Recent advances of carbon dots in lithium battery materials
Abstract
Carbon dots (CDs), an emerging class of zero-dimensional carbon nanomaterials, have attracted extensive attention for lithium-based energy storage due to their high specific surface area, tunable surface chemistry, excellent electronic conductivity, and abundant, readily functionalized surface states. Recent advances have demonstrated that CDs can serve as conductive bridges, chemical regulators, and interfacial stabilizers across all key components of lithium batteries, enabling the simultaneous optimization of electronic and ionic transport, as well as interfacial reactions, in cathodes, anodes, and electrolytes. This review systematically summarizes the synthesis strategies and structural classifications of CDs, emphasizing how precursor selection, heteroatom doping, and surface functionalization determine their core-shell structures, defect states, and chemical reactivity. Subsequently, the applications of CDs in cathode modification, anode reinforcement, and electrolyte optimization are discussed in detail, highlighting their roles in enhancing charge-transfer kinetics, modulating ion transport, stabilizing interphases, and suppressing lithium dendrite formation. Special attention is given to interfacial reconstruction mechanisms driven by heteroatom-doped or functionalized CDs, which simultaneously promote ionic conduction and electron blocking at solid-solid interfaces. Finally, current challenges and future directions are outlined, including predictive synthesis design, interfacial chemistry optimization, multiscale composite construction, and scalable green fabrication. Overall, this review aims to deepen the understanding of CD-mediated interfacial engineering and to provide design guidelines for the development of safe, long-life, and high-energy-density lithium-based batteries.
Keywords
INTRODUCTION
In recent years, lithium-based battery technologies have made significant advancements across multiple application areas, aiming to meet the increasing demand for high-efficiency energy storage systems in sectors such as electric vehicles and portable electronic devices. These improvements primarily aim to enhance energy density, reduce charging time, and improve battery safety. The continuous pursuit of high-energy-density and safe lithium-based batteries has driven intensive efforts to overcome critical challenges, such as sluggish ion and electron transport[1,2], unstable interfacial chemistry[3-5], and dendritic lithium growth[6,7]. Currently, strategies for improving the performance of lithium-ion batteries (LIBs) are generally categorized into material optimization, structural design, and process enhancement[8-11]. Material optimization focuses on selecting positive and negative electrode materials, designing high-stability electrolytes, and developing solid-state or gel polymer composite electrolytes[12,13]. For example, cathode material development has concentrated on representative oxide systems, including lithium cobalt oxide, lithium nickel oxide, and lithium manganese-based oxides[14-16]. Suitable anode materials such as graphite, silicon, and carbon nanotubes (CNTs) are employed to improve battery energy storage efficiency and charge-discharge rates[17,18]. In addition, high-concentration electrolytes, stable solvents, and functional additives are utilized, while solid-state or gel polymer composite electrolytes are developed to enhance ionic conductivity and dendrite resistance[19-21]. Regarding structural design, adopting multilevel electrode architectures, optimizing the ratio of positive to negative electrode materials, and applying surface coating techniques can enhance energy density, cycle life, and charge-discharge rates[22-24]. Process enhancement involves improvements in battery production, assembly, and encapsulation, as well as the optimization of internal structures and manufacturing techniques to enhance consistency and reliability[25-27]. For instance, micro/nano processing technologies and thin-film preparation methods can produce electrode materials with uniform structures and high specific surface areas, further boosting energy density and cycle life[28-31]. Additionally, automated production lines and efficient encapsulation technologies improve manufacturing efficiency and product quality[32-34]. In summary, enhancing the performance of LIBs requires the integrated application of multiple strategies, with material optimization as the top priority[35]. As new materials, technologies, and processes continue to emerge, the potential for performance improvement will expand further, providing more reliable and efficient energy solutions for sustainable development and new energy applications[36-38].
Among various improvement strategies, carbon materials have emerged as key components for enhancing the performance of cathodes[39], anodes[40], electrolytes[41], and separators, owing to their excellent electronic conductivity[42], facile chemical tunability[43], and multifunctional roles within electrodes and interfaces[44]. Carbon materials can serve not only as conductive networks and hosts for lithium metal deposition in lithium metal batteries (LMBs)[45], but also as catalytic sites, protective layers[46], or interfacial engineering elements, thereby significantly contributing to improvements in energy density and cycling stability[47]. Several intrinsic properties make carbon materials particularly well-suited for lithium battery applications. Their high electronic conductivity reduces internal electrode resistance and enhances rate capability[48]. A large specific surface area and tunable porosity increase reaction interfaces[49], enable higher active material loading, and buffer volume changes[50]. Additionally, heteroatom doping[45], surface functionalization, and defect engineering allow modulation of interactions with Li+ and soluble intermediates by creating lithiophilic sites or chemical anchoring points[51]. At the microscopic level, the primary mechanisms by which carbon materials improve electrochemical performance include: (1) constructing continuous electronic pathways to facilitate charge transport; (2) introducing lithiophilic sites through doping or functional groups to lower lithium nucleation overpotentials and promote uniform deposition, thereby suppressing dendrite formation; (3) providing porous structures and chemically active sites for the physical adsorption or chemical anchoring of soluble intermediates (e.g., polysulfides), which mitigates shuttle effects and enhances reversibility; and (4) serving as carbon-based artificial interphases or additive precursors to tailor the solid electrolyte interphase (SEI) and interfacial composition, thereby improving cycling stability[52].
Traditional carbon materials such as graphite, CNTs, and graphene suffer from limited surface reactivity, large particle sizes, and poor tunability, which restrict their ability to regulate interfacial processes at the nanoscale[53]. In contrast, carbon dots (CDs) can significantly enhance the electrochemical performance of materials, including improved cycling stability, Coulombic efficiency, and lithium-ion diffusion, factors that are crucial for the development of next-generation lithium batteries. Compared to traditional carbon-based materials, CDs can be easily modified and prepared into battery materials, exhibiting high Coulombic efficiency, extended cycle life, strong interfacial wettability, and high specific surface area due to their abundant surface functional groups. This versatility is primarily attributed to the diverse structural composition of CDs and their excellent interfacial compatibility with various battery materials[54]. CDs possess ultrasmall particle sizes, high specific surface areas, and abundant surface functional groups (e.g., -OH, -COOH, -NH2), enabling precise modulation of their electronic structure[55], surface polarity[56], and interfacial chemistry at the molecular level[54]. Structurally, CDs consist of a graphitic or amorphous carbon core encapsulated by a functionalized shell, combining the conductivity of sp2-carbon frameworks with the chemical reactivity of surface moieties[57].
Since their discovery in 2004, CDs have evolved into a tunable carbon platform that bridges molecular and graphitic domains[53]. By controlling precursor design, synthesis temperature, and heteroatom doping (e.g., N, S, F, P), their electronic and surface properties can be precisely tailored for specific lithium battery components. Such tunability enhances conductivity and stability in cathodes, improves SEI uniformity and mechanical robustness in anodes, and facilitates the formation of ion-conductive yet electron-blocking layers at electrolyte-solid interfaces[58]. Figure 1 illustrates the publication trend from 2014 to 2024, showing an exponential increase in studies combining the keywords “carbon dots” and “Li battery”. The corresponding keyword co-occurrence network in Figure 2 further reveals that “lithium-ion batteries,” “cathode,” “anode,” and “performance” serve as central research hubs, interconnected with emerging topics such as heteroatom doping and interfacial engineering. In the realm of electrode materials [Table 1], excellent cycling stability is achieved through various strategies, including doping or composite modification. These approaches optimize the stability of the negative electrode interface, facilitate the uniform deposition of lithium ions, suppress the formation of lithium dendrites, and enhance lithium storage capacity and cycling performance via microstructural design of composite materials. Regarding electrolytes, the functionalization of CDs (e.g., controlling surface states and doping with heterogeneous elements) optimizes electrolyte microstructure and interfacial compatibility with electrodes, thereby enabling stable cycling at high rates.
