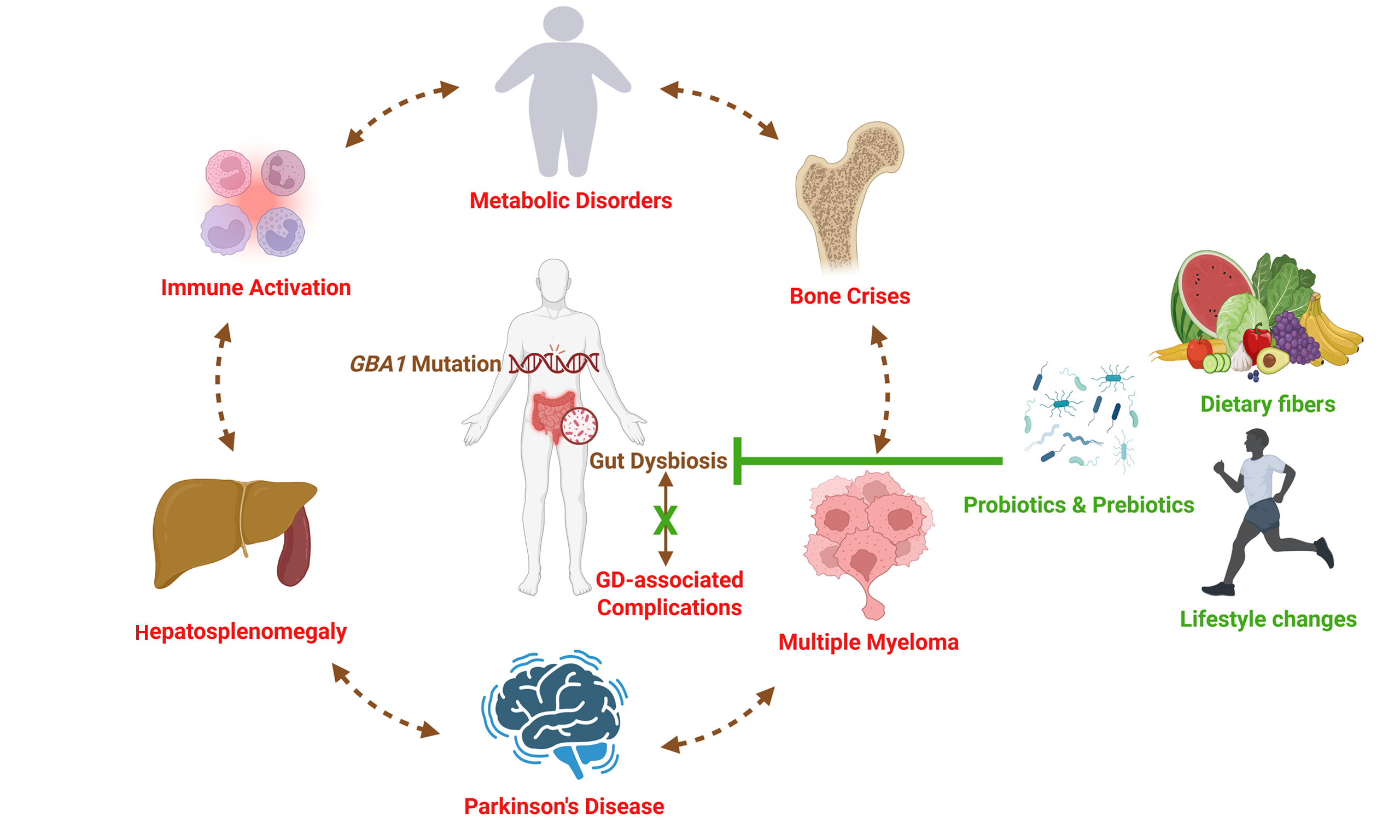
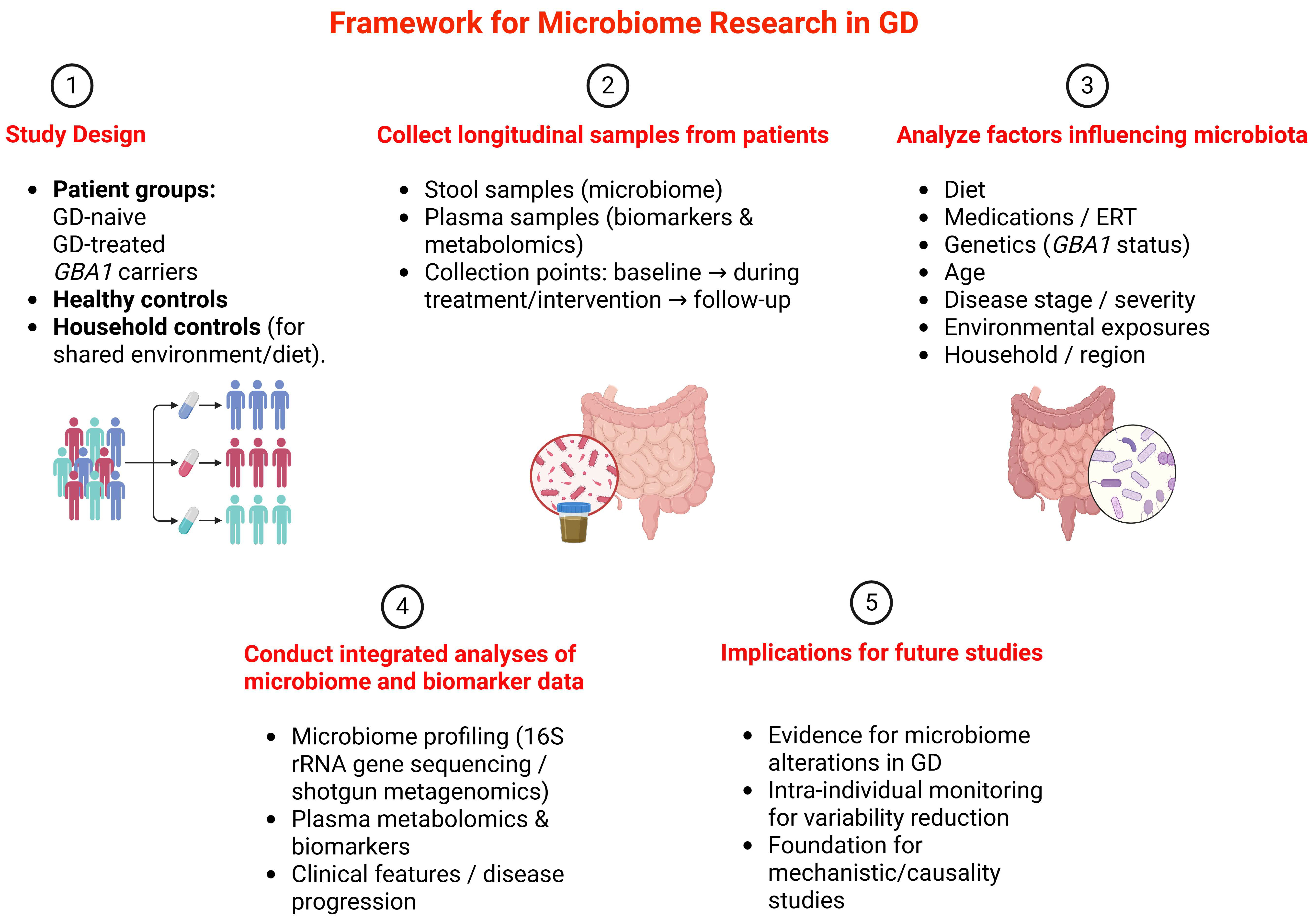
This review highlights preclinical evidence of GBA1-associated gut dysbiosis and describes potential microbiome-mediated pathways that may contribute to the pathogenesis of GD and associated complications. To date, no clinical studies have systematically characterized the gut microbiome in patients with GD; current knowledge is limited to preclinical GBA1-based models in Drosophila and mice, as well as insights drawn from related complications. The novelty of this work lies in (i) integrating immune-metabolic, skeletal, and neurological perspectives within a single microbiome-focused framework specific to GD; and (ii) translating these preclinical observations into a hypothesis-driven clinical framework [Figure 1], with all proposed mechanisms considered exploratory and warranting further research alongside established GD therapies.
