Improving utility and value of wastewater-based epidemiology data for community stakeholders
Abstract
The field of wastewater-based epidemiology (WBE) has experienced significant adoption in response to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic and current initiatives will soon apply the novel technique to environmental matrices outside of municipal wastewater samples. Many well-designed surveillance programs have been abandoned or underutilized due to a disconnect between data providers and end users. As the breadth of wastewater surveillance applications continues to increase, it will be essential to improve upon the core principles, standardization procedures, and data analysis techniques that are central to the field. This review evaluates strategies to improve the practical utility and expand the multidisciplinary value of WBE data for community stakeholders, examining how expanding analytical capabilities towards multi-target surveillance, standardizing laboratory methodologies, improving data normalization and modeling approaches, and prioritizing data transparency can bridge this gap. Improving the utility and increasing the value of WBE data may lead to increased adoption of the methodology and foster the multidisciplinary utilization of wastewater surveillance datasets. Particular attention is given to lessons learned from COVID-19 (coronavirus disease 2019) surveillance, representing the largest WBE deployment to date, with an emphasis on generalizable principles applicable across narcotics monitoring, multi-pathogen surveillance, and emerging contaminant tracking. Furthermore, the importance of serving needs-based communities, economically disadvantaged communities, and developing nations is highlighted as a core principle of WBE, serving not only to promote universal health equity but to contribute to the global understanding of communicable disease prevalence. Consideration of these and similar principles may support continued project buy-in, strengthen longitudinal WBE datasets, and increase the global adoption of the technique.
Keywords
INTRODUCTION
Modern wastewater-based epidemiology (WBE) has had a greater societal impact today than it has at any other point since the technique’s first proof-of-concept project in 2005[1]. While initially purposed for the surveillance of narcotics consumption at the community level, WBE has experienced worldwide adoption in response to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic in early 2020. The methodology sits at the intersection of human health, civil and environmental engineering, analytical chemistry and biology, and bioinformatics to provide a near-real-time source of health data at the community level. The most notable wastewater surveillance effort in the United States remains the Centers for Disease Control and Prevention (CDC) National Wastewater Surveillance System (NWSS) program[2]. A significant breadth of projects spearheaded by state and local government organizations have also expanded the U.S. WBE SARS-CoV-2 surveillance to include monitoring at primary and secondary education systems[3,4], within tribal communities[5], universities[6-8], correctional facilities[9], other congregate living settings[10,11], commercial passenger aircraft[12], and event-specific applications[13].
There currently exists a multidisciplinary effort across academia, government, and industry to expand the breadth of WBE’s analytical capability. While the two primary applications of WBE to date are (i) the quantification of narcotics consumption[14,15] and (ii) community surveillance of SARS-CoV-2[16,17], these aspects represent a subset of the total potential applications of the technique. Researchers have expanded the methodology to monitor pesticide exposure[18,19], food consumption and diet[20-22], endogenous stress biomarkers[23], antibiotic resistance[24-31], and hazard exposure[32,33]. The application has been hypothesized to be a robust system for monitoring for chemical and biological agents of terror attacks[34]. The methodology has also been adapted to monitor communicable diseases such as influenza A and B[35,36], norovirus[37], MPox[38], respiratory syncytial virus (RSV)[39], measles[40], West Nile virus[41], poliovirus[42], sexually transmitted infections[43], and hepatitis[44]. Recent proposals in the literature have identified feasible approaches for extending surveillance to additional targets of interest, including vector-borne[45] and non-communicable diseases[46].
Despite the rapid expansion of wastewater surveillance globally, a notable gap persists in stakeholder and decision-maker understanding of how to interpret and act on surveillance data[47,48]. A 2024 survey conducted by the Association of Public Health Laboratories (APHL) identified data use uncertainty as the largest barrier to WBE program implementation[48]. Diamond et al. further reported a persistent gap between public health officials’ recognition of the value of wastewater surveillance and its actual integration into pandemic decision-making[49]. Collectively, these gaps have left otherwise robust monitoring programs underutilized or prematurely discontinued. Bridging this disconnect will be essential in establishing robust, long-term wastewater monitoring programs and leveraging them effectively to promote positive community health outcomes. Furthermore, it is important for the field to explore options for increasing the value and subsequent utility of wastewater surveillance data. If WBE data is adopted and utilized by a wider array of professionals, it is likely that WBE would experience a higher rate of project implementation worldwide. This would not only serve the communities where projects are established but also contribute to the global understanding of community-level resource consumption and health patterns.
METHODOLOGY
This review was conducted through a narrative synthesis of peer-reviewed literature identified via Google Scholar and PubMed. Search terms were developed around core WBE themes including WBE, wastewater surveillance, community health monitoring, data normalization, health equity, and specific biological/chemical analyte classes including SARS-CoV-2, narcotics, and antimicrobial resistance. No formal date restrictions were applied, though emphasis was placed on literature published from 2015 onward to reflect the contemporary state of the field, with foundational works cited where historically relevant. Reference lists of key publications were reviewed to identify additional sources. Given the cross-disciplinary scope of this review, manuscript inclusion was guided by relevance to the central objective of evaluating strategies to improve the utility and value of WBE data rather than by strict methodological criteria. Studies were included if they contributed to the evidence base for any of the thematic areas addressed, including analytical capability, standardization, data modeling, community equity, or stakeholder engagement.
All figures presented in this manuscript are original works created by the author. Figure 1 and the graphical abstract were created using Microsoft PowerPoint. Figures 2 and 3 were generated programmatically using Python in Google Colab, with coding assistance provided by Claude (Anthropic). Table 1 was constructed in Microsoft Excel. No figures were reproduced or adapted from previously published works, and no third-party templates or symbol libraries were used in their creation.
Figure 1. Decision tree for selecting between RT-PCR and ddPCR platforms in wastewater surveillance programs. Green boxes indicate scenarios where RT-PCR is sufficient or preferred, blue boxes indicate where ddPCR is preferred or strongly recommended, and yellow boxes indicate either platform is viable. Key decision points include data output requirements, target abundance expectations, PCR inhibition prevalence, sample throughput needs, inter-laboratory data harmonization requirements, budget constraints, and anticipated participation in the NWSS. RT-PCR: Reverse transcription polymerase chain reaction; ddPCR: droplet digital polymerase chain reaction; NWSS: National Wastewater Surveillance System.
Figure 2. (A) Absolute number of NWSS-participating counties in each median household income bracket in 2023 (n = 584) and 2025
Figure 3. Net change in NWSS county coverage by median household income bracket from 2023 to 2025. Green bars indicate net gains in participating counties while red bars indicate net losses, demonstrating that program expansion heavily favored higher-income communities (≥ $60 K) while lower-income counties (< $60 K) experienced stagnation or decline, with the $40-50 K bracket losing 25 counties. NWSS: National Wastewater Surveillance System.
Proposed cross-domain applications of wastewater surveillance integrating chemical and biological targets for comprehensive community health assessment
| Application | Chemical targets | Biological targets | Cross-domain value | Supporting evidence |
| Substance use + associated disease burden | Opioids, stimulants, benzodiazepines + antiretroviral drugs (tenofovir, emtricitabine) + HCV antivirals (sofosbuvir) | Hepatitis C RNA, HIV RNA | Links substance use patterns to disease prevalence AND treatment adherence in affected populations; identifies gaps in harm reduction services | Substances[14,15,50] ARVs[51-53] Hepatitis[54,55] HIV[56] |
| Lockdown mental health impact | Cortisol metabolites, alcohol biomarkers, tobacco metabolites, antidepressants (SSRIs), anxiolytics | SARS-CoV-2 variants, influenza (for comparison to pre-pandemic baselines) | Quantifies biological disease burden alongside chemical indicators of population stress, coping mechanisms, and mental health treatment utilization | COVID[6,57,58] Stress hormones[23] Pharmaceuticals[59,60] |
| Foodborne outbreak + exposure source | Pesticide residues, food additives, mycotoxins | Norovirus, Salmonella, E. coli pathotypes, hepatitis A | Links pathogen detection to chemical signatures of food contamination; helps identify outbreak sources (agricultural runoff, food processing) | Norovirus WBE[61,62] Pesticides:[18,19,63] Foodborne pathogens[64,65] |
| Antimicrobial stewardship | Antibiotics (fluoroquinolones, beta-lactams, macrolides), antifungals, disinfectants (triclosan, quaternary ammonium compounds | AMR genes (blaNDM, mcr-1, vanA), pathogenic bacteria markers (STEC, C. difficile) | Correlates antimicrobial use with resistance emergence; evaluates intervention effectiveness; detects hospital vs. community sources | Antibiotics in WW[66] AMR genes[27,31,67] Pathogen markers[43,58,68] |
| Environmental exposure + immune vulnerability | PFAS, phthalates, heavy metals, air pollution markers (PAH metabolites) | Respiratory viruses (RSV, influenza, COVID-19), inflammatory biomarkers (if detectable) | Assesses how environmental contaminant exposure may influence population susceptibility to infectious disease during seasonal surges | PFAS[69,70] Heavy metals[59,71] Respiratory surveillance[17,39,72] |
| Veterinary-human health interface | Veterinary antibiotics, livestock hormones, pesticides from agriculture | Zoonotic pathogens (Campylobacter, Cryptosporidium), AR genes of agricultural origin | Monitors One Health concerns in communities with intensive agriculture; tracks zoonotic spillover risk and agricultural AMR contribution | Vet drugs[73,74] Zoonotic pathogens[75-77] Agricultural AMR[78-80] |
EXPANDING ANALYTICAL CAPABILITY
Narcotics consumption surveillance and SARS-CoV-2 surveillance are the two most well-developed aspects of WBE, but represent only a fraction of the field’s demonstrated applications. Recent projects have begun to expand the breadth of WBE analyses to additional communicable diseases, environmental exposures, and endogenous biomarkers. One straightforward way to improve the utility and value of wastewater data is to expand the target analytes to include chemical or biological indicators linked to high-impact human health threats. Multiple analytical methods could be applied to a single wastewater sample to maximize the information obtained, which could increase the value and return on investment (ROI) of wastewater surveillance projects.
While there are many efforts focused on the expansion of WBE analytical capabilities, there is perhaps less attention being dedicated to creating synergistic projects leveraging both biological and chemical analyses. For instance, a SARS-CoV-2 surveillance project during a lockdown could be improved by also screening for endogenous stress hormones, narcotics consumption indicators, alcohol consumption indicators, and tobacco consumption indicators [Table 1]. Such a design would not only provide insight into the impact of a lockdown on disease spread but also provide an understanding of stressors and coping mechanisms that individuals in these communities may be subjected to in such circumstances. While at least one project has successfully demonstrated this proof of concept through the correlation of residual antibiotic concentrations and the relative abundance of their respective antibiotic resistance genes (ARGs) in hospital wastewater[81], WBE studies that formally integrate chemical and biological targets remain scarce, representing a significant gap and an opportunity for future cross-domain surveillance work. Projects which utilize a wider array of assays will ultimately produce a greater quantity of data which likely will increase the utility of the data across multiple professions. Furthermore, advanced mass spectrometry techniques, including non-targeted and isotope-resolved analyses, can add meaningful new layers of value to both existing and novel wastewater surveillance programs. Approaching projects in this way may increase the adoption of wastewater surveillance programs and support the long-term sustainability of these efforts.
Consideration should also be paid to on-site wastewater testing and the use of in situ chemical and/or biological sensors for deployment in wastewater collection networks. Current WBE methods are able to provide information in near real time, ideally within 48 to 72 h following the collection of a wastewater sample[82]. It is often recommended to use overnight shipping to send samples to a WBE laboratory, resulting in a delay of 12-24 h depending on the time the wastewater sample was originally collected. Utilizing on-site testing would remove this delay and result in data being provided faster, but true real-time wastewater data may only be accomplished through the integration of in situ sensors[72,83-86] and Internet of Things (IoT). If in situ sensors can reliably detect target analytes at the concentrations present within wastewater, the value and utility of wastewater data will increase exponentially. Not only will true real-time data provide the best information for community stakeholders, but it can also be integrated via IoT for complex dataset analysis, analysis by artificial intelligence (AI), and digital twin technologies. Such advancements would substantially transform the capabilities of wastewater surveillance.
LABORATORY PROCESS AND REGULATION
As WBE programs still operate without regulatory guidance, there can be significant differences in the analytical approaches that may be utilized for a project. With respect to SARS-CoV-2 polymerase chain reaction (PCR) analyses, users of WBE data may receive a qualitative analysis (presence or absence of the SARS-CoV-2 gene) or a quantitative analysis (viral copies per liter of wastewater). While a qualitative analysis may be sufficient in select situations, it does not allow data users to understand the temporal fluctuations of the virus. Thus, the utility of qualitative WBE data may be limited to small wastewater catchment regions with minimal transient populations. Conversely, many users of wastewater data would benefit from quantitative measurements - but the quality of quantitative WBE data relies heavily on the technical rigor of laboratory personnel conducting the analyses.