Figure 1. Annual publication trends (2014-2024) for studies containing the keywords “carbon dots Li battery”. (Data source: Web of Science) .
Figure 2. Keyword co-occurrence network of “carbon dots Li battery” over the past 10 years. (Data source: Web of Science) .
Representative CD-modified electrode materials and their electrochemical performance
| CD-based electrode | Rate performance (mAh g-1) | Cycling performance (mAh g-1) | Ref. |
| LC@CG (LiFePO4C/CNT with GQDs) | 107.1 at 20 C | 153.5 at 1 C after 300 cycles | [96] |
| GQDs@FFNA (FeF3·0.33H2O nanosheet array with GQDs) | 161.5 at 1 C, 117 at 20 C | 96 at 2 C after 1,000 cycles | [97] |
| C-VO CQD (VO2 nanoribbons on carbon cloth with CQDs) | 402 at 0.3 C, 168 at 60 C | 196 at 19.2 A g-1 after 500 cycles | [98] |
| NSCDs/S (N,S co-doped CDs-modified sulfur cathode) | 1,207.2 at 0.2 C, 894.9 at 2 C | 482.2 at 2 C after 400 cycles | [99] |
| LPC@S (lignin-derived porous carbon/CQDs-S cathode) | 1,066 at 1 C, 643 at 3 C | 659 at 1 C after 400 cycles | [100] |
| Lotus stem-derived porous carbon/GQDs hybrid | 738 at 0.2 C, 440 at 5 C | 440 at 5 C after 1,000 cycles | [104] |
| BN-CQDs (B,N co-doped CQDs self-supporting anode) | 130.4 at 3,000 mA g-1 | 485.13 at 0.1 A g-1 after 1,000 cycles | [105] |
| S-CDs/rGO (S-doped CDs embedded in reduced graphene oxide) | 938.8 at 0.1 A g-1, 294.1 at 3 A g-1 | 203.3 at 0.5 A g-1 after 1,000 cycles | [110] |
| Bi2Se3/CDs-200 (Bi2Se3 nanosheets with CDs) | 502 at 1 A g-1, 165 at 20 A g-1 | 502 at 1 A g-1 after 950 cycles | [111] |
| Si/CDs (CD-coated Si anode) | - | 1,000 at 1.77 A g-1 after 400 cycles | [113] |
| GQD/SiOx/C (GQD-decorated SiOx/C composite) | ~500 at 0.5 C | ~1,000 at 0.2 C after 1,000 cycles | [114] |
| GDY-Li QDs-modified Li metal (graphdiyne Li-rich QDs on Li) | ~200 at 0.1 C, ~140 at 5 C | 150 at 3 C after 120 cycles | [116] |
Despite rapid progress, a major bottleneck remains: establishing quantitative relationships among CD synthesis, structure, and battery performance, especially through interfacial mechanisms. To translate fragmented reports into actionable design principles, this review is organized according to a Structure-Function-Performance (S-F-P) framework [Figure 3] for lithium-based batteries, connecting (i) CD structural parameters (size, sp2/sp3 ratio, crystallinity/defect density, surface group density/charge, and heteroatom doping) to (ii) battery-relevant interfacial functions (conductive bridging, SEI/cathode electrolyte interphase (CEI) regulation, Li+ transport modulation/anion immobilization, and mechanical buffering), and ultimately to (iii) measurable electrochemical metrics (capacity retention, rate capability, Coulombic efficiency, polarization, lithium ion transference number (tLi+), and interfacial resistance).
Figure 3. Structure-Function-Performance (S-F-P) analytical framework for CDs in lithium-based batteries.
To explicitly establish and trace performance contributions, the interfacial functions discussed herein are described in terms of commonly reported electrochemical indicators. Conductive bridging is typically reflected in reduced polarization and slower impedance growth[59]; SEI/CEI regulation by higher Coulombic efficiency and stabilized interfacial resistance[60,61]; Li+ transport modulation/anion immobilization by higher ionic conductivity and increased tLi+ with reduced concentration polarization[62,63]. In solid electrolytes, electron-blocking interphase reconstruction is associated with increased critical current density (CCD) and prolonged symmetric-cell stability[64,65]. Where available, these relationships are further corroborated by interphase-sensitive characterization techniques (e.g., X-ray photoelectron spectroscopy (XPS), time-of-flight secondary ion mass spectrometry (ToF-SIMS), and operando Raman spectroscopy)[66,67], as well as deposition/morphology evidence.
CLASSIFICATION AND SYNTHETIC STRATEGIES OF CARBON DOTS
As an emerging material, CDs have attracted considerable attention in electrochemical energy storage and related applications due to their facile synthesis and high specific surface area. CDs are typically considered to have a core-shell structure, with the core comprising polycrystalline nanodomains containing small carbon clusters surrounded by amorphous domains, while the polymer surface imparts specific physicochemical properties. Structurally, CDs have at least one dimension smaller than 10 nm and comprise high-density nuclei of sp2/sp3-hybridized carbon atoms and heteroatoms[68,69]. As shown in Figure 4A, based on the carbon core and structural features, traditional classification systems categorize CDs into three types: graphene quantum dots (GQDs), carbon quantum dots (CQDs), and carbonized polymer dots (CPDs). This classification is based on differences in carbon-core morphology, the sp2/sp3 carbon ratio, surface chemistry, and the origin of photoluminescence[70,71]. From a battery design viewpoint, GQDs, CQDs, and CPDs differ in their dominant structural characteristics and therefore tend to excel in different interfacial roles. GQDs usually possess higher crystallinity and extended sp2 domains, favoring electronic percolation and the formation of robust conductive coatings[72]. CQDs commonly feature a balance between graphitic domains and abundant polar surface groups, along with tunable charge/doping, making them versatile as interfacial modifiers or additives[73]. CPDs typically contain an amorphous/polymeric shell with densely packed functional groups, which can enhance adhesion, mechanical buffering, and polymer compatibility in composite electrodes/electrolytes[74]. These battery-oriented distinctions are summarized in Table 2[75,76].
Figure 4. Classification and synthetic strategies of CDs. (A) Structural classification of CDs. (B) Schematic illustration of the two primary synthetic routes.