Figure 1. Hypothesis-driven framework for a pilot longitudinal microbiome and biomarker study in GD. GD: Gaucher disease; GBA1: glucosylceramidase beta 1; ERT: enzyme replacement therapy; 16S rRNA: 16S ribosomal RNA. Created in BioRender. Sharma, R.R. (2026) https://BioRender.com/w94arl2 Created using BioRender: .
HUMAN GUT MICROBIOME
The human gut microbiota comprises a complex and dynamic network of trillions of microorganisms, including bacteria, viruses, fungi, archaea, and protozoa, with the majority residing in the GI tract. Microbes play a crucial role in various physiological processes, including immune system regulation, digestion, metabolism, and neurological function[2]. Numerous factors, including age, diet, environment, genetics, lifestyle, geography, and antibiotic therapies, influence the gut microbiome. These factors collectively determine the composition and function of the gut microbiota, which are crucial to overall human health[3,38].
Human gut microbiome composition
Most bacteria in the human gut microbiota fall within four main phyla: Pseudomonadota (formerly Proteobacteria), Actinomycetota (Actinobacteria), Bacteroidota (Bacteroidetes), and Pseudomonadota (Firmicutes). These are central to several essential biological processes, such as carbohydrate metabolism, dietary fiber fermentation, and immunological response regulation on host health and disease[39,40]. Immunological responses and gut homeostasis are also significantly regulated by other microbes, including fungi, viruses, and archaea[41]. The phage-dominated gut virome shapes bacterial communities and influences host immune responses[42]. The microbiome evolves throughout life. Aging raises the number of inflammation-related microbes and lowers microbial diversity[43].
Gut microbiome role in health and disease
Recent epidemiological, physiological, and omics-based studies conducted at both preclinical and clinical levels have demonstrated that the intestinal microbiota plays a crucial role in health and disease. Alterations in the GI microbiota are linked to several major human diseases, including obesity, diabetes, cardiovascular disorders, cancer, hypertension, and inflammatory bowel diseases (IBDs)[1]. Many of these detrimental changes have been linked to Western diets characterized by low fiber content and high consumption of processed foods, with consequent significant harmful effects on microbiota composition, including reductions in beneficial bacteria such as Bifidobacterium spp. and Faecalibacterium prausnitzii (F. prausnitzii) in the gut[44]. Individuals who are obese have been shown to have an increased Firmicutes-to-Bacteroidetes ratio, which is associated with increased energy intake from food and increased adiposity[45]. However, biomarkers for an obesity-associated microbiome remain elusive[46]. Type 2 diabetes and insulin resistance have been associated with changes in the gut microbiota, particularly a reduction in microbial-derived metabolites, such as short-chain fatty acids (SCFAs), which play a crucial role in regulating energy balance[47]. It has been shown that alterations in gut microbiota composition significantly influence neurological disorders (NDs) such as anxiety, depression, autism, PD, and Alzheimer's disease via neuroinflammation and microglial activation[35]. The microbiota also influences brain function through immune modulation, the production of neurotransmitters such as serotonin and gamma-aminobutyric acid (GABA), and signaling via the vagus nerve[48]. A study has revealed that a decrease in microbial diversity is associated with IBD, particularly a loss of butyrate-producing bacteria, such as F. prausnitzii[49]. Moreover, dysbiosis has also been implicated in colorectal cancer, linked to species such as Fusobacterium nucleatum, Bacteroides fragilis, and Escherichia coli, which promote tumorigenesis through immune modulation and disruption of the intestinal barrier[50]. In addition to direct effects, dysbiosis can indirectly influence drug metabolism and toxicity by modulating host drug metabolism and disposition and by competing with bacterial-derived metabolites for xenobiotic metabolism pathways[51]. However, there is some preclinical evidence showing gut dysbiosis in preclinical models harboring GBA1 gene variants. To date, no clinical studies have confirmed the gut microbiome's pivotal role in GD modulation.
Factors affecting gut microbiome
The gut microbiome is influenced by intrinsic and extrinsic factors that affect its structure and function. The growth of beneficial bacteria can be induced by nutritional interventions such as fiber- and polyphenol-rich diets, whereas high-fat diets alter the diversity of microbes, leading to dysbiosis[52]. Genetic background also plays a role in shaping the gut microbiome. Specific genetic mutations, including those in the nucleotide-binding oligomerization domain 2 (NOD2) gene, influence the microbial composition and establish susceptibility to IBD[53]. Probiotics such as Lactobacillus and Bifidobacterium help restore microbial homeostasis, whereas antibiotics cause disruption of gut flora and lead to dysbiosis[54]. Prebiotics such as inulin and fructooligosaccharides selectively promote the growth of beneficial microbes, enhancing gut barrier function and minimizing inflammation, restoring homeostasis[55]. FMT has successfully restored gut microbial balance in individuals with dysbiosis and has cured recurrent Clostridioides difficile infections, highlighting how changes in microbiota composition can directly influence disease susceptibility and recovery[56]. In Fabry disease, globotriaosylsphingosine (lyso-Gb3) has been shown to influence bacterial growth, biofilm formation, and SCFAs production, suggesting a direct metabolite-microbiota interaction. By analogy, metabolites accumulating in GD, such as glucosylsphingosine (lyso-GL1), may similarly modulate gut microbial function and contribute to disease complications. Further studies are required to elucidate these mechanisms and their clinical significance[57]. These observations support the concept that modulating the gut microbiota may have therapeutic potential.
GUT MICROBIOME AS A POTENTIAL MODULATOR OF GD-ASSOCIATED COMPLICATIONS
GD is linked with multi-systemic complications. The manifestations include hepatosplenomegaly, anemia, thrombocytopenia, and a variety of skeletal issues, such as osteopenia, lytic lesions, fractures, chronic bone pain, bone crisis, bone infarction, osteonecrosis, and skeletal deformities[58]. Patients with GD are at an increased risk of developing PD compared to the general population[59]. It has been shown that microbial metabolites, such as increased bacterial lipopolysaccharides (LPS) and decreased SCFAs, can affect neuroinflammation and dopaminergic neuronal degeneration, which are essential events in the pathogenesis of PD[60]. GI complications are rarely reported in GD and typically occur in patients with more severe phenotypes[61]. As a result, available reports are primarily limited, most often involving neuronopathic variants. In the context of GD, it is plausible that GBA1 gene variants are associated with gut microbial alterations, suggesting that microbiome-targeted strategies may be conceptually informative for understanding disease heterogeneity. However, direct clinical evidence supporting such approaches in GD is currently lacking.