Differences in laboratory instrumentation may also induce unintended data biases or influences that may not be immediately apparent to the end user. To date, a significant number of SARS-CoV-2-focused WBE projects have utilized reverse transcription PCR (RT-PCR) for both qualitative and quantitative analyses[87,88], but published evidence suggests that variability in reverse transcription quantitative PCR (RT-qPCR) assay parameters renders quantification via RT-qPCR unreliable for wastewater surveillance purposes[89]. Advancement of the digital PCR (dPCR) and droplet digital PCR (ddPCR) platforms may provide a viable alternative to traditional RT-qPCR approaches. These digital platforms perform an absolute quantitative measurement of genetic material in a sample independent of a standard curve resulting in a more reliable and consistent quantitative measurement[90]. Furthermore, the increased signal-to-noise ratio and lessened impact of PCR biases[90] inherent to dPCR and ddPCR instrumentation may provide advantages when analyzing complex environmental matrices such as wastewater. Current studies leveraging these dPCR platforms have noted increased analytical sensitivity of dPCR platforms compared to traditional RT-qPCR approaches[91-93], and publication activity referencing digital and ddPCR applications to WBE has increased year-over-year since 2020. It is important to note that while dPCR platforms may offer several advantages over traditional RT-qPCR platforms, the selection of the appropriate instrumentation is likely project-specific [Figure 1]. The higher instrumentation costs of digital platforms may not be ideal for projects with budget constraints, especially within economically disadvantaged communities and developing nations. Furthermore, the greater analytical sensitivity of digital platforms may not be necessary within samples where high viral loads are expected, or where quantitative measurement is not needed. All instrumentation should be considered on a project-by-project basis, and the appropriate instrumentation selected based on budgetary limitations and project necessities.
As wastewater surveillance projects become more commonplace, it will be necessary for regulations to be created, adopted, and enforced for WBE practices. The CDC provides guidance for wastewater sampling, testing, and data reporting and analytics[94] - primarily through its standardized Wastewater Viral Activity Level (WVAL) metric, which enables cross-site comparison by normalizing current viral concentrations against site-specific baselines established from 24 months of historical data. The WVAL methodology, updated in August 2025, utilizes non-normalized concentration data with log-transformation and outlier detection to categorize viral activity into five levels (very low to very high) using virus-specific thresholds for COVID-19 (coronavirus disease 2019), influenza A, and RSV. The CDC recommends twice-weekly sampling with a minimum eight-week data accumulation period before public reporting, and implements data privacy protections including a 3,000-person sewershed minimum and geographic coordinate approximation. However, these recommendations remain voluntary rather than enforceable regulations, and laboratories performing wastewater surveillance are not subject to the same regulatory oversight and proficiency testing requirements that govern clinical diagnostic or environmental testing laboratories[95]. Lack of regulations and method standardization across the field may lead to inconsistencies in data generated from different laboratories that may not be apparent to the end user. This could cause community stakeholders to draw incorrect conclusions from WBE data and act on flawed or inefficient strategies. This is not only a significant negative consequence for the end user but also a negative consequence for the field of WBE as wastewater data could be viewed as an inefficient use of limited funds and resources. Therefore, it becomes incumbent upon all WBE professionals to insist on and assist with the development of regulations and oversight for the field of wastewater surveillance.
While PCR-based platforms have historically dominated biological surveillance workflows, chemical analyses targeting parent compounds and metabolites of high-risk substances have relied primarily on liquid chromatography-tandem mass spectrometry (LC-MS/MS), an analytical platform capable of small-molecule detection at the parts-per-trillion (ppt) level[96]. These sensitivities are essential in surveillance contexts focused on highly potent narcotic adulterants, including xylazine, fentanyl analogues, and nitazenes, compounds associated with substantially elevated overdose potential and a range of acute and chronic health impacts[97]. Analogous to biological methods, chemical analytical workflows consist of two fundamental steps: (i) sample preparation, typically achieved through solid-phase extraction (SPE) and liquid-phase reconstitution; and (ii) mass spectral analysis.
Wastewater represents a chemically complex environmental matrix, containing numerous co-occurring compounds and interfering constituents that can suppress or obscure target analyte signals. SPE addresses this challenge by exploiting the physicochemical affinity between a sorbent resin and target analytes, enabling selective retention and concentration of compounds of interest while removing matrix interferences that would otherwise compromise analytical performance[98]. The choice of SPE sorbent chemistry is therefore a consequential methodological decision, as it directly determines both capture efficiency and the breadth of analytes that can be reliably recovered. Within the WBE literature, two sorbent strategies have emerged as predominant. Broad-spectrum sorbents, such as the Oasis Hydrophilic-Lipophilic Balance (HLB) cartridge, leverage balanced hydrophilic and lipophilic retention mechanisms to maximize analyte coverage across acidic, neutral, and basic compound classes[99]. In contrast, targeted sorbents such as the Oasis Mixed-Mode Cation Exchange (MCX) cartridge exploit dual reversed-phase and ion-exchange interactions to achieve superior selectivity and recovery for basic compounds[98], a class that encompasses the majority of illicit drugs and their metabolites commonly quantified in WBE studies[1,14,15]. The selection of SPE sorbent chemistry therefore represents an inherent methodological trade-off between analyte breadth and capture specificity[96], with implications for both method sensitivity and the interpretive scope of resulting surveillance data.
Recent research has demonstrated the utility of non-targeted analysis (NTA) as a complementary approach for broad-spectrum qualitative screening in wastewater surveillance[100]. Rather than querying a sample for a predefined list of analytes, NTA acquires full-scan accurate-mass spectra across the entire detectable chemical space, which are then interrogated against libraries of thousands of known compounds[96]. This approach offers two distinct advantages for WBE programs. First, NTA can characterize the full complement of detectable compounds present in a wastewater sample, providing an empirical basis for prioritizing which targeted LC-MS/MS methods warrant further development. Second, NTA data constitute a digitally preserved chemical record of the sample at a given point in time, one that is not subject to the physical degradation constraints of archived sample material. Should a novel or previously unrecognized adulterant be identified in illicit drug supplies at some future date, archived NTA datasets can be mined to establish when and where that substance first appeared in the wastewater signal, a retrospective capability with significant public health implications[100].
DATA MODELING
With respect to the end user, data normalization, modeling, and visualization are arguably the most important aspects of WBE. Raw instrument data (commonly in units of mass per volume) may not be intuitive or informative to individuals not well-versed in wastewater surveillance. Applying normalization and modeling to wastewater data increases both data understanding and utility to a wider array of professionals and laypersons. Select public-facing wastewater dashboards exist for both opioid consumption surveillance[101] and monitoring of SARS-CoV-2[102-104]. These dashboards visually summarize wastewater surveillance data to enable users to quickly understand changes in health concern prevalence, long-term trends, correlations with external events and circumstances, and enable cross-comparison between different sampling locations. While many dashboards and projects focus on sample collection at the wastewater treatment plant, projects such as the Tempe Opioid Wastewater Collection Data Dashboard provide a higher spatial resolution by sampling and reporting data from strategic sampling locations along the city’s wastewater collection network[101]. While health departments and governments have historically been the primary users of wastewater surveillance data, data modeling and normalization increases the opportunity for the data to be leveraged by a wider array of professionals and community stakeholders.
Traditional normalization approaches in wastewater surveillance have relied on several distinct methodologies, each with varying levels of complexity and associated costs. Physical normalization utilizing wastewater flow measurements provides a straightforward approach to account for dilution effects from stormwater infiltration or variable contributions across different times of day, while population-based normalization has historically used census data or sewershed service population estimates to standardize concentrations on a per-capita basis[105-107]. Chemical biomarkers such as creatinine, ammonia, phosphate, conductivity, and chemical oxygen demand (COD) have been employed to normalize for the number of individuals contributing to a sample, with each marker offering different advantages depending on temporal stability and susceptibility to confounding factors such as industrial inputs or dietary variations[108-110]. Biological indicators including pepper mild mottle virus (PMMoV) and crAssphage have gained traction as population normalization markers due to their ubiquitous presence in human fecal matter and relatively stable excretion patterns across diverse populations[2,57]. More recently, mobile device usage data and cell phone ping analytics have been successfully integrated into normalization strategies, particularly for sewersheds serving transient populations or areas with significant tourism and commuter contributions that may not be accurately captured through residential population estimates alone[111,112]. The selection of an appropriate normalization approach requires careful consideration of the balance between analytical accuracy and the practical costs associated with obtaining normalization data, as comprehensive multi-parameter normalization may theoretically provide the most robust standardization but can substantially increase program costs through additional laboratory analyses, data acquisition fees, or infrastructure requirements. Furthermore, it is important to recognize that normalization inherently introduces additional sources of uncertainty and potential error into wastewater surveillance data, as each normalization parameter carries its own measurement variability, temporal fluctuations, and assumptions about population behavior or wastewater characteristics that may not hold true across all sampling locations or time periods. The extent and complexity of normalization should therefore be carefully weighed against the intended use of the data and the decisions that will be informed by the results, with minimal normalization potentially sufficient for trend detection at a single site over time, whereas cross-site comparisons or attempts to estimate absolute prevalence metrics may justify more sophisticated normalization strategies despite the added complexity and error propagation.
Central to wastewater surveillance modeling and normalization is an understanding of the in vivo kinetics and dynamics of both biological and chemical WBE targets. Narcotic consumption WBE projects commonly convert mass loads to more tangible values such as the estimated amount of narcotic consumed, the estimated number of narcotic users, black market value of observed narcotics, and estimated overdoses and overdose-deaths[15]. These estimations are only possible due to the significant volume of pharmacokinetic research surrounding various pharmaceuticals and illicit narcotics. Pre-2020 wastewater surveillance literature primarily focused on high-risk substances with known and well-established pharmacokinetic profiles, such as cocaine[113-115], heroin[15,116,117], fentanyl[15,118], xylazine[74], nitazenes[119,120], and pharmaceuticals with a high associated risk of abuse[121,122], making downstream data modeling a more straightforward exercise. With the emergence and rapid onboarding of biological methods for COVID-19, there was a significant gap in understanding the urinary and fecal shedding rates of an individual infected with SARS-CoV-2[123-125]. While the monitoring of viral load increases/decreases at a single WBE site would not be impacted by this, problems arise when trying to cross-compare sampling locations, or estimate the number of infected individuals within a catchment area via the observed wastewater load[126,127]. Correlating wastewater concentrations to published clinical case count data would result in an underestimation due to the number of asymptomatic and mildly symptomatic individuals who may not undergo clinical testing procedures. Even when preliminary fecal shedding rates were first published, significant variability in the viral shedding load was observed across individuals, sometimes spanning multiple orders of magnitude[57,128,129]. Even across well-established pharmacokinetic and chemical urinary excretion studies, notable variation across individuals’ excretion rates has been noted[130,131]. While data modeling and extrapolation may improve the tangibility of wastewater surveillance data across a wider breadth of professions and laypersons, such estimations also introduce sources of notable error that are not inherent to raw analytical measurements. The appropriate degree of data modeling is a function of data availability, current understanding of in vivo kinetics, and project goals, and must be evaluated on a case-by-case basis. Regardless of these limitations, it will still be important that research focuses on understanding the fecal excretion profile of the SARS-CoV-2 virus and other communicable diseases which are measured through wastewater surveillance. Providing data in a digestible form broadens the potential users and value of WBE projects and may encourage long-term project retention.
Study design and sampling site selection meaningfully shape the data produced by WBE programs. Historically, sampling has occurred at three levels of spatial resolution: (i) at the wastewater treatment plant (WWTP) influent; (ii) at intermediate network infrastructure such as manholes, lift stations, and pump stations; and (iii) at building-level access points that allow isolation of specific structures. Programs operating at the WWTP capture the broadest possible population catchment[132] but face the highest degree of analyte degradation, driven by temperature and enzymatic hydrolysis leading to deconjugation over extended hydraulic residence times (HRTs)[133-137]. Despite this limitation, plant-level data have demonstrated clear value to public health agencies at the state and federal level[138], where broad surveillance and wide-reaching policy decisions require population-scale signals rather than granular ones[139].
Higher-resolution programs reduce HRT prior to sample collection, limiting analyte decay and tending to produce more actionable datasets for institutional stakeholders[133,140]. Decision-makers in settings such as schools or correctional facilities can respond to site-specific signals with interventions that produce faster, more measurable impact[141-143]. The tradeoff is cost - higher spatial resolution has historically corresponded with substantially higher per-sample expenditure[144]. There is no universally correct approach, and sampling site selection must ultimately be guided by the program’s purpose and the resources available to support it.
As wastewater surveillance projects become more commonplace, it is likely that the target biological and chemical analytes will also increase in breadth. As with SARS-CoV-2, it will be important to understand the in vivo kinetics and dynamics of each analyte that is monitored via wastewater surveillance. Equally important will be understanding the in situ dynamics of target analytes within wastewater collection networks. It is well-known and documented that analyte degradation due to physical, chemical, and biological processes within sewer networks has an impact on wastewater surveillance data[15,59] - but historically the field has not accounted for this analyte loss through back-calculation. While degradation at a single sampling location may be relatively constant over time, the impact may be substantially greater when conducting a cross-comparison between locations with significantly different wastewater HRTs. Sampling sites with shorter HRTs may appear to have higher concentrations than sites with longer HRTs due to the higher analyte degradation profile inherent to large wastewater catchment areas. Studies have attempted to quantify the degradation profile of various chemical and biological analytes within wastewater collection networks[133,140], which are useful for estimating the temporal degradation of a target analyte within a wastewater collection network. While accounting for degradation in data modeling is important, it is equally important not to “over-engineer” the equations used for back-calculation. Degradation rates for different locations may be heavily influenced by factors such as the percentage of industrial input, stormwater infiltration rates, and biological and/or chemical composition of the wastewater. As proposed in the paper by Hart and Halden, estimating an average temporal degradation rate by first-order kinetics may be a favorable method of accounting for in situ analyte degradation[133].