Battery-oriented comparison of GQDs, CQDs, and CPDs
| CDs class | Key structure | Dominant interfacial functions | Best-fit components | Typical limitations |
| GQDs | High crystallinity; extended sp2 domains; low polarity | Conductive bridging; electron percolation; thin coatings | Cathode coatings; conductive networks | Limited anchoring if inert; aggregation; weak standalone interphase control |
| CQDs | Mixed sp2/amorphous structure; rich polar groups; tunable charge/doping | SEI/CEI regulation; lithiophilicity;Li+ transport modulation | Electrolyte additives; surface modifiers; polymer matrices | Side reactions at high voltage if overly reactive; dosage sensitivity; possible electronic leakage (solid-solid) |
| CPDs | Carbon core + polymeric/amorphous shell; abundant functional groups | Adhesion; mechanical buffering; ion-conductive interphases (polymers) | Polymer electrolytes; binders/interlayers; flexible electrodes | Lower intrinsic conductivity; shell-chemistry-dependent stability; reproducibility challenges |
GQDs typically possess a disc-like or quasi-zero-dimensional core composed of single- or few-layer graphene fragments; with edges and defect sites functionalized by groups such as hydroxyl, carboxyl, and amino moieties. The relatively high sp2 carbon content enhances electronic conductivity and graphitic character, making GQDs particularly suitable for electrode modifications that require conductive scaffolds. Preparation methods such as electrochemical exfoliation can achieve an optimal balance between core conductivity and surface functionalization[72]. CQDs generally consist of small sp2 carbon clusters embedded within oxygen- or nitrogen-rich shells. Their photoluminescence is often attributed to surface states or molecular fluorophores derived from precursors[73]. Accordingly, CQDs offer considerable flexibility in surface chemistry and emission tuning. These properties have led to extensive investigations of CQDs as interfacial functional additives or electrolyte fillers to improve ion transport and interfacial behavior[74]. CPDs typically originate from the partial carbonization of polymeric or macromolecular precursors. Structurally, they exhibit a combination of sp2 and sp3-hybridized amorphous or semicrystalline domains while retaining substantial organic shells or residual polymer chains. Their luminescence is primarily dominated by shell-based molecular fluorophores and functional groups rather than a purely graphitic core. This characteristic provides CPDs with advantages for shell-targeted functionalization[75,76].
As illustrated in Figure 4B, the synthesis of CDs can generally be classified into two primary approaches: “top-down” and “bottom-up” strategies. The top-down method produces CDs by cleaving or exfoliating bulk carbon precursors such as graphite, graphene, CNTs, or fullerenes. Common techniques include arc discharge, laser ablation, electrochemical exfoliation, chemical oxidation, ultrasonication, and ball milling[77]. This approach is advantageous for preserving or generating highly graphitized sp2-rich carbon cores, making it valuable for applications that require excellent electronic conductivity, such as electrode surface modification or conductive additives. Among these methods, electrochemical synthesis/exfoliation has been widely recommended in several reviews for energy-material applications due to its controllable operating conditions and relatively mild processing environment[78]. However, certain top-down techniques, particularly those involving strong acid oxidation or high-energy treatments, tend to introduce oxygen-containing functional groups or residual metal and ionic impurities. These residues can react with commonly used lithium salts, compromising the stability of electrolytes and SEI[79]. Therefore, CDs synthesized via top-down methods typically require rigorous purification or post-treatment steps, such as solvent washing, dialysis, or thermal annealing under an inert atmosphere, to remove harmful impurities and tailor surface chemistry, thereby ensuring electrochemical compatibility in lithium battery systems[80,81].
In contrast, the bottom-up approach constructs CDs from small organic molecules or polymer precursors through condensation/polymerization, carbonization, and nucleation-growth processes[82-86]. Common methods include hydrothermal/solvothermal synthesis, microwave-assisted pyrolysis, thermal carbonization, template-assisted synthesis, and controlled polymerization[87]. Numerous studies and reviews have demonstrated that the bottom-up approach enables facile in situ heteroatom doping (e.g., N, S, P) and surface functionalization, while allowing precise control over size distribution, surface chemistry and porosity through appropriate selection of precursors, reaction temperature and time, and template or interfacial engineering[88-90]. These advantages render bottom-up CDs highly versatile as electrode modifiers, fillers in liquid or gel electrolytes, and interfacial stabilizers that promote uniform lithium plating and SEI formation[91]. Moreover, bottom-up synthesis offers inherent scalability and environmental friendliness[92].
Recent advances have also demonstrated that machine learning and closed-loop optimization can effectively guide hydrothermal and microwave-assisted processes to achieve target CD structures and properties[93-98]. Such data-driven approaches improve synthesis efficiency and reproducibility, providing a feasible pathway for scalable fabrication of CDs tailored for energy storage applications[99,100]. Despite these opportunities, the application of carbon-dot materials in lithium battery technology is still at an exploratory stage; issues such as CD aggregation during long-term cycling, interfacial stability under high-rate operation, and batch-to-batch consistency in large-scale production remain to be systematically addressed[101-103].
APPLICATIONS OF CARBON DOTS IN LITHIUM BATTERY COMPONENTS
Cathode materials
Regarding the application of CDs in lithium battery cathode materials, CDs are primarily employed as surface modifiers and nano-building blocks that enhance electrochemical performance through three interrelated mechanisms: (1) improving electronic conductivity and forming conductive bridges at the particle-collector interface[104]; (2) optimizing ion transport pathways by creating nanoscale channels and enhancing wettability[105]; and (3) stabilizing the electrode structure by buffering volume changes and suppressing detrimental side reactions[106]. Collectively, these mechanisms enable higher rate capability, improved capacity retention, and longer cycle life across various cathode chemistries.
For conventional cathode materials, the role of CDs as conductive and structural enhancers has been demonstrated in several representative studies. Kim et al.[107] designed a multidimensional nanocarbon composite (LC@CG) by incorporating GQDs into LiFePO4 (LFP)/CNT composites. In this system
Figure 5. (A) Schematic illustration of the synthesis processes of LC, LC@C, and LC@CG. Transmission electron microscopy (TEM) images of (B and E) LC, (C and F) LC@C, and (D and G) LC@CG powder samples at different magnifications. (A-G) Reprinted with permission from Ref.[107]. Copyright © 2023 Elsevier. (H) Schematic illustration of the synthesis of GQDs@FFNA. (I) Schematic diagram of GQD surface modification to inhibit the formation of an excessive SEI film. (H and I) Reprinted with permission from Ref.[108]. Copyright © 2019 Elsevier. (J) Schematic representation of the C-VO CQD interwoven nanowire electrode, showing efficient electron and (K) Li/Na ion transport pathways. (J and K) Reprinted with permission from Ref.[109]. Copyright © 2016 American Chemical Society.
In Li-S cathode systems, CDs serve complementary yet distinct functions, primarily focusing on chemical confinement and conversion kinetics. Fu et al.[110] synthesized N/S co-doped CDs (NSCDs) and employed them as cathode additives to immobilize soluble lithium polysulfides (LiPSs) and facilitate Li2S nucleation [Figure 6A and B]. The NSCD-modified cathode demonstrated a reversible capacity of 1,207.2 mAh g-1 at
Figure 6. Schematic diagrams of (A) the synthesis process and the application of the cathode in lithium-sulfur batteries (LSBs); (B) comparison between cathodes without NSCDs and with NSCDs; (C) Long-term cycling performance at 1 C and 2 C[110]. Reprinted with permission from Ref.[110]. Copyright © 2025 John Wiley & Sons.
Complementing the additive strategy, Xu et al.[111] demonstrated a NaCl-templated “two-in-one” valorization approach for converting lignin into both porous lignin-derived carbon (LPC) and lignin-derived CQDs [Figure 7A and B]. The LPC@S cathode achieved high sulfur utilization (~66 wt%), an initial discharge capacity of 1,066 mAh g-1 at 1 C, and 659 mAh g-1 after 400 cycles (capacity decay ≈ 0.095% per cycle, Coulombic efficiency > 98%) [Figure 7C and D]. In this combined approach, the hierarchical porosity of LPC confines sulfur and maintains fast ion/electron transport, while the lignin-derived CQDs provide surface functional groups that can be leveraged for interfacial tuning.