Despite the absence of direct clinical evidence in GD, preclinical GBA1 models suggest gut dysbiosis may be associated with complications via immune activation, barrier dysfunction, and metabolite dysregulation [Table 2]. These findings are hypothesis-generating and require validation in well-controlled human GD cohorts, where confounders such as enzyme replacement therapy (ERT)/substrate reduction therapy (SRT) status, diet, and antibiotics must be prospectively controlled.
Immune dysfunction
Patients with GD exhibit chronic inflammation, hypergammaglobulinemia, and cytokine release, but not impaired immune function, unless splenectomized[62]. Alterations in the microbiome can initiate immune responses that impact gut-associated lymphoid tissue and systemic immune cells, leading to increased immune activation and systemic inflammation[63]. Changes in the microbial population increase pro-inflammatory cytokines, thereby activating the immune system. However, microbial-derived metabolites such as SCFAs exert anti-inflammatory effects[64], suggesting that restoring microbial balance through RMT may be relevant for hypothesis generation regarding immune dysfunction in patients with GD.
Hepatic disorders
In GD, hepatic dysfunction is common, particularly in patients who remain untreated for many years or have undergone splenectomy[26]. The accumulation of glucocerebrosides in GD can directly disrupt liver function and also alter bile composition, predisposing patients to cholesterol gallstones[65]. Liver complications are further influenced by gut-liver axis disturbances, where microbial signaling from the GI tract modulates hepatic homeostasis[66]. Changes in the intestinal microbiota triggered by bacterial endotoxins such as LPS, pathogen-associated molecular patterns (PAMPs), and infections such as hepatitis B and C promote liver injury by increasing bacterial overgrowth, immune dysfunction, altered luminal factors, and intestinal permeability. There is a reciprocal relationship between bile composition and the gut microbiota that may amplify hepatic dysfunction and disease progression[65,67,68]. Although direct evidence in GD is limited, studies in other liver diseases suggest that restoring microbial balance with probiotics or prebiotics can help alleviate hepatic injury[69]. The therapeutic potential of such approaches in GD remains speculative and requires dedicated investigation.
Metabolic abnormalities
Patients with GD often experience metabolic abnormalities, such as impaired lipid and glucose metabolism, insulin resistance, and vitamin D insufficiency, which significantly impact disease progression and quality of life. Lifestyle factors may further increase their susceptibility to cardiovascular and liver complications. Careful monitoring of metabolic parameters, along with longitudinal studies, is needed to better define risks and guide preventive and therapeutic strategies[70,71]. The gut microbiota regulates metabolic homeostasis by secreting metabolites such as SCFAs that control lipid and glucose metabolism[72]. In contrast, microbial dysbiosis exacerbates metabolic disturbances by altering the production and composition of gut-derived SCFAs, which play key roles in regulating energy homeostasis and glucose metabolism[73], highlighting pathways of interest for future investigation in patients with GD.
Hematological malignancies
There is an increased risk of hematologic malignancies in patients with GD, along with cytopenias and coagulopathies. In particular, those with Type 1 GD often exhibit immunoglobulin abnormalities such as monoclonal gammopathy of undetermined significance (MGUS) and hypergammaglobulinemia, which are associated with B-cell and plasma-cell malignancies, including multiple myeloma, leukemia, and lymphoma[74]. Therefore, early monitoring is essential for understanding risk trajectories and disease progression, and improving prognosis in patients with GD. As previously noted, gut dysbiosis can trigger the activation of inflammatory cytokines, whereas SCFAs have anti-inflammatory effects[64], suggesting that gut dysbiosis may be relevant to understanding GD-related complications.
Neurological disorders
NDs have a multifactorial etiology with genetic and environmental factors and are closely associated with lysosomal dysfunction, such as in Alzheimer's disease, PD, frontotemporal dementia, and amyotrophic lateral sclerosis[75]. Recent studies have demonstrated that SCFA treatment aids in restoring BBB integrity and promotes the maturation of microglia and oligodendrocytes. At the same time, elevated levels of secondary bile acids may impair BBB function and contribute to central nervous system (CNS) disorders[76]. In patients with GD, gut dysbiosis may promote the excessive production of pro-inflammatory cytokines that can cross the BBB, thereby exacerbating neurodegeneration by activating neuroinflammatory pathways implicated in the pathogenesis of PD[77]. To better understand neurological complications in GD, further studies are needed to explore therapeutic strategies targeting the gut microbiota, in light of growing evidence linking it to PD and other neurodegenerative disorders.
Bone mineral abnormalities in GD
Osteopenia, osteoporosis, and recurrent bone pain are commonly observed in patients with GD[78]. The gut microbial homeostasis is critical for bone health as it regulates immune responses[79]. Increasing gut-derived SCFAs through microbiome restoration has been shown to increase osteoblast activity and inhibit osteoclast differentiation, thus modulating pathways involved in bone synthesis and resorption that jointly contribute to the loss of bone mineral density in patients with GD[80,81]. Moreover, the gut microbiome may affect bone metabolism by impacting systemic inflammation and immune regulation, which have been implicated in regulating bone density and bone turnover[82]. Human and animal studies suggest that probiotics can restore gut microbiota balance and support intestinal health, potentially preventing or mitigating bone loss, although further research is needed to clarify the signaling pathways linking the gut microbiome to bone metabolism[83]. Evidence for such effects in patients with GD is lacking. Further investigation is needed to determine whether microbiome-targeted strategies could represent a viable area for future research regarding GD-related bone manifestations.
PRECLINICAL EVIDENCE OF GUT DYSBIOSIS IN GAUCHER DISEASE
To date, evidence of gut dysbiosis in GD has been derived exclusively from GBA1-based animal models. No human clinical studies examining the gut microbiome in GD have been reported. Consequently, these preclinical findings form the primary basis for hypothesizing a role for the microbiome in the pathophysiology of GD. However, species-specific differences between mice and humans must be carefully considered when interpreting and extrapolating these results[84]. Studies in Drosophila and murine models carrying GBA1 variants revealed the role of gut dysbiosis in these pathologies. In Drosophila models of GD, deleting the GBA1 gene has been shown to significantly disrupt gut microbiome composition, leading to pronounced gut dysbiosis. This microbial imbalance is associated with increased intestinal permeability and an enhanced innate immune response, highlighting a breakdown in intestinal homeostasis[85]. Another study in Drosophila with either the D370S or L444P variant showed that D370S caused more severe lysosomal dysfunction in the gut. In contrast, the L444P variant caused damage to brain cells[86]. In a mouse PD model, heterozygous A53T-L444P mice exposed to environmental stressors showed gut dysbiosis and cognitive impairments. PD-related symptoms appeared only in A53T-L444P mice. These changes were linked to a loss of Lactobacillus sp. and altered behavior, suggesting a key role for the gut microbiome in neurodegeneration[87]. In another mouse model with GBA1 L444P heterozygous variants, it has been suggested that GBA1 mutations alone may not be sufficient to cause gut dysbiosis. However, longitudinal studies have shown that the GBA1 L444P/WT mouse model does experience a microbial imbalance. This imbalance was associated with systemic inflammation and altered metabolism[88]. These studies, summarized in Table 2, reflect the clinical complications of GD and advanced PD.