The rapid advancement of AI and machine learning (ML) provides an additional opportunity to maximize the value obtained through wastewater surveillance programs. ML techniques have historically been applied across WBE projects, which include population normalization, wastewater flow normalization, fecal indicator normalization, predictive modeling to forecast disease incidence, hospitalizations, and antimicrobial resistance gene prediction[15,145,146]. While much of the growth observed in WBE occurring in the early 2020s was in response to the SARS-CoV-2 pandemic, subsequent years saw the emergence of increasingly sophisticated analytical frameworks. For example, Mathematica’s development of its CovidSURGE algorithm in 2023[147] demonstrated the potential of data-driven approaches to support traditional interpretation of WBE data sources.
More recently, researchers have proposed using trained AI/ML models to evaluate hypothetical policy interventions[148], positioning WBE data as not only a public health surveillance tool but as an additional consideration for public health decision-making. One significant advantage of AI/ML integration is the speed at which complex datasets can be analyzed, reducing a process that once took weeks or months to minutes. This acceleration directly enhances the public health value of WBE by shortening the time between sample collection and actionable insight[148,149]. This has been demonstrated in practice, with Xu et al. showing that large language models (LLMs) adapted for water and wastewater applications can achieve prediction accuracies exceeding 95% on complex domain-specific tasks[150], and Sun et al. identifying that LLM-based data agents can autonomously handle complex analytical workflows with minimal technical expertise required[151].
Another opportunity arises with the convergence of modern AI/ML techniques, digital twin technologies, and in situ sensors, enabling advanced modeling and forecasting techniques approaching real-time. With a comprehensive network of in situ sensors that could detect target analytes to the ppt or ideally parts-per-quadrillion (ppq) level, coupled with digital twin models of existing wastewater infrastructure networks, all integrated via IoT, unique degradation profiles could be generated for individual analytes with respect to the unique characteristics of that specific sewershed instead of relying on rough first- or second-order decay equations to estimate degradation rates. Such programs could substantially improve data generated from WBE methods, especially those focused on the WWTP. While such a WBE application has not yet been attempted, Zehnder et al. demonstrated the theoretical possibility of rapid back-tracing of biomarkers in a simulated wastewater network[152]. Such networks would also have substantial value to the larger environmental forensics field when engaging in activities such as identification of an entity that is illegally discharging contaminants into a wastewater system.
Beyond analytical computation, recent work has also examined how surveillance networks themselves can be optimized. Downsampling strategies, including enumerative and iterative hierarchical approaches, have been explored as methods to identify the minimum sampling site density and frequency required to preserve observed transmission trends, offering a pathway to cost-effective network design without sacrificing public health utility[153].
DATA TRANSPARENCY AND WBE ETHICS
Data transparency is paramount to the scientific advancement of the field, the preservation of ethical considerations, and is essential to maximizing the utility of the data. Several ongoing WBE projects and WBE research groups provide a public-facing source of data[154,155], but the transparency of this data can vary considerably. Some public-facing sources of data provide limited datasets focusing on either the presence/absence of indicator compounds or percent changes at a specific location over time. Other public-facing wastewater data sources report values normalized to observed wastewater flow and/or contributing population. While the former translates wastewater data to a format that may be more understandable to a layperson or professional outside the field of WBE, the latter ultimately provides a more useful source of data to researchers and professionals conducting WBE meta-analyses or trying to compare wastewater data from across multiple sampling locations.
The noted variation in the transparency of WBE data is likely influenced by many factors, including but not limited to sensitivities around personally identifiable information (PII), perceived risks and/or potential liabilities, national/regional security concerns, and political climates. Addressing these factors through both regulation and community education offers opportunities to dispel concerns related to wastewater surveillance practices and institute a greater number of WBE projects. As the number of projects increases, it will be important to institute a level of transparency across these projects to ensure continued community buy-in and maximize the efficacy of the resulting data produced. It will be equally important to ensure that wastewater data is not used in ways that can negatively impact the communities that projects are created to serve. Data mining has become a common practice among large organizations to improve their products, increase profits, and drive forward business development goals. While these practices certainly provide value to an organization, they have recently come under scrutiny from organizations such as the Federal Trade Commission due to the data use practices and ethics surrounding these near-ubiquitous practices[156]. Wastewater surveillance may be especially prone to unethical data practices due to the significant breadth of population health and activity information that can be obtained through the method. Furthermore, residents within a community cannot “opt-out” of wastewater data collection.
Additional ethical considerations must be addressed throughout the entire wastewater surveillance process to safeguard the privacy and civil liberties of individuals who reside and work within monitored catchment areas. The field has generally operated under a “minimum contributing population” principle[157], though significant variation exists in the proposed minimum number of contributing individuals[2,158,159]. This threshold approach, however, could limit valuable building-level surveillance applications such as antimicrobial resistance monitoring at hospitals and healthcare facilities, where contributing populations often fall below even the most conservative published thresholds. Rather than relying solely on minimum population thresholds, leveraging established or community-engaged ethical frameworks throughout the WBE process may provide a more structured yet adaptable approach to ethical oversight. The success of such frameworks has been demonstrated by Clarke et al. through their community-engaged bioethics model applied to the Rubbertown Air Toxics and Health Assessment (RATHA) program[160], and by Arefin et al. in their application of Nancy Fraser’s framework of Justice to evaluate site-specific practices, identifying maldistribution, misrecognition, and exclusion[161]. Adopting these framework-based approaches may address situational ethical concerns without unnecessarily limiting the development of otherwise valuable wastewater surveillance applications.
While AI/ML and LLM technologies have clear roles in data analysis, modeling, and visualization, they also present meaningful opportunities to strengthen the ethical frameworks and transparency infrastructure of WBE programs. Explainable AI (XAI), a set of processes and methods developed to allow human users to comprehend, trust, and interrogate AI-generated outputs, has been proposed as a transparency mechanism in healthcare and epidemiological surveillance contexts[162] and could be similarly applied to wastewater surveillance programs. Beyond interpretability, AI/ML tools could be leveraged to automatically identify geographic and demographic coverage gaps across surveillance networks, providing an equity-focused audit mechanism that operates independently of manual review. These same capabilities extend naturally to data governance, where automated audit trail generation across the full analytical pipeline, from sample collection through to public health reporting, would address a critical accountability gap. Given that wastewater surveillance data informs population-level public health decisions and policy, a verifiable and reproducible record of data provenance, processing decisions, and model outputs is not merely useful but necessary. Importantly, the value of AI/ML in this space is not limited to the applications described here, and continued development of these tools must remain grounded in the ethical principles and community considerations that define responsible wastewater surveillance practice[163].
In the absence of regulation, it becomes imperative that individuals and organizations operating in the field of WBE adhere to ethical considerations throughout all projects. This includes but is not limited to (i) transparency with analytical methods, target analytes, and project goals; (ii) adhering to a “minimum population threshold” in selection of sampling locations; (iii) establishing clear contracts and material transfer agreements which prevent the use of samples and data outside the scope of the intended purposes; (iv) championing public buy-in and ongoing project support; and (v) adhering to ethical business practices. If such considerations are not adhered to, it is possible that the public perception of wastewater surveillance could sour, which may result in a reduction in the adoption of the methodology or complete nonparticipation in the process altogether. Unsuccessful legislation preventing wastewater surveillance has already been proposed in the U.S. at the state level[164], and such initiatives could gain traction if wastewater surveillance data is used for controversial or unethical purposes. Therefore, the future of the field of WBE hinges on researchers and organizations conducting their wastewater surveillance projects transparently and ethically.
SERVING NEEDS-BASED COMMUNITIES AND PROMOTING UNIVERSAL HEALTH EQUITY
Wastewater surveillance provides a significant economic advantage over other methods of health data collection, often costing significantly less than other commonly used methods (such as individual clinical testing)[19,33,165,166]. This makes the methodology especially appealing to needs-based communities and developing nations[167,168], but these types of communities have not experienced the same level of WBE adoption as developed nations and well-resourced communities[168]. Even within the United States, many smaller communities, rural areas, and underserved communities have not experienced the same access to wastewater surveillance as larger cities. To better understand the coverage and trajectory of the NWSS program, an economic analysis was performed on participating NWSS communities in Q1 2023 and Q4 2025. County-level wastewater surveillance site information was obtained from the NWSS obtained in 2023 and 2025[94]. The primary source for median household income data was the U.S. Department of Labor’s Unemployment Statistics dataset, which provides 2022 median household income estimates derived from the USDA Economic Research Service (ERS) County-level Data Sets[169]. For jurisdictions not included in the primary dataset, supplemental data were obtained from individual searches. Connecticut county income data (Fairfield, Litchfield, New Haven, New London, and Tolland counties) were sourced from the Census Bureau’s American Community Survey (ACS) 5-year estimates (2022) via Data USA and Neilsberg analytics platforms[170]. Guam’s median household income was obtained from the Census Bureau’s ACS estimates for U.S. territories[171]. Border counties misattributed in the original NWSS data (Cumberland County, ME and Oxford County, ME) were corrected using the primary dataset. Some counties listed in the NWSS dataset were not actually located within their associated state but were identified as border counties in adjacent states; in these cases, it was assumed that the border county represented the correct jurisdiction for purposes of this analysis, likely reflecting wastewater treatment plants that serve cross-border populations.
The CDC’s NWSS program coverage has grown from nearly 600 counties in 2023 to over 750 counties by 2025 [Figure 2A][104]. However, this expansion has not been evenly distributed across income levels. In 2023, the distribution of participating counties was concentrated in the $50-60 K and $60-70 K median household income brackets, which together accounted for over 53% of all participating counties. By 2025, the distribution shifted upward, with the $60-70 K and $70-80 K brackets now representing nearly 48% of participants [Figure 2B]. The largest net gains in county participation between 2023 and 2025 occurred in the $70-80 K (+60 counties), $60-70 K (+59 counties), $80-90 K (+35 counties), and $90-100 K (+17 counties) brackets [Figure 3]. In contrast, lower-income brackets experienced net losses, with the $40-50 K bracket declining by 25 counties and the < $30 K, $30-40 K, and $50-60 K brackets each losing one to two counties. While the overall growth of the NWSS program represents an important achievement for public health infrastructure, the expansion has not proportionally increased representation among lower-income communities. As a percentage of total participants, counties with median household incomes below $50,000 declined from approximately 11.7% in 2023 to 5.3% in 2025, while counties above $80,000 grew from approximately 17.9% to 22.7%[94,169].
It is important to note that the U.S. NWSS program is a dynamically evolving federal initiative, born out of emergency response to the COVID-19 pandemic and built through collaboration across the CDC, state and local health departments, academic institutions, non-profit organizations, and commercial contractors. Coverage gaps, where they exist, reflect the inherent complexity of establishing a national surveillance infrastructure, with funding availability, wastewater infrastructure, and programmatic priorities all playing a role rather than any single decision-maker or institution. With this being considered, understanding these trends in the program’s natural evolution should inform future resource allocation decisions as the NWSS continues to mature. Several communities currently participating in the program may be able to fund similar wastewater surveillance efforts through alternative funding mechanisms. The City of Tempe, in partnership with Arizona State University, funded its initial wastewater opioid surveillance efforts through the Tempe Innovation Fund[172]. If larger municipalities and well-resourced communities can fund wastewater surveillance projects through alternative means that do not rely on federal funding, a larger portion of federal dollars could be dedicated to increasing wastewater surveillance presence in needs-based communities. This would not only serve as an important effort in helping bridge disparities in health equity but also provide the United States with a more comprehensive wastewater surveillance program.
A core vision of wastewater surveillance has historically centered on developing a cost-effective global environmental monitoring network. While meaningful global growth has occurred, evidence suggests that low- and middle-income countries (LMICs) have experienced substantially less program development compared to more economically advantaged nations across North America, Europe, and Australia[173,174]. This disparity likely reflects a combination of factors. The earliest wastewater surveillance programs in all three of these regions predated the global expansion driven by the COVID-19 pandemic[1,14,175], meaning considerable field expertise was already established in these regions, but socioeconomic factors have likely continued to compound this gap. Political orientation has been shown to influence WBE adoption rates across the United States[159,176,177], and it is reasonable to expect that political considerations have similarly affected uptake in select LMICs.
Drawing from field experiences across Bangladesh, Ghana, Malawi, and South Africa, Truyens et al. noted that while 81% of the global population resides in LMICs, the vast majority of wastewater surveillance success stories during the COVID-19 pandemic emerged from high-income countries, with LMIC attention remaining largely focused on polio-specific surveillance despite substantial risk from other epidemic-prone pathogens[178]. Furthermore, Ghana leveraged existing polio surveillance infrastructure to pilot COVID-19 wastewater monitoring between November 2020 and May 2021 with philanthropic support, but samples collected outside the capital required batching, cold storage, and weekly shipment for analysis. This logistical burden substantially undermined the timeliness that otherwise may have been achieved while giving wastewater surveillance its public health utility[179].