Figure 7. (A) Schematic of the conversion mechanism from lignin to porous carbon, including aromatic rebounding, refusion, and deoxygenation reactions. (B) Schematic illustration of a NaCl-templated strategy for the preparation of carbon quantum dots and porous carbon from lignin. (C) Rate performance at different current densities of 0.1, 0.2, 0.5, 1, 2, and 3 C, and (D) long-term cycling performance and corresponding Coulombic efficiencies at 1 C for 400 cycles. Reprinted with permission from Ref.[111]. Copyright © 2021 Elsevier.
Taken together, these studies reveal a consistent S-F-P relationship across cathode chemistries. In conventional intercalation cathodes, CDs mainly act as nanoscale conductive bridges and morphological stabilizers, filling interfacial gaps to preserve percolation networks and mitigate particle/electrode detachment under high-rate cycling. In Li-S systems, surface-functionalized CDs additionally provide adsorption and nucleation sites that regulate polysulfide conversion and reaction kinetics. Despite differences in application targets, both cases follow the same design rule: optimizing the sp2 carbon network continuity and the surface chemistry/spatial distribution of CDs enables simultaneous tuning of electronic connectivity, ion transport, and interfacial reactivity/CEI evolution. These functions are typically reflected in reduced polarization, slower impedance growth, and improved rate capability and capacity retention during long-term cycling.
Anode materials
Intercalation-type, conversion-type, and alloy-type anodes, along with lithium metal, constitute the principal classes of anodes in lithium-based batteries. Intercalation materials are valued for their cycling stability and high Coulombic efficiency; however, their rate capability and practical specific energy are limited by slow Li+ diffusion and insufficient electronic percolation. In contrast, conversion and alloy-type anodes offer substantially higher theoretical capacities but suffer from significant volume changes, structural
Intercalation-type anode
Intercalation materials offer good cycling stability, high Coulombic efficiency, and relatively controllable interfaces and structures. However, their specific capacity and energy density are limited by the number of available intercalation sites and, often, sluggish Li+ diffusion. Additionally, enhanced electronic percolation is typically required to achieve high-rate performance[112-114]. Sun et al.[115] employed a self-templated hydrothermal route combined with chemical activation to produce a lotus stem-derived hybrid porous anode [Figure 8A]. The resulting material comprises a carbon nanowire framework and a hierarchical pore network that promotes electrolyte infiltration and wetting, thereby increasing the electrochemically accessible surface area and reducing interfacial mass-transport resistance [Figure 8B-G]. GQDs embedded in or attached to the carbon skeleton strengthen electronic pathways and, together with the porous structure, buffer volume changes during cycling. The composite exhibits excellent electrochemical performance in both Li-ion and Na-ion systems, with reversible capacities of 738 and 460 mAh·g-1 at 0.2 C, respectively, and maintains high capacity under prolonged high-rate cycling. These results demonstrate that the GQD-based design simultaneously improves interfacial contact, charge and ion transport, and mechanical stability.
Figure 8. (A) Schematic illustration of the synthesis procedure for LPC materials. (B) Low-magnification transmission electron microscopy (TEM) image of the LPC sample. (C) High-magnification TEM image of LPC showing a reticular structure, with an inset of a locally magnified image of GQDs. (D) Corresponding high-resolution transmission electron microscopy (HRTEM) image of GQDs. (E) High-magnification TEM image of LPC showing a self-supporting porous structure. (F) Local magnified image of the regions indicated by the yellow squares in (E). (G) Corresponding HRTEM image. Reprinted with permission from Ref.[115]. Copyright © 2024 John Wiley & Sons.
Kim et al.[116] synthesized boron and nitrogen co-doped CQDs (BN-CQDs) via a facile hydrothermal method and demonstrated their application as a self-supporting anode material for LIBs [Figure 9A]. The 0.5 BN-CQD electrode delivered a capacity of 485.13 mAh·g-1 at 100 mA·g-1 and maintained 130.4 mAh·g-1 at an ultrahigh current density of 3,000 mA·g-1 (approximately 27.9% of the initial value), with 88% capacity retention after 1,000 cycles. Mechanistically, B/N co-doping enhances electronic conductivity, supporting ultrafast charge transport, while B incorporation forms C-B bonds and promotes the formation of surface C=O groups; these surface defect sites increase Li+ affinity and provide additional reversible storage sites [Figure 9B]. By tailoring surface functionality, BN-CQDs overcome the limitations of oxygen-rich pristine CQDs (restricted Li+ accessibility and low conductivity) and achieve synergistic improvements in conductivity, Li+ affinity, and reversible capacity, positioning BN-CQDs as promising carbon-based self-supporting anode materials. Khan et al.[117] constructed a layered anode comprising lithium titanate (LTO), N-doped GQDs (N-GQDs), and Super-P conductive carbon. N-GQDs form a dense transport layer on the LTO surface that protects against air and electrolyte-induced degradation, shortens electron transport pathways, and slightly reduces the surface valence state of Ti, collectively enhancing electronic conductivity [Figure 9C]. The LTO-N-GQD composite achieves approximately a 23% improvement in rate performance at 50 C and demonstrates robust stability at high rates, indicating that N-GQDs function both as conductive conduits and protective surface layers.
Figure 9. (A) Schematic of the experimental procedure for preparing N-CQDs and BN-CQDs. (B) Illustration of the advantages of the surface functional group-tailored BN-CQD structure. (A and B) Reprinted with permission from Ref.[116]. Copyright © John Wiley & Sons. (C) Schematic diagrams showing the formation of the hierarchical LTO/N-GQD/Super-P structure. (C) Reprinted with permission from Ref.[117]. Copyright © 2019 Elsevier. BN-CQDs, boron and nitrogen co-doped carbon quantum dots; N-CQDs, nitrogen-doped carbon quantum dots.
Conversion-type anodes
Conversion-type anodes, while offering high capacity, often undergo significant volume changes and loss of electrical contact. In these systems, CDs commonly serve a dual function: as conductive and flexible scaffolds that maintain electron transport networks, and as surface-active agents that modulate reaction pathways and product nucleation[118-120]. Gottam et al.[121] synthesized spherical S-doped CDs embedded in reduced graphene oxide (S-CDs/rGO) by annealing benzyl disulfide (BDS) with graphene oxide (GO) [Figure 10A]. The composite exhibits a high initial capacity (938.8 mAh·g-1 in the first cycle) and strong long-term retention at high rates (~78.8% at 1,000 mA·g-1). In addition, an S-CDs/rGO|LFP full cell retains
Figure 10. (A) Schematic representation of S-CDs/rGO preparation via the annealing of GO and BDS in a tube furnace. (B) Schematic illustration of the LIB full cell with LiFePO4 as the cathode and S-CDs/rGO-900 as the anode. (A and B) Reprinted with permission from Ref.[121]. Copyright © 2021 John Wiley & Sons. (C) Schematic illustration of the fabrication process of pure Bi2Se3 and Bi2Se3/CDs composites, along with a mechanism analysis of lithium storage in the Bi2Se3/CDs-200 electrode. (D and E) In situ X-ray diffraction (XRD) patterns with corresponding galvanostatic charge-discharge (GCD) curves, and (F) atomic model illustrating the phase change of the Bi2Se3/CDs-200 electrode during the initial cycle. Nyquist plots of Bi2Se3/CDs-200 at different voltage points during the third discharge (G) and charge (H) processes. (C-H) Reprinted with permission from Ref.[122]. Copyright © 2022 Elsevier.