Recent studies also highlight that gut dysbiosis plays a central role in maintaining intestinal barrier integrity and regulating systemic inflammation, which can lead to neurological, metabolic, and GI dysfunction[89,90]. This can further exacerbate bone conditions, such as osteopenia and fractures, highlighting the potential relevance of gut microbial balance in the context of GD pathophysiology[91].
These findings highlight the potential role of gut dysbiosis in the pathogenesis of GD-associated complications and underscore the need for further research to explore the underlying mechanisms that may be relevant to overall health in patients with GD. Therefore, the hypothesis that reestablishing microbial balance can represent a conceptual framework for future adjunctive strategies, pending rigorous preclinical and clinical validation.
Pertinently, distinguishing cause from consequence is essential. We therefore recommend conducting systematic gnotobiotic transfer experiments, in which germ-free mice are colonized with feces from GBA1-mutant donors versus controls, to determine whether GD-associated microbiota can transmit systemic inflammation, neuroinflammatory signatures, and bone phenotypes. Conversely, colonization of mutant mice with healthy humanized microbiota or defined consortia will test reversibility.
CLINICAL EVALUATION OF GUT MICROBIOTA
Stool sample collection is the most practical and widely used approach for evaluating gut microbiota, both for clinical and research purposes, as it is sufficient to assess the dense and diverse microbial communities. It is non-invasive, patient-friendly, and can be performed at home using standardized collection kits[92]. Patients are instructed to refrigerate or freeze the sample before submitting it to the laboratory for analysis, typically within 24 h of collection. Techniques such as 16S rRNA (ribosomal RNA) gene sequencing and shotgun metagenomics are then employed to characterize microbial composition and function[93,94]. It is crucial to remember that stool samples may not accurately reflect the microbiome of the upper GI tract (e.g., the small intestine), where microbial populations vary widely in density and composition. Instead, they represent the luminal microbiota of the distal gut. More invasive sampling techniques, including endoscopic biopsies, luminal brushing, and aspirated intestinal fluid, may be considered if upper gut involvement is suspected or if further study is required. However, these techniques are usually only used for research purposes because of their procedural complexity[95]. A comprehensive clinical evaluation should accompany microbiota sampling to enhance the interpretability of results. Clinicians should assess GI symptoms such as bloating, early satiety, constipation, diarrhea, and abdominal discomfort[96].
During clinical visits, standardized symptom questionnaires can help document bowel patterns in patients with GD. It is also essential to consider other factors, such as diet, antibiotic use, and GD therapies, as they have a significant impact on gut microbial dynamics. However, in family-based studies, collecting samples from first-degree relatives or cohabiting family members, such as parents or siblings, can offer valuable insights into the interplay of genetic predisposition and shared environmental exposures on microbiome composition in patients with GD. Integrating gut microbiota analysis into research protocols may provide deeper insight into GI symptoms and inform the design of microbiome-targeted research studies. A pragmatic, initial biomarker panel should include fecal SCFAs such as acetate, propionate, butyrate; fecal calprotectin and zonulin; and shotgun stool metagenomics; serum lyso-GL1; and a systemic inflammatory cytokine panel. These assays capture microbial function, gut barrier integrity, and disease burden and are suitable for longitudinal, within-person analyses.
THERAPEUTIC IMPLICATIONS
If gut dysbiosis is shown to play a causal role in GD pathogenesis, interest in RMT as an investigational approach would increase. In addition to traditional GD indicators such as lyso-GL1 and Chitotriosidase[97]. Several surrogate biomarkers can be employed to monitor the impact of RMT for patients with GD, such as monitoring SCFAs (butyrate, acetate, and propionate) that demonstrate the action of beneficial gut bacteria[98]. Calprotectin and zonulin are markers of intestinal inflammation and barrier permeability, and their levels in both serum and fecal samples are elevated in individuals with PD[99]. This may be useful for exploratory monitoring in future interventional studies in GD research. The low levels of the gut bacterium Faecalibacterium are linked to conditions such as IBD, colorectal cancer, and depression. Due to its role in supporting gut health, Faecalibacterium is being explored as a promising new probiotic therapy[100]. Therefore, therapies such as probiotics, prebiotics, synbiotics, dietary, and lifestyle interventions are discussed here as investigational strategies with a mechanistic rationale, rather than established therapeutic options. Here, we discuss the conceptual rationale for these approaches in the context of GD pathophysiology and disease complexity. RMT holds mechanistic promise but carries particular safety concerns for patients with GD with immune alterations, including splenectomized individuals. Therefore, any clinical RMT program should follow a staged pathway, beginning with preclinical safety in relevant models, followed by Phase I safety trials with rigorous donor screening, and only then planning randomized efficacy trials.
Probiotics
Probiotics, live microorganisms that confer health benefits when administered correctly, have been shown to improve gut health. Lactobacillus and Bifidobacterium are two of the most extensively researched probiotics, as they can enhance gut barrier function and immune responses[101]. These probiotics can decrease gut permeability, strengthen intestinal epithelial cells, and improve gut immune tolerance[102]. This may be relevant for future investigation in GD with chronic inflammation and GI symptoms as a component of their disease process, and in relation to quality-of-life measures.
Prebiotics and synbiotics
Prebiotics are substances that enhance the activity and growth of beneficial gut microbiota. They are usually indigestible fiber or oligosaccharides, and create a favorable microbial habitat by promoting the development of beneficial gut bacteria[103]. Consumption of a high-fiber diet comprising whole foods has been demonstrated to change gut flora and boost SCFAs, which strengthen gut health[104]. Prebiotics enhance microbiome diversity and can influence downstream pathways associated with inflammation and metabolism. Prebiotics also enhance gut motility and reduce GI distress. Synbiotics, which combine probiotics and prebiotics, offer a synergistic approach to improving gut health[105,106]. Combinational therapies can supply healthy microbes to the gut and offer substrates to support their increased growth and activity[107], which may influence pathways relevant to GD symptoms.
Dietary interventions
Diet is one of the key determinants of gut microbiota organization and function. A diet that promotes a healthy gut microbiome can influence disease-related pathways and symptom management[108]. Prebiotic-rich, high-fiber diets are linked with greater microbiome diversity and less inflammation. Recent studies have emphasized the importance of macronutrient balance and specific dietary modifications in maintaining enzymatic function and metabolic homeostasis[109]. Particular dietary constituents, including polyphenols, found in foods such as fruits, vegetables, and cereals, have been shown to be responsible for prebiotic stimulation of beneficial bacterial growth and inhibition of pathogenic bacterial populations[110,111]. Dietary interventions can enhance the composition and functional potential of the gut microbiome, thereby indirectly influencing disease-related pathways and biomarker trajectories. These effects may drive metabolic reprogramming, modulate immune responses, and potentially enhance the clinical effectiveness of therapies across various diseases[112]. The link between diet, gut microbiota, and disease progression is now more clearly understood. Therefore, personalized dietary treatment can be considered an adjunctive, investigational strategy to help minimize GI symptoms and inflammation characteristic of the disease, while promoting overall gut health and well-being.