Resource availability represents another significant barrier. LMICs have historically faced limited access to both economic and physical resources[178,180,181], which constrains WBE program development along several dimensions. First, there is the direct challenge of funding, as wastewater surveillance programs carry meaningful costs in personnel time, instrumentation, and consumables[182,183]. A survey of twelve wastewater monitoring programs across LMICs found that per-sample costs varied considerably, ranging from $34/sample in India to $517/sample in Costa Rica. The recurring costs across these programs were between 19% and 93% of total program expenditures, with funding primarily coming from government and philanthropic sources. This underscores the degree to which LMIC programs remain dependent on external financial support rather than sustained domestic investment[49]. Second, where instrumentation does exist within LMIC laboratories, it is typically already dedicated to an existing purpose, such as clinical RT-qPCR analysis, and cannot easily be repurposed for environmental applications[178]. Third, even where infrastructure and funding are present, many LMIC laboratories struggle to obtain necessary consumables due to cost, limited availability, and import barriers[184].
Beyond funding and equipment, the physical infrastructure of wastewater collection in LMICs introduces an additional layer of complexity. Many LMICs rely on rudimentary systems such as pit latrines rather than the centralized treatment infrastructure common in developed countries[68], meaning that even pilot-scale surveillance programs require substantially greater planning and personnel investment to deploy. A multi-matrix environmental surveillance study in Maputo, Mozambique, detected a broad range of pathogens across multiple wastewater treatment-associated matrices, revealing exposure pathways which conventional surveillance methods failed to observe[185]. These studies suggest that the barriers to WBE equity in LMICs extend beyond economics; considerations including population density, education level, gender representation, and community engagement remain largely unaddressed in WBE contexts. Consideration of these aspects is essential for the equitable development of WBE programs in underserved populations.
Interestingly, development of a global wastewater surveillance network through the analysis of international aircraft and airport effluent has been proposed in recent years as a method of global pandemic monitoring. The CDC developed the Traveler-Based Genomic Surveillance program in 2023 as an exploratory pilot project to determine the efficacy of monitoring SARS-CoV-2 variants of concern entering the U.S. from international travel[186]. The focus of this strategic biosurveillance project was three-fold: (i) to enable the timely detection of communicable diseases of public health concern; (ii) to limit the spread of communicable diseases to local communities; and (iii) to reduce the need for border interventions and disruptions to travel and trade. Similar programs were adopted across airports outside of the United States as well[67,187-189]. In a recently published article, St-Onge et al. demonstrated that establishment of 10 to 20 strategically located global wastewater sentinel sites at select airports would effectively function as a global early warning system for pandemic readiness[190]. Similar programs have also been suggested within other forms of common international travel, such as cruise ships[58,191,192]. This application illustrates how continued innovation in WBE can yield more economical tools capable of serving underserved populations globally.
As wastewater surveillance efforts continue to expand, it is imperative that both domestic and international needs-based communities are not overlooked. Select projects have increased wastewater surveillance access to underserved communities, including areas with high poverty rates in the United States[193,194], tribal communities[5], and developing nations[68]. These projects provide significant advantages that extend far beyond the immediate communities that they serve. With the highly transient nature of today’s society, human health threats such as communicable diseases are a global concern and thus must be monitored globally. Nearly 83% of the world’s population resides in developing countries[195], and thus understanding public health in these areas is essential in the global management of a human health crisis such as the SARS-CoV-2 pandemic. The cost-effective nature of wastewater surveillance makes it an ideal method to continuously monitor health threats in needs-based communities.
CONCLUSION
Increasing the value and subsequent utility of wastewater data is essential to securing the long-term viability of the field. While the SARS-CoV-2 pandemic provided the opportunity for increased adoption and recognition of the methodology, there is uncertainty regarding the future direction that WBE will take. It is likely that wastewater data could be pertinent to many fields and professionals not directly linked to public health - but it will take a reexamination of experimental design and laboratory processes to adapt the technology for new applications and user communities. This manuscript has proposed approaches including expanding analytical methodology, standardizing laboratory processes, instituting regulatory oversight, improving data modeling and visualization, adhering to minimum transparency standards, and serving needs-based communities to bridge healthcare equity gaps as options to improve the value of WBE data. It is important to consider that these suggestions may not increase the value or utility of WBE data in all situations, nor are they a comprehensive list of all potential methods for increasing the value and utility. It will take a coordinated effort across government, industry, and academia to ensure that future WBE efforts are robust, scientifically defensible, and serve the community to the fullest extent possible.
It is important to note that the near-term feasibility of these recommendations varies considerably. Standardizing laboratory processes, improving data modeling and visualization, and adhering to minimum data transparency standards represent the most immediately actionable priorities, as the infrastructure and knowledge necessary to advance these goals are already well established across the field. Expanding biological and chemical methodologies and increasing surveillance coverage in needs-based communities are equally essential, but require substantial investment and strategic cross-sector partnerships to achieve at meaningful scale. Advancing formal regulation around WBE is perhaps the most complex undertaking of those proposed, yet also one of considerable importance to the long-term legitimacy and sustainability of the field. Critically, however, the order in which any of these priorities are pursued should not be determined by the research community alone. Meaningful community input, spanning both the breadth of professional disciplines that contribute to wastewater surveillance programs and the residents of the communities those programs serve, should guide how and when these recommendations are implemented.
Beyond the well-documented economic advantages of wastewater surveillance, the methodology provides several unique epidemiological benefits that are difficult or impossible to achieve through traditional clinical surveillance approaches. Wastewater monitoring captures population-level health data from all individuals contributing to a sewershed regardless of their access to healthcare, testing availability, or willingness to seek medical attention, providing an unbiased view of community disease burden that includes asymptomatic and mildly symptomatic individuals who would otherwise go undetected. The near-real-time nature of wastewater data enables early warning capabilities that can detect increases in pathogen prevalence days to weeks before corresponding rises in clinical cases, hospitalizations, or emergency department visits, allowing public health officials to implement interventions proactively rather than reactively. Furthermore, wastewater surveillance maintains individual privacy by aggregating data at the community level, avoiding the ethical and logistical challenges associated with individual-level health data collection while still providing actionable intelligence for public health decision-making. The ability to simultaneously monitor multiple pathogens, chemical exposures, and antimicrobial resistance genes from a single sample creates opportunities for comprehensive community health assessments that would be prohibitively expensive and logistically complex using traditional surveillance methods.
While wastewater surveillance has recently experienced widespread adoption of the technique, it is important to recognize that the field is still in its infancy. The direction that the field will take over the next decade will solidify WBE’s place in modern society, though that direction has yet to be determined. It is vital that the core values that started this field are adhered to and guide the future development of wastewater monitoring. Ultimately, wastewater surveillance was developed to serve the community, and this principle should guide all WBE initiatives.
DECLARATIONS
Authors’ contributions
The author contributed solely to the article.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool Claude (Anthropic, version Claude Sonnet 4.6, released 2025) was used solely for Python code development (Figures 2 and 3) and language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
None.
Conflicts of interest
Gushgari, A. was affiliated with Eurofins Environment Testing USA as Senior Director of Emerging Contaminants at the time of manuscript submission. The employer had no role in the study design, data analysis, interpretation, or the decision to submit the manuscript for publication.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Zuccato, E.; Chiabrando, C.; Castiglioni, S.; et al. Cocaine in surface waters: a new evidence-based tool to monitor community drug abuse. Environ. Health. 2005, 4, 14.
2. Kirby, A. E.; Walters, M. S.; Jennings, W. C.; et al. Using wastewater surveillance data to support the COVID-19 response - United States, 2020-2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 1242-4.
3. Fielding-Miller, R.; Karthikeyan, S.; Gaines, T.; et al. Wastewater and surface monitoring to detect COVID-19 in elementary school settings: the Safer at School Early Alert project. medRxiv 2021.
4. Crowe, J.; Schnaubelt, A. T.; Schmidtbonne, S.; et al. Assessment of a program for SARS-CoV-2 screening and environmental monitoring in an urban public school district. JAMA. Netw. Open. 2021, 4, e2126447.
5. Driver, E. M.; Bowes, D. A.; Halden, R. U.; Conroy-Ben, O. Implementing wastewater monitoring on American Indian reservations to assess community health indicators. Sci. Total. Environ. 2022, 823, 153882.
6. Wright, J.; Driver, E. M.; Bowes, D. A.; Johnston, B.; Halden, R. U. Comparison of high-frequency in-pipe SARS-CoV-2 wastewater-based surveillance to concurrent COVID-19 random clinical testing on a public U.S. university campus. Sci. Total. Environ. 2022, 820, 152877.
7. Harris-Lovett, S.; Nelson, K. L.; Beamer, P.; et al. Wastewater surveillance for SARS-CoV-2 on college campuses: initial efforts, lessons learned, and research needs. Int. J. Environ. Res. Public. Health. 2021, 18, 4455.
8. Gibas, C.; Lambirth, K.; Mittal, N.; et al. Implementing building-level SARS-CoV-2 wastewater surveillance on a university campus. Sci. Total. Environ. 2021, 782, 146749.
9. Greenwald, H. D.; Kennedy, L. C.; Hinkle, A.; et al. Tools for interpretation of wastewater SARS-CoV-2 temporal and spatial trends demonstrated with data collected in the San Francisco Bay Area. Water. Research. X. 2021, 12, 100111.
10. Colosi, L. M.; Barry, K. E.; Kotay, S. M.; et al. Development of wastewater pooled surveillance of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from congregate living settings. Appl. Environ. Microbiol. 2021, 87, e0043321.
11. Yaglom, H. D.; Maurer, M.; Collins, B.; et al. One health genomic surveillance and response to a university-based outbreak of the SARS-CoV-2 Delta AY.25 lineage, Arizona, 2021. PLoS. ONE. 2022, 17, e0272830.
12. Ginkgo Bioworks. Ginkgo Bioworks and XpresCheck receive award to expand CDC’s traveler-based SARS-CoV-2 genomic surveillance program in U.S. airports. 2022. https://www.prnewswire.com/news-releases/ginkgo-bioworks-and-xprescheck-receive-award-to-expand-cdcs-traveler-based-sars-cov-2-genomic-surveillance-program-in-us-airports-301605653.html. (accessed 2026-04-28).
13. Montgomery, A. B.; O’Rourke, C. E.; Subedi, B. Basketball and drugs: wastewater-based epidemiological estimation of discharged drugs during basketball games in Kentucky. Sci. Total. Environ. 2021, 752, 141712.
14. Gushgari, A. J.; Driver, E. M.; Steele, J. C.; Halden, R. U. Tracking narcotics consumption at a Southwestern U.S. university campus by wastewater-based epidemiology. J. Hazard. Mater. 2018, 359, 437-44.
15. Gushgari, A. J.; Venkatesan, A. K.; Chen, J.; Steele, J. C.; Halden, R. U. Long-term tracking of opioid consumption in two United States cities using wastewater-based epidemiology approach. Water. Res. 2019, 161, 171-80.
16. Fontenele, R. S.; Kraberger, S.; Hadfield, J.; et al. High-throughput sequencing of SARS-CoV-2 in wastewater provides insights into circulating variants. Water. Res. 2021, 205, 117710.
17. Hart, O. E.; Halden, R. U. Computational analysis of SARS-CoV-2/COVID-19 surveillance by wastewater-based epidemiology locally and globally: feasibility, economy, opportunities and challenges. Sci. Total. Environ. 2020, 730, 138875.
18. Devault, D. A.; Karolak, S.; Lévi, Y.; Rousis, N. I.; Zuccato, E.; Castiglioni, S. Exposure of an urban population to pesticides assessed by wastewater-based epidemiology in a Caribbean island. Sci. Total. Environ. 2018, 644, 129-36.
19. Rousis, N. I.; Gracia-Lor, E.; Zuccato, E.; et al. Wastewater-based epidemiology to assess pan-European pesticide exposure. Water. Res. 2017, 121, 270-9.
20. Subedi, B., Burgard, D. Wastewater-based epidemiology as a complementary approach to the conventional survey-based approach for the estimation of community consumption of drugs. In Wastewater-Based Epidemiology: Estimation of Community Consumption of Drugs and Diets; Subedi, B., Burgard, D. A., Loganathan, B. G., Eds.; ACS Symposium Series, Vol. 1319; American Chemical Society, 2019; pp 3-21.
21. Bowes, D. A.; Halden, R. U. Theoretical evaluation of using wastewater-based epidemiology to assess the nutritional status of human populations. Curr. Opin. Environ. Sci. Health. 2019, 9, 58-63.
22. Choi, P. M.; Tscharke, B. J.; Donner, E.; et al. Wastewater-based epidemiology biomarkers: past, present and future. TrAC. Trends. Anal. Chem. 2018, 105, 453-69.
23. Driver, E. M.; Gushgari, A. J.; Steele, J. C.; Bowes, D. A.; Halden, R. U. Assessing population-level stress through glucocorticoid hormone monitoring in wastewater. Sci. Total. Environ. 2022, 838, 155961.
24. Mtetwa, H. N.; Amoah, I. D.; Kumari, S.; Bux, F.; Reddy, P. Wastewater-based surveillance of antibiotic resistance genes associated with tuberculosis treatment regimen in KwaZulu Natal, South Africa. Antibiotics 2021, 10, 1362.
25. Riquelme, M. V.; Garner, E.; Gupta, S.; et al. Wastewater based epidemiology enabled surveillance of antibiotic resistance. medRxiv 2021.