Alloy-type anodes
Alloy-type anodes, particularly Si, offer remarkably high theoretical capacities but are prone to severe volume expansion. CDs function as “nano-glues” or conductive coatings that reinforce particle-binder interfaces, enhance mechanical integrity, and preserve electrical contact[123]. Du et al.[124] prepared Si/CDs composites via impregnation: CDs uniformly coat Si particles and improve interactions with the binder, thereby mitigating pulverization and contact loss [Figure 11A-E]. Compared to bare Si, the Si/CD electrode retains 64.0% of its capacity after 150 cycles at 0.2 C (approximately twice that of the pristine Si electrode) and operates stably at 1,000 mAh·g-1 for 400 cycles. These results demonstrate that CDs act as flexible conductive layers and adhesion enhancers, maintaining long-term electrical connectivity and structural integrity. Hwang et al.[125] designed GQD/SiOx/C composites in which GQDs and carbon nanoparticles form internal cavities and conductive networks within a SiOx matrix [Figure 11F]. An optimized composition (~15 wt% SiOx) delivers an initial capacity of 768 mAh·g-1 (initial Coulombic efficiency of 83%) and retains approximately 81% of the initial capacity at 2 C relative to 0.1 C, highlighting the dual roles of GQDs in buffering volume changes and enhancing conductivity.
Figure 11. (A and B) HRTEM images of Si nanoparticles; (C and D) HRTEM images of Si/CDs. (E) Schematic illustration of the interaction between Si/CDs and the binder. (A-E) Reprinted with permission from Ref.[124]. Copyright © 2021 Springer. (F) SEM and TEM images of the GQD/SiOx/C composite. (F) Reprinted with permission from Ref.[125]. Copyright © 2024 MDPI.
Lithium metal anodes
Lithium metal anodes offer the highest theoretical capacity but suffer from dendrite formation, an unstable SEI, and parasitic reactions. CD-based approaches address these failure modes through ion adsorption and nucleation control, as well as by introducing lithium-rich CD species that serve as active lithium reservoirs and nucleation directors[126]. Shen et al.[127] synthesized graphdiyne-lithium quantum dots (GDY-Li QDs) approximately 2.6 nm in size, characterized by high lithium content and strong ion adsorption. When applied to lithium metal anodes, GDY-Li QDs concentrate Li+ ions, modulate the solvation structure, and provide uniform nucleation sites, creating a nano-confinement effect that suppresses dendrite growth and achieves Coulombic efficiencies of up to approximately 99.3% in carbonate electrolytes [Figure 12A-D]. Full cells pairing GDY-Li QD-treated lithium anodes with LiNi0.8Co0.1Mn0.1O2 (NCM811) cathodes exhibit excellent high-rate stability, demonstrating the potential of Li-rich CDs as active functional additives for Li metal protection.
Figure 12. Synthesis and structure of self-assembled GDY-Li QDs. (A) General preparation strategy of self-assembled metallized GDY QDs. (B) Synthesis route of HEB-Li. (C) Charge density distribution (left: isosurface, 0.03 e Å-3; right: slice) of HEB and HEB-Li molecules. (D) Schematic illustration of the formation of self-assembled GDY-Li QDs. Reprinted with permission from Ref.[127]. Copyright © 2024 Chinese Chemical Society.
Across intercalation-, conversion-, alloy-, and lithium metal-based anodes, a set of convergent, structure-enabled functions is enabled by CDs. Conductive bridging is facilitated by the nanoscale dimensions and sp2-rich domains of CDs, whereby interparticle voids are filled and percolation pathways restored, thereby lowering ohmic losses and supporting high-rate operation. Interfacial chemical regulation is further promoted by heteroatom doping and polar surface functionalities, which provide adsorption/nucleation sites, steer conversion pathways, tune SEI chemistry, and homogenize Li deposition. In parallel, electrode integrity and architecture are reinforced via nano-adhesion/coating effects and, in selected cases, through self-assembly into robust conductive frameworks, leading to improved particle-binder cohesion and preserved electrical contact under large volume changes[128]. From a S-F-P perspective, CD structural features (sp2 continuity, defect/edge density, and surface polarity) translate into interfacial functions (charge-transport bridging, reaction/deposition modulation, and SEI stabilization), which are reflected in reduced charge-transfer resistance, diminished polarization/voltage hysteresis, improved rate capability, and enhanced capacity retention. A practical boundary condition should be considered, in which excessive CD loading or voltage-mismatched surface chemistry may be associated with intensified parasitic reactions and accelerated interphase growth, thereby lowering the initial Coulombic efficiency. Therefore, voltage- and component-specific optimization of CD composition and dosage is recommended.
Electrolytes
Electrolytes govern ion transport efficiency and interfacial stability in both Li-ion and Li-metal batteries. Compared with electrodes, electrolytes more directly determine bulk ionic conductivity, the Li+ transference number, and the formation/evolution of SEI/CEI layers during long-term cycling. Liquid electrolytes are characterized by intrinsically high ionic conductivity and excellent wetting properties; however, they suffer from flammability, relatively narrow electrochemical stability windows, and a strong tendency to trigger uncontrolled lithium dendrite growth and parasitic side reactions at high current densities. Conversely, solid electrolytes are nonflammable and mechanically robust but are generally limited by low room-temperature ionic conductivity, high interfacial resistance, and challenges in engineering and processing. In this context, CDs, owing to their tailorable surface functionalization, quantum size effects, and nanometer-scale dimensions, have been widely explored to tune the microenvironment and interfacial chemistry of liquid, polymer, and oxide electrolytes. By simultaneously serving as ion-coordination centers, interfacial modifiers, and mechanical/structural regulators, CDs constitute a multifunctional tool to improve battery safety and lifespan. In the following subsections, we categorize representative CD-based electrolyte designs by electrolyte type, summarize the underlying mechanisms, and highlight typical performance improvements[129].
Liquid electrolytes
Incorporating functional additives into liquid electrolytes is a direct and effective strategy for suppressing dendrite growth[130-132]. Compared with conventional molecular additives, nanoscale CDs provide a higher density of lithiophilic sites and a larger effective surface area, enabling more efficient regulation of Li+ solvation and deposition behavior at the electrode-electrolyte interface. Li et al.[133] proposed and demonstrated the use of nitrogen and sulfur co-doped CDs (NS-CDs) as deposition-regulating additives in conventional ether-based electrolytes. At a concentration of 0.3 mg·mL-1, NS-CDs are well dispersed in the electrolyte; their abundant N- and S-containing surface groups impart pronounced lithiophilicity, enabling the CDs to adsorb surrounding Li+ ions and co-deposit on the current collector to form a dense and uniform Li deposition layer. This process was directly observed using confocal laser scanning microscopy
Figure 13. (A) Schematic illustration of the role of CDs in stabilizing lithium metal anodes. (B) Model structures showing the binding energies between Li ions and distinct lithiophilic sites based on DFT calculations. (C) Schematic illustration of the formation mechanism of NS-CDs. (A-C) Reprinted with permission from Ref.[133]. Copyright © 2021 Elsevier. (D) Schematic illustration of the synthesis of M-CPDs and H-CPDs. (E) Model structures of graphite-N, pyrrolic-N, and pyridinic-N. (F) Digital photographs of electrolytes containing M-CPDs and H-CPDs under ultraviolet (UV) light. Schematic illustration of Li deposition on Cu foils in the electrolyte. (G) Without CPD additives; with additives of (J) M-CPDs, (M) H-CPDs. SEM images of (H, K, N) Cu foil surfaces and (I, L, O) Li sheet surfaces after 50 cycles of Li//Cu half-cells at a current density of 1 mA cm-2. (D-N) Reprinted with permission from Ref.[134]. Copyright © 2023 John Wiley & Sons.