Microbiome interactions with standard GD therapies
ERT and SRT are the cornerstones of GD management[113,114]. However, their interplay with the gut microbiome remains virtually unexplored. Several questions arise that warrant systematic investigation. First, does ERT, by reducing substrate accumulation and systemic inflammation, also normalize gut microbial composition or function? Preclinical models and clinical cohorts can test whether longitudinal microbiome signatures shift after therapy initiation and whether residual dysbiosis persists despite a biochemical response. Second, does SRT directly or indirectly alter microbial metabolic pathways? Given that oral SRT agents act systemically and may reach the gut lumen, it is plausible that they influence microbial sphingolipid metabolism, bile acid pools, or SCFA production. Finally, could baseline microbiome composition or functional capacity predict therapeutic response or side effect profile? For instance, microbial taxa or metabolites linked to immune activation or drug metabolism may correlate with treatment efficacy or tolerability. Addressing these questions would not only deepen our understanding of host-microbiome interactions in GD but could also enable microbiome-informed patient stratification for optimized therapy.
Microbiome interactions with standard GD therapies (ERT/SRT) remain entirely unexplored. Hypothetically, restoring barrier function might enhance ERT tissue penetration; however, this requires longitudinal testing with paired microbiome/biomarker sampling before and after therapy initiation. Oral SRT agents could plausibly alter microbial sphingolipid metabolism, though no data exist. Future studies should include ERT/SRT status/timing as core variables.
Lifestyle interventions
Lifestyle changes, such as stress management and exercise, are also helpful for maintaining gut health. Exercise has been found to positively influence gut microbiota composition by enhancing microbial diversity and decreasing inflammation[115]. Anti-inflammatory diets, such as the Mediterranean diet, as well as omega-3 fatty acids, vitamin D, and selenium, can modulate the immune system and improve disease outcomes. Moreover, regular exercise is vital for improving inflammatory profiles in autoimmune diseases by reducing inflammation, enhancing immune regulation, and controlling complications[116]. Likewise, lifestyle intervention-mediated stress-reduction techniques, such as mindfulness, meditation, or yoga, can help reduce systemic inflammation and neurological issues[117]. Combining lifestyle changes with other treatments has been shown to restore GI health, normalize immune responses, and reduce inflammation and neurological symptoms, strategies that may be considered in future studies in GD for holistic management of the condition. This conceptual framework may broaden avenues for future investigation in GD. Individualized treatment regimens using such interventions may influence disease-related parameters and quality of life.
CURRENT LIMITATIONS AND FUTURE PROSPECTS
Studying the gut microbiome in patients with GD presents unique challenges, as lifestyle, diet, and medications can independently influence microbial composition, regardless of disease status. Preclinical studies must account for factors such as coprophagy and housing conditions in mice to ensure reproducible results[118,119]. Dysbiosis should be verified in both human donors and recipient animals following fecal microbiota transplantation to gain mechanistic insight into host-microbe interactions.
Emerging ecological frameworks offer a more holistic approach to evaluating microbiome changes[120], and in some contexts, community stability may be more informative than the abundance of individual taxa[121]. Machine learning methods, including random forest regression, can identify potential biomarkers, yet these findings require careful interpretation, given the need for large sample sizes, cross-validation, and mechanistic follow-up.
The low prevalence of GD further complicates the identification of robust, replicable microbiome signatures, limiting sample sizes. Longitudinal, intra-individual study designs are therefore essential, as they allow each individual to serve as their own control and enable intra-individual tracking of biomarkers before, during, and after interventions. These designs help control for clinical heterogeneity and treatment effects, while minimizing inter-individual variability, for instance, by collecting samples at consistent intervals or from individuals within the same household or under similar environmental conditions.
We propose a hypothesis-driven pilot clinical framework [Figure 1] that emphasizes longitudinal, intra-individual follow-up to investigate the link between the microbiome and GD. This includes patients naive to GD and those treated, GBA1 carriers, and healthy controls, with serial stool and plasma sampling at baseline, 6-12 months, and key treatment milestones (ERT/SRT initiation) coupled to integrated microbiome and metabolomic analyses. Participants should be stratified by GD type, treatment-naïve versus treated status, and splenectomy. Unavoidable confounders, such as recent antibiotics or significant dietary changes, should be documented systematically. Cross-sectional studies are discouraged due to high inter-individual variability.
This hypothesis-driven framework can identify gut microbial alterations associated with GD and generate testable hypotheses regarding disease mechanisms. However, causal inference will require further mechanistic or extended longitudinal studies. While identifying specific microbial species linked to GD complications may inform future research directions, any clinical translation remains premature at this stage. It could inform the development of adjunct microbiome-targeted therapies and diagnostic tools, ultimately improving outcomes for individuals with GD.
CONCLUSION
Looking ahead, the trajectory of this field can be envisioned across short-, medium-, and long-term goals. In the short term (1-3 years), robust, standardized pilot cohorts are needed to define reproducible microbiome signatures in GD and establish correlations with established biomarkers such as lyso-GL1 and systemic inflammatory mediators. Small randomized controlled trials may be considered to evaluate the safety and biomarker effects of probiotics, synbiotics, or dietary interventions. Over the medium term (3-6 years), validated biomarker panels predictive of disease progression, including prodromal neurological features such as Parkinsonism or skeletal complications, will be critical, alongside the development of defined microbial consortia as therapeutics to replace less controlled interventions such as FMT. Integration with existing GD registries can accelerate recruitment and ensure standardized data collection, thereby enhancing the overall effectiveness of the process. In the long term (6+ years), the goal is to develop precision adjunctive microbiome-based therapies tailored to an individual’s microbiome and metabolome profile, used in combination with ERT or SRT. If causal relationships are firmly established, such approaches may eventually help clarify mechanisms underlying symptom burden and disease heterogeneity, particularly in neurological and skeletal manifestations of GD.
Gut dysbiosis is descriptively associated with metabolic, immunological, and neurological processes that may be relevant to GD-related complications. Preclinical GBA1 models link dysbiosis with chronic immune activation, lysosomal abnormalities, and autophagic dysfunction. However, these findings remain correlational and model-dependent. Adjunct microbiome-targeted strategies provide a conceptual framework for biomarker discovery and hypothesis generation, rather than immediate clinical application. They may help identify novel candidate biomarkers for early diagnosis and prognosis, thereby informing future research directions relevant to patient-centered outcomes in GD. We propose consideration of an international GD-microbiome working group to harmonize sampling and share data.
DECLARATIONS
Acknowledgments
The graphic abstract was created with BioRender.com. (Created in BioRender. Sharma, R. (2025) https://BioRender.com/7fl80ac).