26. Foxman, B.; Salzman, E.; Gesierich, C.; et al. Wastewater surveillance of antibiotic-resistant bacteria for public health action: potential and challenges. Am. J. Epidemiol. 2025, 194, 1192-9.
27. Conforti, S.; Pruden, A.; Acosta, N.; et al. Strengthening policy relevance of wastewater-based surveillance for antimicrobial resistance. Environ. Sci. Technol. 2025, 59, 2339-43.
28. Chaudhury, S.; Oransathit, W.; Peerapongpaisarn, D.; et al. Identification of clinically relevant multi-drug resistant ESKAPEE isolates from hospital wastewater surveillance in Thailand. Front. Microbiol. 2025, 16, 1657219.
29. Goetgeluck, C.; Olojo, O.; Hilton, S. P.; et al. Using wastewater surveillance to investigate community-level differences in antibacterial resistance in a major urban center, USA. Appl. Environ. Microbiol. 2025, 91, e0168425.
30. Sousa-Carmo, R. R.; Lincopan, N. Importance of wastewater-based and environmental surveillance for monitoring resistance to next-generation antibiotics. Future. Microbiol. 2025, 20, 849-54.
31. Alford, B.; Hughes, C.; Gilpin, D.; Mcgrath, J. Monitoring antimicrobial resistance in care homes through wastewater surveillance: a scoping review. J. Hosp. Infect. 2025, 164, 8-17.
32. Wang, H.; Liu, Z.; Zhang, J.; Huang, R.; Yin, H.; Dang, Z. Human exposure of bisphenol A and its analogues: understandings from human urinary excretion data and wastewater-based epidemiology. Environ. Sci. Pollut. Res. Int. 2019, 27, 3247-56.
33. Gracia-Lor, E.; Zuccato, E.; Hernández, F.; Castiglioni, S. Wastewater-based epidemiology for tracking human exposure to mycotoxins. J. Hazard. Mater. 2020, 382, 121108.
34. Lajoie, A. S.; Holm, R. H.; Anderson, L. B.; Ness, H. D.; Smith, T. Survey of nationwide public perceptions regarding acceptance of wastewater used for community health monitoring in the United States. medRxiv 2022.
35. Wolfe, M. K.; Duong, D.; Bakker, K. M.; et al. Wastewater-based detection of two influenza outbreaks. Environ. Sci. Technol. Lett. 2022, 9, 687-92.
36. Jones, M. J.; Ibrahim, R.; Clark, S.; et al. Wastewater surveillance of SARS-CoV-2 and influenza in a dynamic university community: understanding how wastewater measurements correspond to reported cases. Sci. Total. Environ. 2025, 1000, 180377.
37. Guo, Y.; Li, J.; O’Brien, J.; Sivakumar, M.; Jiang, G. Back-estimation of norovirus infections through wastewater-based epidemiology: a systematic review and parameter sensitivity. Water. Res. 2022, 219, 118610.
38. Wolfe, M. K.; Duong, D.; Hughes, B.; Chan-Herur, V.; White, B. J.; Boehm, A. B. Detection of monkeypox viral DNA in a routine wastewater monitoring program. medRxiv 2022.
39. Hughes, B.; Duong, D.; White, B. J.; et al. Respiratory syncytial virus (RSV) RNA in wastewater settled solids reflects RSV clinical positivity rates. Environ. Sci. Technol. Lett. 2022, 9, 173-8.
40. Chen, W.; Bibby, K. Temporal, spatial, and methodological considerations in evaluating the viability of measles wastewater surveillance. Sci. Total. Environ. 2025, 959, 178141.
41. Kuhn, K. G.; Shelton, K.; Sanchez, G.; et al. Wastewater surveillance as a tool for understanding West Nile virus transmission and distribution in Oklahoma. Sci. Total. Environ. 2025, 983, 179707.
42. Falman, J. C.; Fagnant-Sperati, C. S.; Kossik, A. L.; Boyle, D. S.; Meschke, J. S. Evaluation of secondary concentration methods for poliovirus detection in wastewater. Food. Environ. Virol. 2019, 11, 20-31.
43. Alshehri, B.; Birch, O. N.; Greaves, J. C. Monitoring multiple sexually transmitted pathogens through wastewater surveillance. Pathogens 2025, 14, 562.
44. Hou, C.; Hua, Z.; Xu, P.; et al. Estimating the prevalence of hepatitis B by wastewater-based epidemiology in 19 cities in China. Sci. Total. Environ. 2020, 740, 139696.
45. Ahmed, T.; Zulli, A.; Ishtiaq, F.; et al. Charting the future of wastewater-based epidemiology for vector-borne diseases: opportunities, challenges, and climate-driven needs. Environ. Sci. Water. Res. Technol. 2026, 12, 499-507.
46. D’Aoust, P. M. Why wastewater-based epidemiology must tackle noncommunicable diseases. Curr. Med. Res. Opin. 2025, 41, 977-81.
47. Kreuter, M. W.; Garg, R.; Marsh, A. K.; et al. Wastewater monitoring: improving public awareness and understanding in the United States, May 2024. Public. Health. Rep. 2025, 141, 114-21.
48. Farkas, K.; Kaya, D.; Maal-Bared, R.; et al. Communicating wastewater-based surveillance data to drive action. J. Water. Health. 2025, 23, 1095-108.
49. Diamond, M. B.; Keshaviah, A.; Bento, A. I.; et al. Wastewater surveillance of pathogens can inform public health responses. Nat. Med. 2022, 28, 1992-5.
50. Devault, D. A.; Peyré, A.; Cottereau, V.; Pleignet, E.; Daveluy, A. How synthetic cannabinoid user profiles and consumption patterns can affect wastewater-based epidemiology. Environ. Sci. Pollut. Res. 2025, 32, 31108-20.
51. Abafe, O. A.; Späth, J.; Fick, J.; et al. LC-MS/MS determination of antiretroviral drugs in influents and effluents from wastewater treatment plants in KwaZulu-Natal, South Africa. Chemosphere 2018, 200, 660-70.
52. Mashiane M, S. C. Quantification of selected antiretroviral drugs in a wastewater treatment works in South Africa using GC-TOFMS. J. Chromatogr. Sep. Tech. 2015, 6, 4.
53. Tsotetsi, N. T.; Rasifudi, N. F.; Seedat, N.; Maxakato, N. W.; Mekuto, L. Monitoring the performance of two wastewater treatment plants in the treatment of selected antiretrovirals in the Gauteng Province, South Africa. Clean. Water. 2025, 4, 100146.
54. Mccall, C.; Wu, H.; Miyani, B.; Xagoraraki, I. Identification of multiple potential viral diseases in a large urban center using wastewater surveillance. Water. Res. 2020, 184, 116160.
55. Casares-Jimenez, M.; Garcia-Garcia, T.; Suárez-Cárdenas, J. M.; et al. Correlation of hepatitis E and rat hepatitis E viruses urban wastewater monitoring and clinical cases. Sci. Total. Environ. 2024, 908, 168203.
56. Wolfe, M. K.; Varkila, M. R. J.; Zulli, A.; Parsonnet, J.; Boehm, A. B. Detection and quantification of human immunodeficiency virus-1 (HIV-1) total nucleic acids in wastewater settled solids from two California communities. Appl. Environ. Microbiol. 2024, 90, e0147724.
57. Chen, Y.; Chen, L.; Deng, Q.; et al. The presence of SARS‐CoV‐2 RNA in the feces of COVID‐19 patients. J. Med. Virol. 2020, 92, 833-40.
58. Jones, D. L.; Bridgman, M.; Pellett, C.; et al. Use of wastewater from passenger ships to assess the movement of COVID-19 and other pathogenic viruses across maritime international boundaries. Front. Public. Health. 2024, 12, 1377996.
59. Erickson, T. B.; Endo, N.; Duvallet, C.; et al. “Waste Not, Want Not” - leveraging sewer systems and wastewater-based epidemiology for drug use trends and pharmaceutical monitoring. J. Med. Toxicol. 2021, 17, 397-410.
60. Osena, G.; Fick, J.; Flach, C.; Kristiansson, E.; Joakim Larsson, D. Evaluating wastewater surveillance for estimating pharmaceutical use. Environ. Int. 2025, 204, 109807.
61. Ammerman, M. L.; Mullapudi, S.; Gilbert, J.; et al. Norovirus GII wastewater monitoring for epidemiological surveillance. PLOS. Water. 2024, 3, e0000198.
62. Walker, D. I.; Witt, J.; Rostant, W.; et al. Piloting wastewater-based surveillance of norovirus in England. Water. Res. 2024, 263, 122152.
63. Bijlsma, L.; Campos-Mañas, M.; Hernández, F.; et al. Wastewater surveillance for assessing human exposure to pesticides: investigating populations living near flower bulb fields. J. Environ. Chem. Eng. 2025, 13, 117090.
64. Troja, F.; Indio, V.; Savini, F.; et al. Monitoring and preventing foodborne outbreaks: are we missing wastewater as a key data source? Ital. J. Food. Saf. 2024, 13, 12725.
65. Kuhn, K. G.; Shukla, R.; Mannell, M.; et al. Using wastewater surveillance to monitor gastrointestinal pathogen infections in the state of Oklahoma. Microorganisms 2023, 11, 2193.
66. Wang, Z.; Cai, M.; Du, P.; Li, X. Wastewater surveillance for antibiotics and resistance genes in a river catchment: spatiotemporal variations and the main drivers. Water. Res. 2024, 251, 121090.
67. Knight, M. E.; Farkas, K.; Wade, M.; et al. Wastewater-based analysis of antimicrobial resistance at UK airports: evaluating the potential opportunities and challenges. Environ. Int. 2025, 195, 109260.
68. Capone, D.; Chigwechokha, P.; De Los Reyes, F. L.; et al. Impact of sampling depth on pathogen detection in pit latrines. PLoS. Negl. Trop. Dis. 2021, 15, e0009176.
69. Camacho, C. G.; Antonison, A.; Oldnettle, A.; et al. Statewide surveillance and mapping of PFAS in Florida surface water. ACS. EST. Water. 2024, 4, 4343-55.
70. Oza, S.; Bell, K. Y.; Xu, Z.; et al. Surveillance of PFAS in sludge and biosolids at 12 water resource recovery facilities. J. Environ. Qual. 2024, 54, 6-19.
71. Anchidin-Norocel, L.; Bosancu, A.; Iatcu, O. C.; Lobiuc, A.; Covasa, M. Real-time detection of heavy metals and some other pollutants in wastewater using chemical sensors: a strategy to limit the spread of antibiotic-resistant bacteria. Chemosensors 2025, 13, 352.
72. Wang, Y.; Amarasiri, M.; Oishi, W.; et al. Aptamer-based biosensors for wastewater surveillance of influenza virus, SARS-CoV-2, and norovirus: a comprehensive review. Water. Res. 2025, 279, 123484.
73. Delcher, C.; Quesinberry, D.; Torabi, S.; et al. Wastewater surveillance for xylazine in Kentucky. AJPM. Focus. 2024, 3, 100203.
74. Joseph, K. M.; Parikh, D.; Xuan, Q.; et al. First detection of xylazine in Texas wastewater and its association with fentanyl use. medRxiv 2024.
75. Grassly, N. C.; Shaw, A. G.; Owusu, M. Global wastewater surveillance for pathogens with pandemic potential: opportunities and challenges. Lancet. Microbe. 2025, 6, 100939.
76. Xiao, K.; Zhang, L. Wastewater pathogen surveillance based on One Health approach. Lancet. Microbe. 2023, 4, e297.
77. Ahmed, W.; Liu, Y.; Smith, W.; et al. Leveraging wastewater surveillance to detect viral diseases in livestock settings. Sci. Total. Environ. 2024, 931, 172593.
78. Kelbrick, M.; Hesse, E.; O’Brien, S. Cultivating antimicrobial resistance: how intensive agriculture ploughs the way for antibiotic resistance. Microbiology 2023, 169, 001384.
79. Tang, K. W. K.; Millar, B. C.; Moore, J. E. Antimicrobial resistance (AMR). Br. J. Biomed. Sci. 2023, 80, 11387.
80. Kaviani Rad, A.; Astaykina, A.; Streletskii, R.; et al. An overview of antibiotic resistance and abiotic stresses affecting antimicrobial resistance in agricultural soils. Int. J. Environ. Res. Public. Health. 2022, 19, 4666.
81. Silvester, R.; Perry, W. B.; Webster, G.; et al. Metagenomic profiling of hospital wastewater: a comprehensive national scale analysis of antimicrobial resistance genes and opportunistic pathogens. J. Infect. 2025, 90, 106503.
82. Wu, F.; Xiao, A.; Zhang, J.; et al. Wastewater surveillance of SARS-CoV-2 across 40 U.S. states from February to June 2020. Water. Res. 2021, 202, 117400.
83. Ramírez-Chavarría, R. G.; Castillo-Villanueva, E.; Alvarez-Serna, B. E.; et al. Loop-mediated isothermal amplification-based electrochemical sensor for detecting SARS-CoV-2 in wastewater samples. J. Environ. Chem. Eng. 2022, 10, 107488.
84. Jiménez-Rodríguez, M. G.; Silva-Lance, F.; Parra-Arroyo, L.; et al. Biosensors for the detection of disease outbreaks through wastewater-based epidemiology. TrAC. Trends. Anal. Chem. 2022, 155, 116585.