Wang et al.[134] designed CPDs as electrolyte additives, synthesizing them via microwave (M-CPDs) and hydrothermal (H-CPDs) methods. H-CPDs, produced under milder conditions [Figure 13D], contain a higher concentration of lithiophilic functional groups (pyridinic-N, pyrrolic-N, -COOH) and remain stable in conventional electrolytes for at least three months [Figure 13E and F]. CPDs form stable Li-CPD clusters with Li+ ions through electrostatic interactions, acting as adsorption-nucleation sites that guide uniform lithium deposition and suppress tip effects and dendrite formation [Figure 13G-O]. As the content of lithiophilic groups increases, CPDs more effectively disrupt Li+ solvation, promoting faster Li+ transport and diffusion. In terms of performance, H-CPDs enable highly uniform Li+ transport: Li|Li symmetric cells with H-CPDs cycle stably for over 3,700 h with a voltage hysteresis of approximately 0.38 mV, and Li|LFP full cells (1 C) retain 93.8% of their capacity after 200 cycles (compared to 61.2% without additives and 83.7% with M-CPDs). Overall, CPDs are easy to synthesize, chemically stable in electrolytes, and offer a practical engineering approach to suppress dendrite formation and enhance Li-metal battery stability by providing lithiophilic sites, tuning the solvation structure, and creating uniform nucleation channels.
Polymer electrolytes
Introducing CDs into polymer electrolytes enables precise microstructural control. Unlike liquid additives, which primarily influence deposition behavior, CDs embedded within polymer matrices serve as both functional fillers and physical-chemical cross-linkers, thereby intrinsically modifying polymer crystallinity and ion transport pathways[135-140]. Liu et al.[141] designed multifunctional (O, N, S and F) carbon dots (NSFCDs) and incorporated them in situ with poly(ethylene glycol) diacrylate (PEGDA) into a polyethylene oxide (PEO)-based matrix via free-radical polymerization to form a cross-linked hybrid polymer electrolyte (HPE) [Figure 14A-F]. NSFCDs serve as both functional fillers and cross-linking units, creating a dense network that enhances mechanical strength and interfacial adhesion. Their surface polyfunctional groups reconfigure polymer chain conformations and Li+ coordination, reducing Li+ migration barriers and establishing efficient ion transport channels. Simultaneously, NSFCDs interact with lithium salt anions to modulate SEI composition and morphology, promoting uniform Li deposition and suppressing dendrite growth
Figure 14. (A) Schematic diagram of the formation of NSFCDs. (B) Schematic diagram of cross-linking between NSFCDs and PEGDA under UV irradiation. 1H nuclear magnetic resonance (NMR) (C) and 13C NMR (D) spectra of NSFCDs. (E) TEM image of NSFCDs. (F) HRTEM image of NSFCDs. (G) Schematic diagram of polymer conformation in HPE. (A-G) Reprinted with permission from Ref.[141]. Copyright © Elsevier. (H) Solvothermal synthesis of CDs from citric acid (CA) and ethylenediamine (EDA), and the subsequent reaction between CDs and PEG to produce (I) PEG-CDs (DCC and DMAP are 1,3-dicyclohexylcarbodiimide and 4-dimethyl aminopyridine, respectively). (J) TEM image of CDs and the inset HRTEM image. (K) TEM image of PEG-CDs and the inset HRTEM image. (L) Fourier transform infrared (FTIR) spectra of CDs and PEG-CDs. (M) Schematic illustration of the mechanisms by which PCCE improves ion conductivity and lithium ion transference number. (H-M) Reprinted with permission from Ref.[142]. Copyright © John Wiley & Sons.
Huang et al.[142] prepared a cross-linked polyethylene glycol (PEG)-CD network via a 1,3-dicyclohexylcarbodiimide (DCC)/4-dimethylaminopyridine (DMAP)-catalyzed reaction between PEG and CDs, and subsequently combined it with polyvinylidene fluoride (PVDF) and a liquid electrolyte to form a composite electrolyte (PCCE). PEG-CDs reduce PVDF crystallinity, forming continuous amorphous conductive domains, while also providing a robust framework that improves mechanical, thermal, and electrochemical properties [Figure 14H-M]. The PCCE shows an increased Li+ transference number and promotes the formation of a stable SEI, resulting in uniform Li deposition and effective dendrite suppression. Room-temperature long-term cycling of Li|PCCE|LFP and Li|PCCE|LiCoO2 cells demonstrates performance and capacity retention superior to PVDF-based gel polymer electrolytes (PVDF-GPEs) and liquid electrolyte controls. Combined with its low cost, the PCCE is an attractive candidate for gel polymer electrolytes and separators in Li-metal batteries.
Liu et al.[41] proposed a CDs-induced supramolecular physical cross-linking strategy to significantly enhance ion transport and interfacial stability in polymer electrolytes [Figure 15A]. By exploiting the quantum size effect and abundant surface functional groups of CDs, the authors constructed a hydrogen-bonded physically cross-linked network between CDs and polymer chains, which suppressed crystallinity and created diverse, efficient ion transport channels [Figure 15B]. As a result, the ionic conductivity reached 3.20 mS·cm-1 at
Figure 15. (A) Schematic illustration of the preparation process of CDPE. (B) Schematic illustration of the structural differences between PDOL and CDP. (C) SEM images of the lithium metal anode surface obtained after 50 cycles in Li||Li symmetric cells. (D) Schematic illustration of the mechanisms of SEI formation and lithium-ion deposition in PDOL and CDPE. (E-G) XPS analysis of the lithium metal surface after long-term cycling: (E) C 1s spectrum, (F) F 1s spectrum, and (G) Li 1s spectrum. (H) Schematic illustration of lithium-ion transport pathways in CDPE. (A-H) Reprinted with permission from Ref.[41]. Copyright © John Wiley & Sons. (I) Schematic diagram of the synthesis process of HF-CDs. (J) Digital photographs of PEO/P-FCD electrolyte slurry, electrolyte membrane, and membrane bending and stretching. (K and L) SEM cross-sectional images of the PEO/P-FCD composite electrolyte at different magnifications. (M) Elemental mapping images of chlorine (Cl) and fluorine (F). (N) Fluorescence image of the composite electrolyte. (I-K) Reprinted with permission from Ref.[143]. Copyright © American Chemical Society.