Authors’ contributions
Developed the concept of the review, performed the literature review, and drafted and revised the manuscript: Sharma RR
Contributed to conceptual input, manuscript revision, and proofreading: Staley C, Subramanian S, Weinreb NJ, Kartha RV
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors would like to acknowledge the Translational Pharmacology Research Fellowship grant from Sanofi.
Conflict of interests
Weinreb NJ is Guest Editor of the Special Issue “Topic: Innovations in Gaucher Disease Research: Progress, Perspectives, and Promising Therapies” and Associate Editor of the journal Rare Disease and Orphan Drugs Journal. Kartha RV is an Editorial Board Member of the journal. Weinreb NJ and Kartha RV were not involved in any steps of the editorial process, notably including reviewer selection, manuscript handling, or decision-making, while the other authors have declared that they have no conflicts of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Ding R, Goh W, Wu R, et al. Revisit gut microbiota and its impact on human health and disease. J Food Drug Anal. 2019;27:623-31.
2. Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R. Diversity, stability and resilience of the human gut microbiota. Nature. 2012;489:220-30.
4. Shreiner AB, Kao JY, Young VB. The gut microbiome in health and in disease. Curr Opin Gastroenterol. 2015;31:69-75.
5. Rooks MG, Garrett WS. Gut microbiota, metabolites and host immunity. Nat Rev Immunol. 2016;16:341-52.
7. Sharon G, Sampson TR, Geschwind DH, Mazmanian SK. The central nervous system and the gut microbiome. Cell. 2016;167:915-32.
8. Takagi T, Naito Y, Inoue R, et al. Differences in gut microbiota associated with age, sex, and stool consistency in healthy Japanese subjects. J Gastroenterol. 2018;54:53-63.
9. Clemente JC, Manasson J, Scher JU. The role of the gut microbiome in systemic inflammatory disease. BMJ. 2018;360:j5145.
10. Dandamudi BJ, Dimaano KAM, Shah N, et al. Neurodegenerative disorders and the gut-microbiome-brain axis: a literature review. Cureus. 2024;16:e72427.
11. Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol. 2020;19:55-71.
12. Cryan JF, O'riordan KJ, Cowan CSM, et al. The microbiota-gut-brain axis. Physiol Rev. 2019;99:1877-2013.
13. Gubert C, Kong G, Renoir T, Hannan AJ. Exercise, diet and stress as modulators of gut microbiota: implications for neurodegenerative diseases. Neurobiol Dis. 2020;134:104621.
14. Vos WM, Tilg H, Van Hul M, Cani PD. Gut microbiome and health: mechanistic insights. Gut. 2022;71:1020-32.
15. Wang P, Deng X, Zhang C, Yuan H. Gut microbiota and metabolic syndrome. Chin Med J. 2020;133:808-16.
16. Tripathi A, Debelius J, Brenner DA, et al. The gut-liver axis and the intersection with the microbiome. Nat Rev Gastroenterol Hepatol. 2018;15:397-411.
17. Fülling C, Dinan TG, Cryan JF. Gut microbe to brain signaling: what happens in vagus…. Neuron. 2019;101:998-1002.
18. Mayer EA, Knight R, Mazmanian SK, Cryan JF, Tillisch K. Gut microbes and the brain: paradigm shift in neuroscience. J Neurosci. 2014;34:15490-6.
19. Kulkarni S, Ganz J, Bayrer J, Becker L, Bogunovic M, Rao M. Advances in enteric neurobiology: the “brain” in the gut in health and disease. J Neurosci. 2018;38:9346-54.
20. Dekaboruah E, Suryavanshi MV, Chettri D, Verma AK. Human microbiome: an academic update on human body site specific surveillance and its possible role. Arch Microbiol. 2020;202:2147-67.
21. Nguyen Y, Stirnemann J, Belmatoug N. La maladie de Gaucher: quand y penser? Rev Med Interne. 2019;40:313-22.
22. Riboldi GM, Di Fonzo AB. GBA, Gaucher disease, and Parkinson’s disease: from genetic to clinic to new therapeutic approaches. Cells. 2019;8:364.
23. Pandey MK, Burrow TA, Rani R, et al. Complement drives glucosylceramide accumulation and tissue inflammation in Gaucher disease. Nature. 2017;543:108-12.
24. Horowitz M, Braunstein H, Zimran A, Revel-Vilk S, Goker-Alpan O. Lysosomal functions and dysfunctions: molecular and cellular mechanisms underlying Gaucher disease and its association with Parkinson disease. Adv Drug Delivery Rev. 2022;187:114402.
25. Luettel DM, Terluk MR, Roh J, Weinreb NJ, Kartha RV. Emerging biomarkers in Gaucher disease. Adv Clin Chem. 2025;124:1-56.
26. Stirnemann J, Belmatoug N, Camou F, et al. A review of Gaucher disease pathophysiology, clinical presentation and treatments. Int J Mol Sci. 2017;18:441.
27. Mohamed RNES, Elnahal WA, Furtado C, Zeitoun R, Abdel-Wahab N. Skeletal manifestations of Gaucher's disease: a case report and literature review. Semin Musculoskelet Radiol. 2024;28:505-10.
28. Méndez-cobián DA, Guzmán-Silahua S, García-Hernández D, et al. An overview of Gaucher disease. Diagnostics. 2024;14:2840.
29. Lal T, Sidransky E. The spectrum of neurological manifestations associated with Gaucher disease. Diseases. 2017;5:10.
30. Castillon G, Chang S, Moride Y. Global incidence and prevalence of Gaucher disease: a targeted literature review. JCM. 2022;12:85.
31. Roh J, Subramanian S, Weinreb NJ, Kartha RV. Gaucher disease - more than just a rare lipid storage disease. J Mol Med. 2022;100:499-518.
32. Lapaquette P, Bizeau J, Acar N, Bringer M. Reciprocal interactions between gut microbiota and autophagy. World J Gastroenterol. 2021;27:8283-301.
33. Chidambaram SB, Essa MM, Rathipriya A, et al. Gut dysbiosis, defective autophagy and altered immune responses in neurodegenerative diseases: tales of a vicious cycle. Pharmacol Ther. 2022;231:107988.
34. Murphy KE, Gysbers AM, Abbott SK, et al. Reduced glucocerebrosidase is associated with increased α-synuclein in sporadic Parkinson’s disease. Brain. 2014;137:834-48.
35. Ashique S, Mohanto S, Ahmed MG, et al. Gut-brain axis: a cutting-edge approach to target neurological disorders and potential synbiotic application. Heliyon. 2024;10:e34092.
36. Travagli RA, Browning KN, Camilleri M. Parkinson disease and the gut: new insights into pathogenesis and clinical relevance. Nat Rev Gastroenterol Hepatol. 2020;17:673-85.
38. Gilbert JA, Blaser MJ, Caporaso JG, Jansson JK, Lynch SV, Knight R. Current understanding of the human microbiome. Nat Med. 2018;24:392-400.
39. Ridaura VK, Faith JJ, Rey FE, et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science. 2013;341:1241214.
40. Rowland I, Gibson G, Heinken A, et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr. 2017;57:1-24.
41. O'callaghan A, Van Sinderen D. Bifidobacteria and their role as members of the human gut microbiota. Front Microbiol. 2016;7:925.