85. Mahmoudi, T.; Naghdi, T.; Morales-Narváez, E.; Golmohammadi, H. Toward smart diagnosis of pandemic infectious diseases using wastewater-based epidemiology. TrAC. Trends. Anal. Chem. 2022, 153, 116635.
86. Xu, Q.; Guo, Y.; Niu, G.; Wu, H.; Coulon, F.; Yang, Z. Advances in biosensor technology for illicit drug detection enable effective wastewater surveillance. Chem. Bio. Eng. 2025, 3, 87-103.
87. Peccia, J.; Zulli, A.; Brackney, D. E.; et al. Measurement of SARS-CoV-2 RNA in wastewater tracks community infection dynamics. Nat. Biotechnol. 2020, 38, 1164-7.
88. Nemudryi, A.; Nemudraia, A.; Wiegand, T.; et al. Temporal detection and phylogenetic assessment of SARS-CoV-2 in municipal wastewater. Cell. Reports. Medicine. 2020, 1, 100098.
89. Bivins, A.; Kaya, D.; Bibby, K.; et al. Variability in RT-qPCR assay parameters indicates unreliable SARS-CoV-2 RNA quantification for wastewater surveillance. Water. Res. 2021, 203, 117516.
90. Bio-Rad. Droplet Digital PCR (ddPCR) Technology. https://www.bio-rad.com/en-us/life-science/learning-center/introduction-to-digital-pcr/what-is-droplet-digital-pcr. (accessed 2026-04-28).
91. Ciesielski, M.; Blackwood, D.; Clerkin, T.; et al. Assessing sensitivity and reproducibility of RT-ddPCR and RT-qPCR for the quantification of SARS-CoV-2 in wastewater. J. Virol. Methods. 2021, 297, 114230.
92. Gonzalez, R.; Larson, A.; Thompson, H.; Carter, E.; Cassi, X. F. Redesigning SARS-CoV-2 clinical RT-qPCR assays for wastewater RT-ddPCR. Epidemiology 2021.
93. Flood, M. T.; D’Souza, N.; Rose, J. B.; Aw, T. G. Methods evaluation for rapid concentration and quantification of SARS-CoV-2 in raw wastewater using droplet digital and quantitative RT-PCR. Food. Environ. Virol. 2021, 13, 303-15.
94. CDC. National Wastewater Surveillance System (NWSS): a new public health tool to understand COVID-19 spread in a community. 2022. https://stacks.cdc.gov/view/cdc/114168. (accessed 2026-04-28).
95. Doorn, N. Wastewater research and surveillance: an ethical exploration. Environ. Sci. Water. Res. Technol. 2022, 8, 2431-8.
96. Hernández, F.; Castiglioni, S.; Covaci, A.; et al. Mass spectrometric strategies for the investigation of biomarkers of illicit drug use in wastewater. Mass. Spectrom. Rev. 2016, 37, 258-80.
97. Keller, E. L.; Peake, B.; Simpson, B. S.; White, J. M.; Gerber, C. Comprehensive method to detect nitazene analogues and xylazine in wastewater. Environ. Sci. Pollut. Res. 2025, 32, 30863-70.
98. Baker, D. R.; Kasprzyk-Hordern, B. Critical evaluation of methodology commonly used in sample collection, storage and preparation for the analysis of pharmaceuticals and illicit drugs in surface water and wastewater by solid phase extraction and liquid chromatography–mass spectrometry. J. Chromatogr. A. 2011, 1218, 8036-59.
99. Zuccato, E.; Chiabrando, C.; Castiglioni, S.; Bagnati, R.; Fanelli, R. Estimating community drug abuse by wastewater analysis. Environ. Health. Perspect. 2008, 116, 1027-32.
100. Vogel, E. J.; Neyra, M.; Larsen, D. A.; Zeng, T. Target and nontarget screening to support capacity scaling for substance use assessment through a statewide wastewater surveillance network in New York. Environ. Sci. Technol. 2024, 58, 8518-30.
101. Opioid Wastewater Collection Data Dashboard. https://www.arcgis.com/apps/dashboards/69d996bc23dc461f82d01f47a5d70bfe. (accessed 2026-04-28).
102. Healthy Central Valley Together. Healthy Central Valley Together wastewater data. https://healthycvtogether.org/data/. (accessed 2026-04-28).
103. COVIDPoops19 Dashboard. https://www.arcgis.com/apps/dashboards/c778145ea5bb4daeb58d31afee389082. (accessed 2026-04-28).
104. CDC. Surveillance and data analytics. https://covid.cdc.gov/covid-data-tracker/#wastewater-surveillance. (accessed 2026-04-28).
105. Maal-Bared, R.; Qiu, Y.; Li, Q.; et al. Does normalization of SARS-CoV-2 concentrations by Pepper Mild Mottle Virus improve correlations and lead time between wastewater surveillance and clinical data in Alberta (Canada): comparing twelve SARS-CoV-2 normalization approaches. Sci. Total. Environ. 2023, 856, 158964.
106. Nguyen Quoc, B.; Saingam, P.; Redcorn, R.; et al. Case Study: Impact of diurnal variations and stormwater dilution on SARS-CoV-2 RNA signal intensity at neighborhood scale wastewater pumping stations. ACS. EST. Water. 2022, 2, 1964-75.
107. Price, M.; Tscharke, B.; Chappell, A.; et al. Testing methods to estimate population size for wastewater treatment plants using census data: Implications for wastewater-based epidemiology. Sci. Total. Environ. 2024, 922, 170974.
108. Oloye, F. F.; Xie, Y.; Challis, J. K.; et al. Understanding common population markers for SARS-CoV-2 RNA normalization in wastewater - a review. Chemosphere 2023, 333, 138682.
109. Hsu, S.; Bayati, M.; Li, C.; et al. Biomarkers selection for population normalization in SARS-CoV-2 wastewater-based epidemiology. Water. Res. 2022, 223, 118985.
110. Maere, T.; Therrien, J. D.; VanRolleghem, P. Normalization practices for SARS-CoV-2 data in wastewater-based epidemiology. Université Laval, 2022. https://nccid.ca/wp-content/uploads/sites/2/2023/02/Normalization-practices-Technical-Report-Final3.pdf. (accessed 2026-04-28).
111. Thomas, K. V.; Amador, A.; Baz-Lomba, J. A.; Reid, M. Use of mobile device data to better estimate dynamic population size for wastewater-based epidemiology. Environ. Sci. Technol. 2017, 51, 11363-70.
112. Rhodes, E. R.; Vogel, J. R.; Lowery, B. C.; et al. Tracking SARS-CoV-2 levels in wastewater during college football events using cell phone data for population dynamics. Environments 2024, 11, 279.
113. Gomes, C. S.; De Jesus Soares Freire, D.; De Souza Ramos Pontes Moura, H.; et al. Wastewater surveillance to assess cocaine and methylenedioxymethamphetamine use trends during a major music festival in Brazil. Drug. Test. Anal. 2024, 17, 88-100.
114. Humphries, M. A.; Bruno, R.; Lai, F. Y.; et al. Evaluation of monitoring schemes for wastewater-based epidemiology to identify drug use trends using cocaine, methamphetamine, MDMA and methadone. Environ. Sci. Technol. 2016, 50, 4760-8.
115. Mcculloch, S.; Bergen-Cico, D.; Zeng, T.; Larsen, D. A. Comparison of self-reported survey and wastewater-based epidemiology measures of cocaine use on a college campus. PLoS. ONE. 2026, 21, e0338791.
116. Gerrity, D.; Barber, C. A.; Trenholm, R. A.; et al. Piloting wastewater-based monitoring on a university campus to inform public health surveillance and response for opioids and other high-risk substances. J. Water. Health. 2026, 24, 65-80.
117. Du, P.; Thai, P. K.; Bai, Y.; et al. Monitoring consumption of methadone and heroin in major Chinese cities by wastewater-based epidemiology. Drug. Alcohol. Depend. 2019, 205, 107532.
118. Jaunay, E. L.; Simpson, B. S.; White, J. M.; Gerber, C. Using wastewater-based epidemiology to evaluate the relative scale of use of opioids. Sci. Total. Environ. 2023, 897, 165148.
119. Bade, R.; Nadarajan, D.; Driver, E. M.; et al. Wastewater-based monitoring of the nitazene analogues: first detection of protonitazene in wastewater. Sci. Total. Environ. 2024, 920, 170781.
120. Bade, R.; Nadarajan, D.; Hall, W.; Brown, J. A.; Schumann, J.; NPS Wastewater Consortium. Early identification of the use of potent benzylbenzimidazoles (nitazenes) through wastewater analysis: two years of data from 22 countries. Addiction 2025, 120, 1739-46.
121. Keller, E.; Tscharke, B.; Peake, B.; et al. Weekend highs: rising ketamine levels in Australian wastewater. Environ. Adv. 2026, 23, 100687.
122. Quireyns, M.; Boogaerts, T.; Van Wichelen, N.; et al. Monitoring community use of benzodiazepines through wastewater‐based epidemiology. Drug. Test. Anal. 2025, 17, 2066-77.
123. Pecson, B. M.; Darby, E.; Haas, C. N.; et al. Reproducibility and sensitivity of 36 methods to quantify the SARS-CoV-2 genetic signal in raw wastewater: findings from an interlaboratory methods evaluation in the U.S. Environ. Sci. Water. Res. Technol. 2021, 7, 504-20.
124. Prasek, S. M.; Pepper, I. L.; Innes, G. K.; et al. Population level SARS-CoV-2 fecal shedding rates determined via wastewater-based epidemiology. Sci. Total. Environ. 2022, 838, 156535.
125. Kashi, A. H.; De la Rosette, J.; Amini, E.; Abdi, H.; Fallah-Karkan, M.; Vaezjalali, M. Urinary viral shedding of COVID-19 and its clinical associations: a systematic review and meta-analysis of observational studies. Urol. J. 2020, 17, 433-41.
126. Parkins, M. D.; Lee, B. E.; Acosta, N.; et al. Wastewater-based surveillance as a tool for public health action: SARS-CoV-2 and beyond. Clin. Microbiol. Rev. 2024, 37, e0010322.
127. Soller, J.; Jennings, W.; Schoen, M.; et al. Modeling infection from SARS-CoV-2 wastewater concentrations: promise, limitations, and future directions. J. Water. Health. 2022, 20, 1197-211.
128. Wang, W.; Xu, Y.; Gao, R.; et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020, 323, 1843-4.
129. Arts, P. J.; Kelly, J. D.; Midgley, C. M.; et al. Longitudinal and quantitative fecal shedding dynamics of SARS-CoV-2, pepper mild mottle virus, and crAssphage. mSphere 2023, 8, e0013223.
130. Eap, C. B.; Buclin, T.; Baumann, P. Interindividual variability of the clinical pharmacokinetics of methadone: implications for the treatment of opioid dependence. Clin. Pharmacokinet. 2002, 41, 1153-93.
131. Aylward, L. L.; Hays, S. M.; Zidek, A. Variation in urinary spot sample, 24 h samples, and longer-term average urinary concentrations of short-lived environmental chemicals: implications for exposure assessment and reverse dosimetry. J. Expo. Sci. Environ. Epidemiol. 2016, 27, 582-90.
132. Sims, N.; Kasprzyk-Hordern, B. Future perspectives of wastewater-based epidemiology: monitoring infectious disease spread and resistance to the community level. Environ. Int. 2020, 139, 105689.
133. Hart, O. E.; Halden, R. U. Modeling wastewater temperature and attenuation of sewage-borne biomarkers globally. Water. Res. 2020, 172, 115473.
134. Li, J.; Choi, P. M.; Gao, J.; et al. In-sewer stability of 31 human health biomarkers and suitability for wastewater-based epidemiology. Water. Res. 2024, 249, 120978.
135. Laimou-Geraniou, M.; Quireyns, M.; Boogaerts, T.; et al. Stability of antipsychotic biomarkers for wastewater-based epidemiology. J. Pharm. Biomed. Anal. 2025, 257, 116702.
136. Mccall, A.; Bade, R.; Kinyua, J.; et al. Critical review on the stability of illicit drugs in sewers and wastewater samples. Water. Res. 2016, 88, 933-47.
137. Wen, J.; Duan, L.; Wang, B.; et al. In-sewer stability assessment of 140 pharmaceuticals, personal care products, pesticides and their metabolites: Implications for wastewater-based epidemiology biomarker screening. Environ. Int. 2024, 184, 108465.
138. Adams, C.; Bias, M.; Welsh, R. M.; et al. The National Wastewater Surveillance System (NWSS): from inception to widespread coverage, 2020-2022, United States. Sci. Total. Environ. 2024, 924, 171566.
139. O’Keeffe, J. Wastewater-based epidemiology: current uses and future opportunities as a public health surveillance tool. Environ. Health. Rev. 2021, 64, 44-52.
140. Mccall, C.; Fang, Z. N.; Li, D.; et al. Modeling SARS-CoV-2 RNA degradation in small and large sewersheds. Environ. Sci. Water. Res. Technol. 2022, 8, 290-300.
141. Betancourt, W. Q.; Schmitz, B. W.; Innes, G. K.; et al. COVID-19 containment on a college campus via wastewater-based epidemiology, targeted clinical testing and an intervention. Sci. Total. Environ. 2021, 779, 146408.