Xu et al.[143] designed highly fluorinated CDs (HF-CDs) for composite solid electrolytes and long-cycle operation. Prepared via a Claisen-Schmidt reaction under ambient conditions [Figure 15I], the HF-CDs achieved a maximum fluorine content of 20.79 at.%. HF-CD-based composite electrolytes exhibited a room-temperature ionic conductivity of 7.5 × 10-5 S·cm-1, along with improved mechanical properties and an expanded electrochemical window compared to the pristine polymer [Figure 15J-N]. The fluorine functionality promotes interfacial fluorination and enhances interfacial stability: Li symmetric cells cycled at 0.4 mA·cm-2 demonstrated low overpotential and stable operation for 1,000 h, while LFP|PEO/HF-CD composite electrolyte (P-FCD)|Li full cells retained 130.3 mAh·g-1 after 300 cycles, corresponding to 95.10% capacity retention. This study illustrates how HF-CDs can be rationally employed to tune PEO-based electrolytes for high-performance solid-state Li batteries.
Oxide electrolytes
Although polymer-composite electrolytes significantly enhance many properties, the greatest value of CDs arguably lies in organic-inorganic interfacial engineering for oxide electrolytes[144]. Liu et al.[145] doped Li6.7La3Zr1.75Ta0.25O12 (LLZTO) with NSFCDs and subjected the mixture to thermal treatment to obtain LLZTOCDs filler, which was subsequently incorporated into a PEO-based composite electrolyte to achieve multi-interface synergistic regulation [Figure 16A]. Thermal treatment converted the surface Li2CO3 into ion-conductive but electron-insulating LiF/Li3N intermediate layers, while CDs self-assembled into an organophilic coating that bridged LLZTO and the polymer matrix, enhancing interfacial compatibility and ion-exchange kinetics [Figure 16B]. Oxygen vacancies at the LLZTOCDs interface generated an anion-confinement effect that promoted salt dissociation and increased the Li+ transference number to approximately 0.85, facilitating the formation of inorganic-rich SEI phases[146]. The LLZTOCD-containing composites exhibited high ionic conductivity (~1.96 × 10-4 S·cm-1 at 25 °C) and effectively suppressed dendrite growth, resulting in solid-state lithium metal cells with excellent electrochemical stability and extended cycle life. This CD-mediated reconstruction of the organic-inorganic interface provides a practical approach for developing high-performance solid electrolytes.
Figure 16. (A) Schematic representation of the synthesis of LLZTOCDs. (B) Schematic diagram of ion transport pathways within two composite polymer electrolytes (CPEs). (A and B) Reprinted with permission from Ref.[145]. Copyright © John Wiley & Sons. Interfacial behaviors of Li-0.5 % FCD with an LLZTO pellet. (C-E) Digital photographs of the LLZTO pellet pre- and post-dipping in Li-0.5 % FCD composite. (F) Cross-sectional SEM images of Li-0.5 % FCD LLZTO. (G) Atomic structures of C, Li2O, LiF, and LLZTO. (H) Interfacial energies of different interfaces. (C-H) Reprinted with permission from Ref.[147]. Copyright © John Wiley & Sons.
Zhu et al.[147] demonstrated an in situ approach to form a Li-FCD composite at the LLZTO|Li interface by reacting trace amounts of fluorinated CDs (0.5 wt% FCDs) with molten lithium. The Li-FCD layer spontaneously concentrated carbon, Li2O, and LiF at the LLZTO interface [Figure 16C-F]. DFT calculations indicated that the significant interfacial energy differences among the C-Li2O-LiF/LLZTO and C-Li2O-Li/LLZTO interfaces favor strong adhesion and low contact angles, thereby promoting intimate wetting and uniform Li+ deposition [Figure 16G-H]. In this system, CDs serve multiple roles: they catalyze the in situ formation of inorganic blocking phases (LiF/Li2O) that suppress electronic conduction and dendrite growth; their carbon component provides moderate electronic conductivity to homogenize the local electric field and guide uniform deposition; and the derived interface lowers Li migration barriers, thereby establishing smooth ion transport channels. The modified interface achieved low interfacial resistance
Similarly, Zhu et al.[148] synthesized Li-NCD composites by reacting N-doped CDs (NCDs) with molten lithium to engineer an LLZTO|Li interface that is ionically conductive yet electronically insulating. This reaction produces Li2O and Li3N at the interface. Finite element simulations and DFT calculations indicate that these phases inhibit electron penetration, while Li3N offers high ionic conductivity, facilitating interfacial ion migration and uniform lithium deposition. The carbon component of the NCDs also provides moderate electron pathways, which help homogenize the electric field and enhance wetting and interfacial adhesion [Figure 17A-C]. The modified interface exhibits a low interfacial resistance of approximately 6.3 Ω·cm2 and a CCD of approximately 1.1 mA·cm-2. Li-NCD|LLZTO|Li-NCD symmetric cells demonstrate excellent cycling stability, lasting over 5,000 h at 0.1 mA·cm-2 and approximately 850 h at 0.4 mA·cm-2. Additionally, LFP|LLZTO|Li-NCD full cells retain approximately 92.7% of their capacity after 100 cycles, delivering
Figure 17. (A) Schematic illustration of the assembly procedure for the semi-solid-state battery. Schematic illustrations of (B) the Li-NCD|LLZTO interface and (C) the Li|LLZTO interface. (D) Cycling performance at 1 C. Reprinted with permission from Ref.[148]. Copyright © 2019 Electrochemical Society.
Multidimensional and synergistic functions are enabled by CDs across diverse electrolyte classes. In liquid electrolytes, lithiophilic adsorption/nucleation sites and solute-solvent clustering effects are introduced, thereby homogenizing Li deposition and regulating nucleation. In polymer electrolytes, crystallinity suppression and the formation of physical/chemical network structures are promoted, together with the development of CD-derived interphases, resulting in enhanced ionic conductivity, increased transference number, and improved interfacial durability. At inorganic oxide-electrolyte interfaces, chemical activation and interfacial self-assembly are facilitated, enabling integrated interface engineering characterized by electron blocking, ion conduction, and improved wetting/contact. From a S-F-P perspective, the structural features of CDs-particularly surface charge/polar functional groups and tailored heteroatom doping-translate into interfacial functions including salt dissociation/anion immobilization, interphase stabilization, and reconstruction of solid-solid interfaces with electron blocking. These functions are reflected in higher ionic conductivity and transference number (t+), reduced interfacial resistance, and, for solid electrolytes, increased CCD and extended transference number symmetric cells. Where feasible, these structure-function relationships are recommended to be further validated through in situ/operando characterization of SEI/CEI evolution and lithium deposition morphology.
CONCLUSION AND OUTLOOK
CDs have been established as a highly tailorable nanocarbon platform for lithium batteries, with their electrochemical relevance arising from the coupled tunability of carbon core structure and surface structure. Across cathodes, anodes, and electrolytes, convergent functions have been enabled, including percolation restoration and nanoscale gap filling for conductive bridging, interphase regulation for stabilized SEI/CEI evolution, and interfacial reconstruction for improved contact and suppressed polarization. Under a unifying S-F-P logic, these functions are most directly reflected by reduced polarization and slower impedance growth, together with improved Coulombic efficiency, rate capability, and capacity retention; for solid electrolytes, additional gains are manifested by reduced interfacial resistance, increased CCD, and extended symmetric-cell stability.
Several constraints remain before broad practical application can be realized:
(1) Limited reinforcement and conductivity as standalone additives. Compared with macroscopic carbon networks, CDs typically provide limited mechanical reinforcement and moderate intrinsic conductivity. As a result, composite co-engineering is frequently required to concurrently satisfy electrical, mechanical, and chemical requirements.