42. Reyes A, Blanton LV, Cao S, et al. Gut DNA viromes of Malawian twins discordant for severe acute malnutrition. Proc Natl Acad Sci USA. 2015;112:11941-6.
43. Shin N, Whon TW, Bae J. Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015;33:496-503.
44. David LA, Maurice CF, Carmody RN, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2013;505:559-63.
45. Magne F, Gotteland M, Gauthier L, et al. The firmicutes/bacteroidetes ratio: a relevant marker of gut dysbiosis in obese patients? Nutrients. 2020;12:1474.
46. Finucane MM, Sharpton TJ, Laurent TJ, Pollard KS. A taxonomic signature of obesity in the microbiome? Getting to the guts of the matter. PLoS ONE. 2014;9:e84689.
47. Clemente JC, Ursell LK, Parfrey LW, Knight R. The impact of the gut microbiota on human health: an integrative view. Cell. 2012;148:1258-70.
48. Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13:701-12.
49. Lopez-Siles M, Enrich-Capó N, Aldeguer X, et al. Alterations in the abundance and co-occurrence of akkermansia muciniphila and faecalibacterium prausnitzii in the colonic mucosa of inflammatory bowel disease subjects. Front Cell Infect Microbiol. 2018;8:281.
50. Kendong SM, Raja Ali RA, Nawawi KNM, Ahmad HF, Mokhtar NM. Gut dysbiosis and intestinal barrier dysfunction: potential explanation for early-onset colorectal cancer. Front Cell Infect Microbiol. 2021;11:744606.
51. Wilson ID, Nicholson JK. Gut microbiome interactions with drug metabolism, efficacy, and toxicity. Transl Res. 2017;179:204-22.
52. Derrien M, Belzer C, De Vos WM. Akkermansia muciniphila and its role in regulating host functions. Microb Pathog. 2017;106:171-81.
53. Manrique P, Dills M, Young M. The human gut phage community and its implications for health and disease. Viruses. 2017;9:141.
54. Underhill DM, Iliev ID. The mycobiota: interactions between commensal fungi and the host immune system. Nat Rev Immunol. 2014;14:405-16.
56. Karimi M, Shirsalimi N, Hashempour Z, et al. Safety and efficacy of fecal microbiota transplantation (FMT) as a modern adjuvant therapy in various diseases and disorders: a comprehensive literature review. Front Immunol. 2024;15:1439176.
57. Aguilera-Correa J, Madrazo-Clemente P, Martínez-Cuesta MDC, et al. Lyso-Gb3 modulates the gut microbiota and decreases butyrate production. Sci Rep. 2019;9:12010.
58. Linari S. Clinical manifestations and management of Gaucher disease. Clin Cases Miner Bone Metab. 2015;12:157-64.
59. Kinghorn K, Asghari A, Castillo-Quan J. The emerging role of autophagic-lysosomal dysfunction in Gaucher disease and Parkinson's disease. Neural Regen Res. 2017;12:380.
60. Sampson TR, Debelius JW, Thron T, et al. Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell. 2016;167:1469-80.e12.
61. Gragnaniello V, Cazzorla C, Gueraldi D, Puma A, Loro C, Burlina AB. The hidden burden: gastrointestinal involvement in lysosomal storage disorders. Metabolites. 2025;15:361.
62. Sønder SU, Limgala RP, Ivanova MM, et al. Persistent immune alterations and comorbidities in splenectomized patients with Gaucher disease. Blood Cells Mol Dis. 2016;59:8-15.
63. Candelli M, Franza L, Pignataro G, et al. Interaction between lipopolysaccharide and gut microbiota in inflammatory bowel diseases. Int J Mol Sci. 2021;22:6242.
64. Lavelle A, Sokol H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2020;17:223-37.
65. Taddei TH, Dziura J, Chen S, et al. High incidence of cholesterol gallstone disease in type 1 Gaucher disease: characterizing the biliary phenotype of type 1 Gaucher disease. J Inherit Metab Dis. 2010;33:291-300.
66. Albillos A, De Gottardi A, Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol. 2020;72:558-77.
67. Larabi AB, Masson HLP, Bäumler AJ. Bile acids as modulators of gut microbiota composition and function. Gut Microbes. 2023;15:2172671.
68. Hu H, Shao W, Liu Q, et al. Gut microbiota promotes cholesterol gallstone formation by modulating bile acid composition and biliary cholesterol secretion. Nat Commun. 2022;13:252.
69. Xu S, Zhao M, Wang Q, et al. Effectiveness of probiotics and prebiotics against acute liver injury: a meta-analysis. Front Med. 2021;8:739337.
70. Nascimbeni F, Dalla Salda A, Carubbi F. Energy balance, glucose and lipid metabolism, cardiovascular risk and liver disease burden in adult patients with type 1 Gaucher disease. Blood Cells Mol Dis. 2018;68:74-80.
71. Kałużna M, Trzeciak I, Ziemnicka K, Machaczka M, Ruchała M. Endocrine and metabolic disorders in patients with Gaucher disease type 1: a review. Orphanet J Rare Dis. 2019;14:275.
72. He J, Zhang P, Shen L, et al. Short-chain fatty acids and their association with signalling pathways in inflammation, glucose and lipid metabolism. Int J Mol Sci. 2020;21:6356.
73. Portincasa P, Bonfrate L, Vacca M, et al. Gut microbiota and short chain fatty acids: implications in glucose homeostasis. Int J Mol Sci. 2022;23:1105.
74. Rosenbloom BE, Cappellini MD, Weinreb NJ, et al. Cancer risk and gammopathies in 2123 adults with Gaucher disease type 1 in the international gaucher group gaucher registry. Am J Hematol. 2022;97:1337-47.
75. Udayar V, Chen Y, Sidransky E, Jagasia R. Lysosomal dysfunction in neurodegeneration: emerging concepts and methods. Trends Neurosci. 2022;45:184-99.
76. Park J, Kim CH. Regulation of common neurological disorders by gut microbial metabolites. Exp Mol Med. 2021;53:1821-33.
77. Hull A, Atilano ML, Gergi L, Kinghorn KJ. Lysosomal storage, impaired autophagy and innate immunity in Gaucher and Parkinson's diseases: insights for drug discovery. Phil Trans R Soc B. 2024;379:20220381.
78. Masi L, Brandi ML. Gaucher disease: the role of the specialist on metabolic bone diseases. Clin Cases Miner Bone Metab. 2015;12:165-9.
79. Qi P, Chen X, Liu H, Ma J, Qi Z, Xie X. Regulatory mechanisms of gut homeostasis and bone metabolism interplay in osteoporosis. Phenomics. 2025;5:435-45.
80. Tu Y, Yang R, Xu X, Zhou X. The microbiota-gut-bone axis and bone health. J Leukocyte Biol. 2021;110:525-37.
81. Marcucci G, Brandi ML. The Diagnosis and therapy of osteoporosis in Gaucher disease. Calcif Tissue Int. 2025;116:31.
82. Behera J, Ison J, Tyagi SC, Tyagi N. The role of gut microbiota in bone homeostasis. Bone. 2020;135:115317.
83. Schepper JD, Irwin R, Kang J, et al. Probiotics in gut-bone signaling. Adv Exp Med Biol. 2017;1033:225-47.
84. Nguyen TLA, Vieira-silva S, Liston A, Raes J. How informative is the mouse for human gut microbiota research? Dis Model Mech. 2015;8:1-16.