142. Kotay, S. M.; Tanabe, K. O.; Colosi, L. M.; et al. Building-level wastewater surveillance for SARS-CoV-2 in occupied university dormitories as an outbreak forecasting tool: one year case study. ACS. EST. Water. 2022, 2, 2094-104.
143. Corchis-Scott, R.; Geng, Q.; Seth, R.; et al. Averting an outbreak of SARS-CoV-2 in a university residence hall through wastewater surveillance. Microbiol. Spectr. 2021, 9, e0079221.
144. Bivins, A.; Kaya, D.; Ahmed, W.; et al. Passive sampling to scale wastewater surveillance of infectious disease: lessons learned from COVID-19. Sci. Total. Environ. 2022, 835, 155347.
145. Ali, M.; Younis, A. B.; Duru, C. I.; Sherchan, S. P. A review of AI/ML approaches in wastewater surveillance advancement. Sci. Total. Environ. 2026, 1015, 181364.
146. Aßmann, E.; Greiner, T.; Richard, H.; et al. Augmentation of wastewater-based epidemiology with machine learning to support global health surveillance. Nat. Water. 2025, 3, 753-63.
147. Keshaviah, A.; Huff, I.; Hu, X. C.; et al. Separating signal from noise in wastewater data: an algorithm to identify community-level COVID-19 surges in real time. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2216021120.
148. Pagsuyoin, S.; Ng, C.; Molejon, N.; Luo, Y. Coupling wastewater‐based epidemiology with data‐driven machine learning for managing public health risks. Risk. Anal. 2025, 45, 2974-82.
149. Zhu, W.; Wang, D.; Li, P.; Deng, H.; Deng, Z. Advances in wastewater-based epidemiology for pandemic surveillance: methodological frameworks and future perspectives. Microorganisms 2025, 13, 1169.
150. Xu, B.; Wu, G.; Li, Z.; et al. Towards domain-adapted large language models for water and wastewater management: methods, datasets and benchmarking. npj. Clean. Water. 2025, 8, 82.
151. Sun, M.; Han, R.; Jiang, B.; et al. A survey on large language model-based agents for statistics and data science. Am. Stat. 2025, 1-14.
152. Zehnder, C.; Béen, F.; Vojinovic, Z.; et al. Machine learning for detecting virus infection hotspots via wastewater‐based epidemiology: the case of SARS‐CoV‐2 RNA. GeoHealth 2023, 7, e2023GH000866.
153. Zheng, X.; Li, Y.; Deng, Y.; et al. Cost-effective sampling strategies for wastewater surveillance: a large-scale longitudinal study in Hong Kong and Shenzhen. Environ. Sci. Technol. 2026, 60, 8315-25.
154. Biomarker: Opioids. https://data.tempe.gov/pages/biomarker-opioids~b77394d180a840e79949b7298f2d7c85. (accessed 2026-04-28).
155. Healthy Central Valley Together Wastewater Data. https://healthycvtogether.org/data/. (accessed 2026-04-28).
156. Federal Trade Commission. FTC explores rules cracking down on commercial surveillance and lax data security practices. 2022. https://www.ftc.gov/news-events/news/press-releases/2022/08/ftc-explores-rules-cracking-down-commercial-surveillance-lax-data-security-practices. (accessed 2026-04-28).
157. National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice; Division on Earth and Life Studies; Water Science and Technology Board; Committee on Community Wastewater-based Infectious Disease Surveillance. Increasing the utility of wastewater-based disease surveillance for public health action: a phase 2 report. Washington (DC): National Academies Press (US); 2024.
158. Ni, K.; Korfmacher, K. S. A scoping review on the ethics of wastewater surveillance for COVID-19. J. Public. Health. Manag. Pract. 2025, 31, E387-95.
159. National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Division on Earth and Life Studies; Board on Population Health and Public Health Practice; Water Science and Technology Board; Committee on Community Wastewater-based Infectious Disease Surveillance. Wastewater-based disease surveillance for public health action. Washington (DC): National Academies Press (US); 2023.
160. Clarke, K.; Anderson, L. B.; Gadson, A.; Holm, R. H.; Kolers, A.; Smith, T. A case study of ethical bridges: wastewater-based epidemiology in the Rubbertown Air Toxics and Health Assessment (RATHA) project. Environ. Sci. Water. Res. Technol. 2025, 11, 1363-8.
161. Arefin, M. R.; Prouse, C.; Wittmer, J.; et al. Making waves: a justice-centred framework for wastewater-based public health surveillance. Water. Res. 2025, 268, 122747.
162. Barredo Arrieta, A.; Díaz-Rodríguez, N.; Del Ser, J.; et al. Explainable artificial intelligence (XAI): concepts, taxonomies, opportunities and challenges toward responsible AI. Inf. Fusion. 2020, 58, 82-115.
163. Yu, M.; Fang, M.; Tian, Z.; Wang, B.; Walker, D. Artificial intelligence and machine learning for environmental health study. Environ. Health. 2025, 3, 1115-6.
164. North Dakota House flushes proposed wastewater testing ban. https://bismarcktribune.com/news/local/govt-and-politics/north-dakota-house-flushes-proposed-wastewater-testing-ban/article_df7ec53a-493e-5bc8-918d-9c60abc10117.html. (accessed 2026-04-28).
165. Laturner, Z. W.; Zong, D. M.; Kalvapalle, P.; et al. Evaluating recovery, cost, and throughput of different concentration methods for SARS-CoV-2 wastewater-based epidemiology. Water. Res. 2021, 197, 117043.
166. Driver, E. M.; Gushgari, A.; Chen, J.; Halden, R. U. Alcohol, nicotine, and caffeine consumption on a public U.S. university campus determined by wastewater-based epidemiology. Sci. Total. Environ. 2020, 727, 138492.
167. Medina, C. Y.; Kadonsky, K. F.; Roman, F. A.; et al. The need of an environmental justice approach for wastewater based epidemiology for rural and disadvantaged communities: a review in California. Curr. Opin. Environm. Sci. Health. 2022, 27, 100348.
168. Shrestha, S.; Yoshinaga, E.; Chapagain, S. K.; Mohan, G.; Gasparatos, A.; Fukushi, K. Wastewater-based epidemiology for cost-effective mass surveillance of COVID-19 in low- and middle-income countries: challenges and opportunities. Water 2021, 13, 2897.
169. USDA. County-level data sets: Unemployment and median household income. https://www.ers.usda.gov/data-products/county-level-data-sets. (accessed 2026-04-28).
170. United States Census Bureau. American community survey 5-year estimates, 2018-2022. Table B19013: median household income in the past 12 months (in 2022 inflation-adjusted dollars). https://data.census.gov/table/ACSDT5Y2022.B19013?g=010XX00US. (accessed 2026-04-28).
171. United States Census Bureau. 2020 Island Areas Censuses: Guam. https://www.census.gov/data/tables/2020/dec/2020-guam.html. (accessed 2026-04-28).
172. ASU News. Tempe, ASU to work together on wastewater monitoring to improve public health. https://news.asu.edu/20180524-solutions-asu-tempe-partner-wastewater-monitoring-system-opioids. (accessed 2026-04-28).
173. Naughton, C. C.; Roman, F. A.; Alvarado, A. G. F.; et al. Show us the data: global COVID-19 wastewater monitoring efforts, equity, and gaps. FEMS. Microbes. 2023, 4, xtad003.
174. Keshaviah, A.; Diamond, M. B.; Wade, M. J.; et al. Wastewater monitoring can anchor global disease surveillance systems. Lancet. Glob. Health. 2023, 11, e976-81.
175. O’Brien, J. W.; Grant, S.; Banks, A. P.; et al. A National Wastewater Monitoring Program for a better understanding of public health: a case study using the Australian Census. Environ. Int. 2019, 122, 400-11.
176. Holm, R. H.; Anderson, L. B.; Ness, H. D.; Lajoie, A. S.; Smith, T. Towards the outbreak tail, what is the public opinion about wastewater surveillance in the United States? J. Water. Health. 2024, 22, 1409-18.
177. Gitter, A.; Ruvalcaba, V.; Clark, K.; et al. Turning analysis into action: opportunities and challenges in implementing wastewater science for public health decision-making. Front. Public. Health. 2025, 13, 1562659.
178. Truyens, C. S.; Berendes, D. M.; Cantrell, M. E.; et al. Advancing wastewater and environmental surveillance in LMICs for public health response and SDG data gaps. npj. Clean. Water. 2025, 8, 95.
179. WHO Africa. How wastewater surveillance is helping the COVID-19 fight. 2022. https://www.afro.who.int/news/how-wastewater-surveillance-helping-covid-19-fight. (accessed 2026-04-28).
180. Khan, M. S.; Wurzbacher, C.; Uchaikina, A.; Pleshkov, B.; Mirshina, O.; Drewes, J. E. A perspective on wastewater and environmental surveillance as a public health tool for low- and middle-income countries. Microorganisms 2025, 13, 238.
181. Adhikari, S.; Halden, R. U. Opportunities and limits of wastewater-based epidemiology for tracking global health and attainment of UN sustainable development goals. Environ. Int. 2022, 163, 107217.
182. Ngwira, L. G.; Sharma, B.; Shrestha, K. B.; et al. Cost of wastewater-based environmental surveillance for SARS-CoV-2: evidence from pilot sites in Blantyre, Malawi and Kathmandu, Nepal. PLOS. Glob. Public. Health. 2022, 2, e0001377.
183. Sanjak, J. S.; Mcauley, E. M.; Raybern, J.; et al. Wastewater surveillance pilot at US military installations: cost model analysis. JMIR. Public. Health. Surveill. 2024, 10, e54750.
184. Boyina, K.; Holm, R. H.; Samarasinghe, H.; et al. A scoping review of the private sector involvement in wastewater and environmental surveillance. Discov. Water. 2025, 5, 84.
185. Capone, D.; Chiluvane, M.; Cumbane, V.; et al. Environmental pathogen surveillance in cities without universal conventional wastewater infrastructure. MedRxiv 2025.
186. CDC. About traveler-based genomic surveillance. 2025. https://www.cdc.gov/traveler-genomic-surveillance/about/index.html. (accessed 2026-04-28).
187. Edson, R.; Rutayisire, R.; El-Khatib, Z.; et al. SARS-CoV-2 detection in international travelers through wastewater-based epidemiology at the Kigali international airport: genomic surveillance study. JMIR. Public. Health. Surveill. 2025, 11, e71104.
188. Nkambule, S.; Johnson, R.; Mathee, A.; et al. Wastewater-based SARS-CoV-2 airport surveillance: key trends at the Cape Town International Airport. J. Water. Health. 2023, 21, 402-8.
189. Zachmann, K.; Mattersdorf, J.; Lackner, S.; Agrawal, S. From crisis to clarity: tackling the sampling strategy challenge in airport wastewater surveillance during COVID-19. Water. Res. 2026, 292, 125229.
190. St-Onge, G.; Davis, J. T.; Hébert-Dufresne, L.; et al. Pandemic monitoring with global aircraft-based wastewater surveillance networks. Nat. Med. 2025, 31, 788-96.
191. Ahmed, W.; Bertsch, P. M.; Angel, N.; et al. Detection of SARS-CoV-2 RNA in commercial passenger aircraft and cruise ship wastewater: a surveillance tool for assessing the presence of COVID-19 infected travellers. J. Travel. Med. 2020, 27, taaa116.
192. Koureas, M.; Kyritsi, M.; Vontas, A.; et al. Pilot implementation of SARS-CoV-2 wastewater surveillance on cruise ships arriving at the port of piraeus from June to November 2021. Med. Sci. Forum. 2022, 13, 6.
193. Lancaster, E.; Byrd, K.; Ai, Y.; Lee, J. Socioeconomic status correlations with confirmed COVID-19 cases and SARS-CoV-2 wastewater concentrations in small-medium sized communities. Environ. Res. 2022, 215, 114290.
194. Kadonsky, K.; Naughton, C.; Tucker, L.; et al. Healthy Central Valley Together: an environmental justice approach for wastewater disease surveillance and health equity in California. In Proceedings of the Water Environment Federation. Water Environment Federation; 2022.
195. UNCTAD. Now 8 billion and counting: where the world’s population has grown most and why that matters. 2022. https://unctad.org/news/now-8-billion-and-counting-where-worlds-population-has-grown-most-and-why-matters. (accessed 2026-04-28).
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data





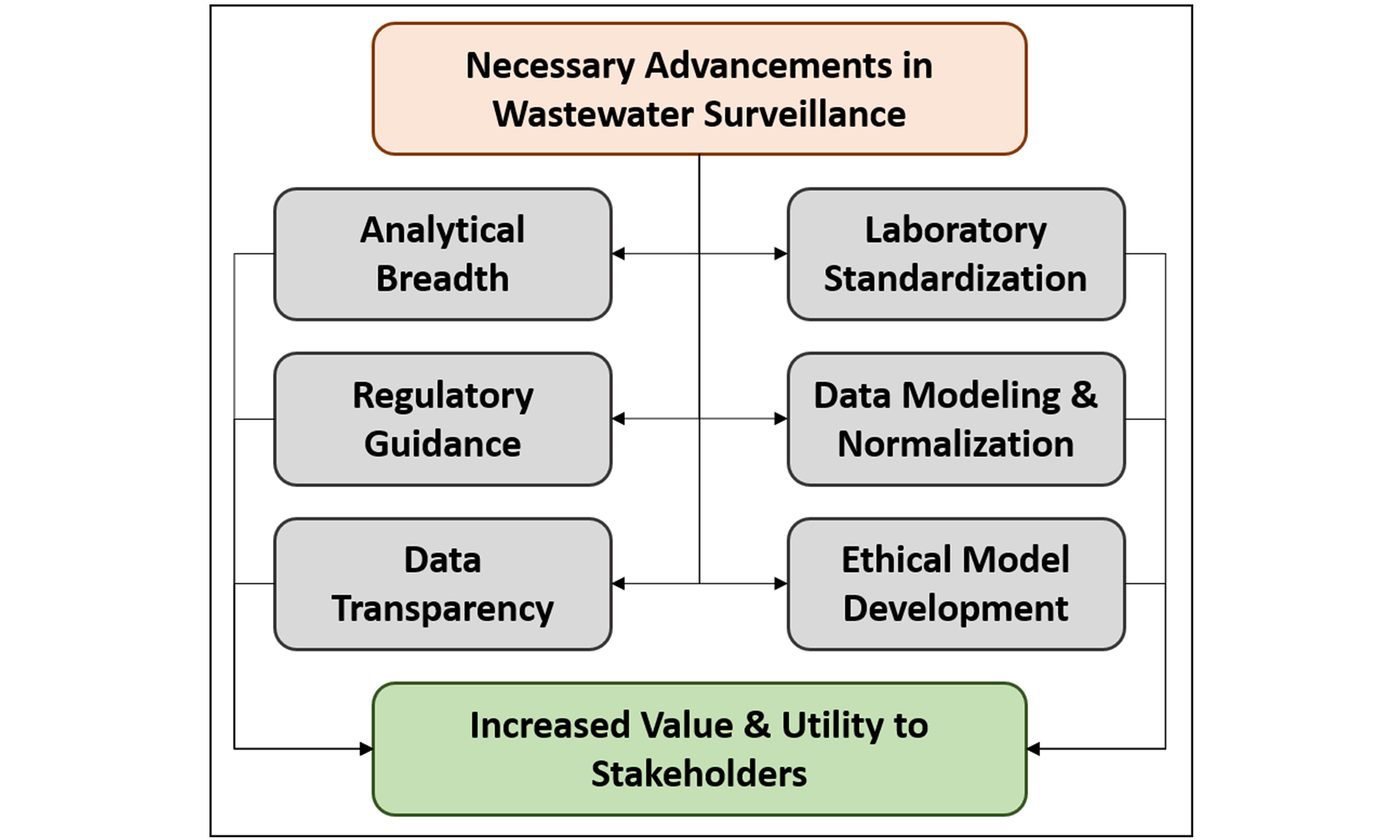
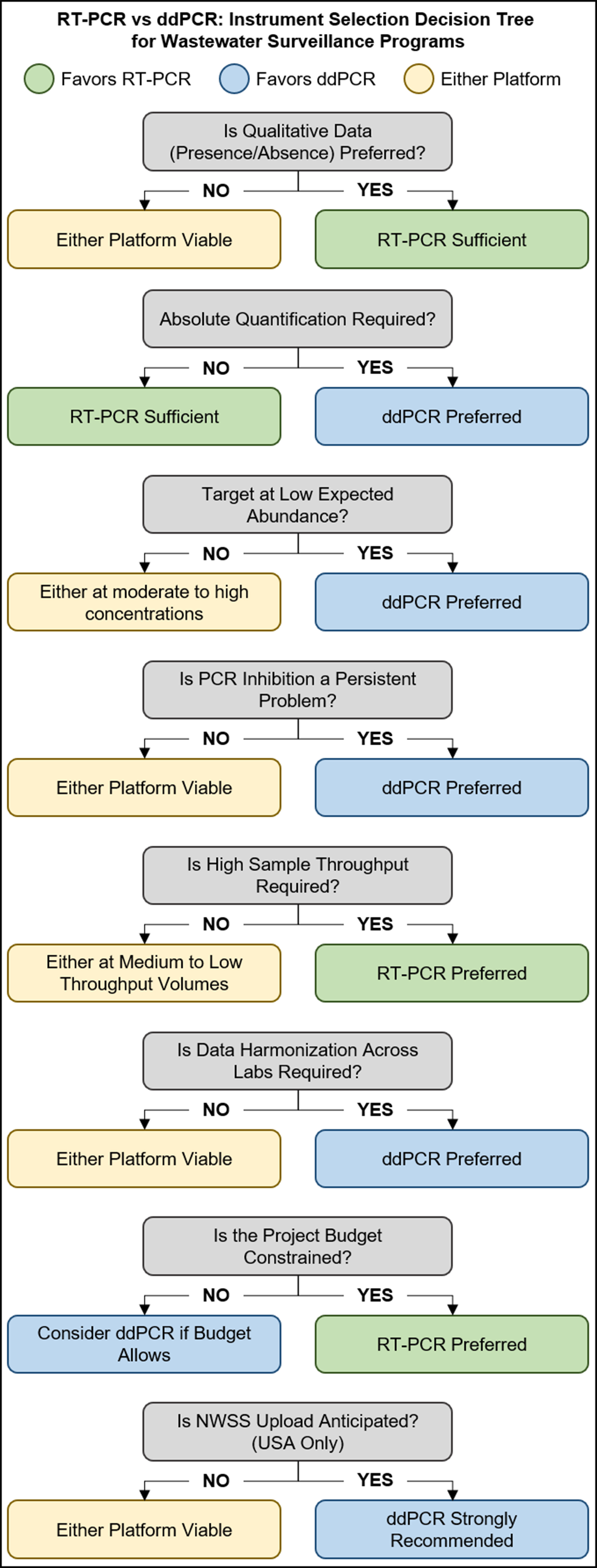
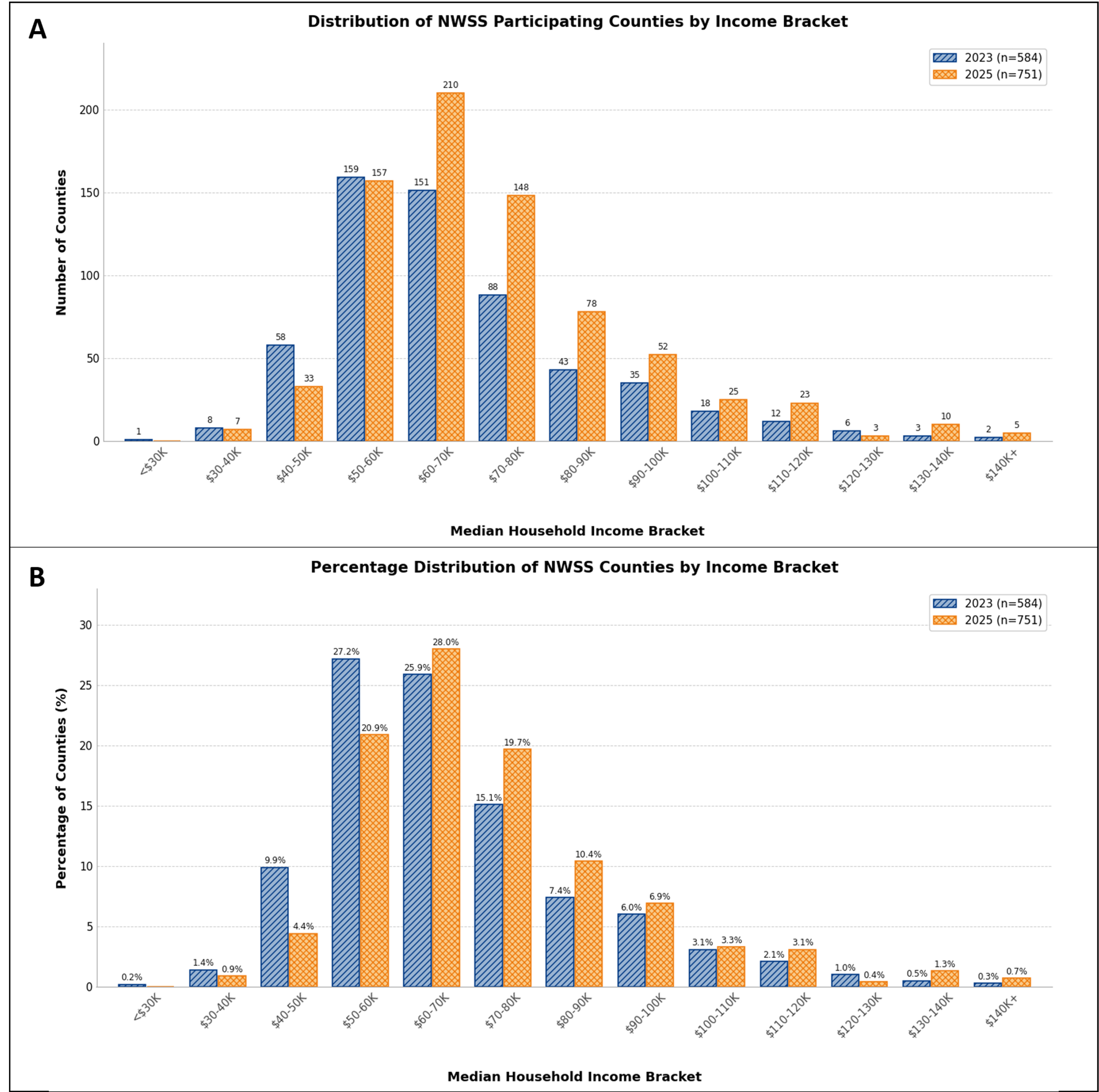
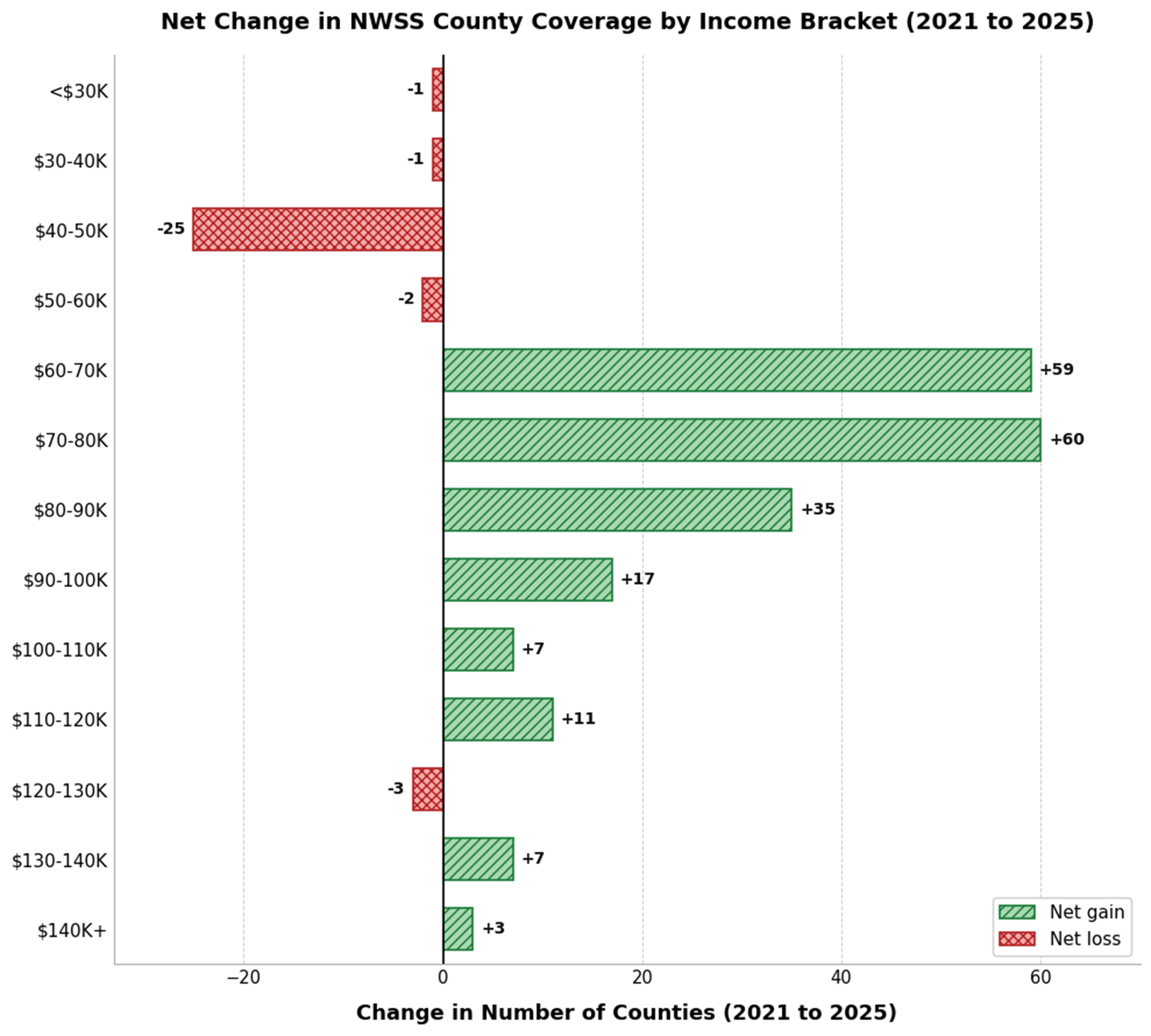











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.