85. Atilano ML, Hull A, Romila C, et al. Autophagic dysfunction and gut microbiota dysbiosis cause chronic immune activation in a Drosophila model of Gaucher disease. PLoS Genet. 2023;19:e1011063.
86. Kuppuramalingam A, Cabasso O, Horowitz M. Functional analysis of human GBA1 missense mutations in drosophila: insights into Gaucher disease pathogenesis and phenotypic consequences. Cells. 2024;13:1619.
87. Chaklai A, O’neil A, Goel S, et al. Effects of paraquat, dextran sulfate sodium, and irradiation on behavioral and cognitive performance and the gut microbiome in A53T and A53T-L444P mice. Genes. 2024;15:282.
88. Menozzi E, Geiger M, Meslier V, et al. Exploring the relationship between GBA1 host genotype and gut microbiome in the GBA1L444P/WT mouse model: implications for Parkinson’s disease pathogenesis. Front Neurosci. 2025;19:1546203.
89. Noble EE, Hsu TM, Kanoski SE. Gut to brain dysbiosis: mechanisms linking western diet consumption, the microbiome, and cognitive impairment. Front Behav Neurosci. 2017;11:9.
90. Du Y, He C, An Y, et al. The role of short chain fatty acids in inflammation and body health. Int J Mol Sci. 2024;25:7379.
91. Lazar V, Ditu L, Pircalabioru GG, et al. Aspects of gut microbiota and immune system interactions in infectious diseases, immunopathology, and cancer. Front Immunol. 2018;9:1830.
92. Jones J, Reinke SN, Ali A, Palmer DJ, Christophersen CT. Fecal sample collection methods and time of day impact microbiome composition and short chain fatty acid concentrations. Sci Rep. 2021;11:13964.
93. Allaband C, Mcdonald D, Vázquez-baeza Y, et al. Microbiome 101: studying, analyzing, and interpreting gut microbiome data for clinicians. Clin Gastroenterol Hepatol. 2019;17:218-30.
94. Vandeputte D, Tito RY, Vanleeuwen R, Falony G, Raes J. Practical considerations for large-scale gut microbiome studies. FEMS Microbiol Rev. 2017;41:S154-67.
95. Tang Q, Jin G, Wang G, et al. Current sampling methods for gut microbiota: a call for more precise devices. Front Cell Infect Microbiol. 2020;10:151.
96. Crucillà S, Caldart F, Michelon M, Marasco G, Costantino A. Functional abdominal bloating and gut microbiota: an update. Microorganisms. 2024;12:1669.
97. Giuffrida G, Markovic U, Condorelli A, et al. Glucosylsphingosine (Lyso-Gb1) as a reliable biomarker in Gaucher disease: a narrative review. Orphanet J Rare Dis. 2023;18:27.
98. Meyers G, Samouda H, Bohn T. Short chain fatty acid metabolism in relation to gut microbiota and genetic variability. Nutrients. 2022;14:5361.
99. Dumitrescu L, Marta D, Dănău A, et al. Serum and fecal markers of intestinal inflammation and intestinal barrier permeability are elevated in Parkinson’S disease. Front Neurosci. 2021;15:689723.
100. Martín R, Rios-covian D, Huillet E, et al. Faecalibacterium: a bacterial genus with promising human health applications. FEMS Microbiol Rev. 2023;47:fuad039.
101. Kim Y, Mills DA. Exploring the gut microbiome: probiotics, prebiotics, synbiotics, and postbiotics as key players in human health and disease improvement. Food Sci Biotechnol. 2024;33:2065-80.
103. You S, Ma Y, Yan B, et al. The promotion mechanism of prebiotics for probiotics: a review. Front Nutr. 2022;9:1000517.
104. Oliver A, Chase AB, Weihe C, et al. High-fiber, whole-food dietary intervention alters the human gut microbiome but not fecal short-chain fatty acids. mSystems. 2021;6:00115-21.
105. Rinninella E, Cintoni M, Raoul P, et al. Food components and dietary habits: keys for a healthy gut microbiota composition. Nutrients. 2019;11:2393.
106. Li H, Zhou D, Gan R, et al. Effects and mechanisms of probiotics, prebiotics, synbiotics, and postbiotics on metabolic diseases targeting gut microbiota: a narrative review. Nutrients. 2021;13:3211.
107. Abe M, Abe H. Lifestyle medicine - an evidence based approach to nutrition, sleep, physical activity, and stress management on health and chronic illness. Pers Med Univ. 2019;8:3-9.
108. Sobek KŁ, Mazur M, Waz D, et al. The impact of diet and lifestyle on the course of autoimmune diseases. Qual Sport. 2024;36:56879.
109. Yadav RK, Magan D, Mehta N, Sharma R, Mahapatra SC. Efficacy of a short-term yoga-based lifestyle intervention in reducing stress and inflammation: preliminary results. J Altern Complement Med. 2012;18:662-7.
110. Călinoiu LF, Vodnar DC. Whole grains and phenolic acids: a review on bioactivity, functionality, health benefits and bioavailability. Nutrients. 2018;10:1615.
111. Roy S, Nag S, Saini A, Choudhury L. Association of human gut microbiota with rare diseases: a close peep through. Intractable Rare Dis Res. 2022;11:52-62.
112. Xiao Y, Gong Y, Qi Y, Shao Z, Jiang Y. Effects of dietary intervention on human diseases: molecular mechanisms and therapeutic potential. Sig Transduct Target Ther. 2024;9:59.
113. Grabowski GA, Antommaria AH, Kolodny EH, Mistry PK. Gaucher disease: basic and translational science needs for more complete therapy and management. Mol Genet Metab. 2021;132:59-75.
114. Gary SE, Ryan E, Steward AM, Sidransky E. Recent advances in the diagnosis and management of Gaucher disease. Expert Rev Endocrinol Metab. 2018;13:107-18.
115. Strasser B, Wolters M, Weyh C, Krüger K, Ticinesi A. The effects of lifestyle and diet on gut microbiota composition, inflammation and muscle performance in our aging society. Nutrients. 2021;13:2045.
116. Kashyap PC, Chia N, Nelson H, Segal E, Elinav E. Microbiome at the frontier of personalized medicine. Mayo Clin Proc. 2017;92:1855-64.
117. Behrouzi A, Nafari AH, Siadat SD. The significance of microbiome in personalized medicine. Clin Transl Med. 2019;8:e16.
118. Walter J, Armet AM, Finlay BB, Shanahan F. Establishing or exaggerating causality for the gut microbiome: lessons from human microbiota-associated rodents. Cell. 2020;180:221-32.
119. Knight R, Vrbanac A, Taylor BC, et al. Best practices for analysing microbiomes. Nat Rev Microbiol. 2018;16:410-22.
120. Walter J, Maldonado-Gómez MX, Martínez I. To engraft or not to engraft: an ecological framework for gut microbiome modulation with live microbes. Curr Opin Biotechnol. 2018;49:129-39.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.