(2) Reproducibility limited by structural variability and impurities. Batch-to-batch variations in size distribution, defect characteristics, and surface chemistry remain a major source of irreproducibility. In addition, impurity residues introduced during synthesis/purification may intensify parasitic reactions with common salts/solvents, thereby destabilizing SEI/CEI chemistry.
(3) Insufficient device-level evidence under practical conditions. Performance and failure statistics under high areal loading, lean electrolyte conditions, elevated temperature, and realistic operation remain underreported. Moreover, quantitative relationships linking CD structure, interfacial evolution, and degradation mechanisms have yet to be systematically established.
Accordingly, three coupled directions are suggested to accelerate progress:
(1) Structure-controlled, impurity-managed, and scalable synthesis. Continuous or modular production routes should be advanced to ensure verifiable batch-to-batch consistency in size distribution, sp2/sp3 hybridization, defect characteristics, surface chemistry, and impurity profiles.
(2) Operando-corroborated interfacial causality and quantitative correlation. In situ/operando tracking of SEI/CEI evolution and metal deposition behavior should be combined with quantitative correlations between CD structural parameters and kinetic/transport metrics (e.g., charge transfer resistance (Rct), diffusivity, t+, and exchange current density), enabling structure-function relationships to be translated into predictive design rules.
(3) Manufacturability as a primary design constraint. Process compatibility with industrial electrode fabrication should be explicitly addressed (slurry rheology, binder interactions, coating uniformity, drying/thermal stability, calendaring tolerance, and electrolyte wetting). Without such considerations, interfacial benefits may be offset by aggregation or processing-induced segregation.
Although this review centers on lithium batteries, these interfacial principles are expected to remain applicable to other interface-limited chemistries (e.g., Na-metal systems), supporting cross-platform relevance[149]. Only voltage-matched, manufacturable, and operando-validated CD interphases are expected to translate laboratory-level gains into robust device-level benefits.
DECLARATIONS
Authors’ contributions
Data sourcing, data collection, and original draft writing: Liu, X.
Literature search, data curation, and original draft writing: Wang, P.; Liu, Q.; Xu, R.; Yao, J.; Lyu, Y.; Liu, D.
Supervision, original draft writing, and manuscript review: Zhai, F.; Wang, X.
All authors reviewed and approved the final version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-Assisted Tools Statement
An artificial intelligence (AI) image-generation tool was used to generate a conceptual schematic element in the Graphical Abstract. The image was created using the ChatGPT (OpenAI) image generation tool via the ChatGPT web interface (the model/version was not disclosed by the platform). The prompt (summary) was: “Generate a horizontal lithium-battery cross-section schematic with carbon-dot nanoparticles distributed in the electrolyte and near both electrode interfaces; no text.” The AI-generated output was used solely as a visual schematic. The final Graphical Abstract layout and all scientific content were developed and verified by the authors.
Financial support and sponsorship
The authors are grateful for the financial support from Project 2024CXPT035 under the Shandong Province Key R&D Program (Competitive Innovation Platform), the Taishan Scholars Program, Project ZR2023ME061 supported by the Shandong Provincial Natural Science Foundation, and the special supporting project for provincial leading talents of Yantai, the National Key Research and Development Program of China (2022YFB4600102).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Lan, J.; Cao, Y.; Geng, C.; et al. Anion regulation for high-performance lithium-sulfur batteries. Adv. Mater. 2025, 37, e2505527.
2. Zhou, Y.; Wang, P.; Wang, K.; et al. Developing high‐performance anode‐free lithium batteries: challenges, strategies, and opportunities. Adv. Funct. Mater. 2025, 35, 2424022.
3. Huang, Z.; Lyu, H.; Greenburg, L. C.; Cui, Y.; Bao, Z. Stabilizing lithium-metal electrodes with polymer coatings. Nat. Energy. 2025, 10, 811-23.
4. Cui, H.; Song, Y.; Ren, D.; Wang, L.; He, X. Electrocapillary boosting electrode wetting for high-energy lithium-ion batteries. Joule 2024, 8, 29-44.
5. Xu, Y.; Liu, X.; Wang, S.; et al. A mechanical strategy of surface anchoring to enhance the electrochemical performance of ZnO/NiCo2O4@nickel foam self-supporting anode for lithium-ion batteries. Adv. Compos. Hybrid. Mater. 2024, 7, 238.
6. Yang, C.; Singh, A.; Pu, X.; et al. Addressing the safety of next-generation batteries. Nature 2025, 645, 603-13.
7. Bae, J.; Choi, K.; Cheong, J. Y.; et al. Emerging surface engineering methods for lithium metal anodes: critical review beyond conventional SEI and surface coatings. Adv. Mater. 2025, 37, e2501959.
9. Ziegler, M. S.; Trancik, J. E. Re-examining rates of lithium-ion battery technology improvement and cost decline. Energy. Environ. Sci. 2021, 14, 1635-51.
10. Wu, F.; Maier, J.; Yu, Y. Guidelines and trends for next-generation rechargeable lithium and lithium-ion batteries. Chem. Soc. Rev. 2020, 49, 1569-614.
11. Goodenough, J. B.; Park, K. S. The Li-ion rechargeable battery: a perspective. J. Am. Chem. Soc. 2013, 135, 1167-76.
12. Manthiram, A. A reflection on lithium-ion battery cathode chemistry. Nat. Commun. 2020, 11, 1550.
13. Li, J.; Fleetwood, J.; Hawley, W. B.; Kays, W. From materials to cell: state-of-the-art and prospective technologies for lithium-ion battery electrode processing. Chem. Rev. 2022, 122, 903-56.
14. Li, W.; Erickson, E. M.; Manthiram, A. High-nickel layered oxide cathodes for lithium-based automotive batteries. Nat. Energy. 2020, 5, 26-34.
15. Liu, T.; Yu, L.; Lu, J.; et al. Rational design of mechanically robust Ni-rich cathode materials via concentration gradient strategy. Nat. Commun. 2021, 12, 6024.
16. Lv, Y.; Huang, S.; Zhao, Y.; et al. A review of nickel-rich layered oxide cathodes: synthetic strategies, structural characteristics, failure mechanism, improvement approaches and prospects. Appl. Energy. 2022, 305, 117849.
17. Eshetu, G. G.; Zhang, H.; Judez, X.; et al. Production of high-energy Li-ion batteries comprising silicon-containing anodes and insertion-type cathodes. Nat. Commun. 2021, 12, 5459.
18. Ikonen, T.; Kalidas, N.; Lahtinen, K.; et al. Conjugation with carbon nanotubes improves the performance of mesoporous silicon as Li-ion battery anode. Sci. Rep. 2020, 10, 5589.
19. Efaw, C. M.; Wu, Q.; Gao, N.; et al. Localized high-concentration electrolytes get more localized through micelle-like structures. Nat. Mater. 2023, 22, 1531-9.
20. Ren, F.; Li, Z.; Chen, J.; Huguet, P.; Peng, Z.; Deabate, S. Solvent-diluent interaction-mediated solvation structure of localized high-concentration electrolytes. ACS. Appl. Mater. Interfaces. 2022, 14, 4211-9.
21. Aruchamy, K.; Ramasundaram, S.; Divya, S.; Chandran, M.; Yun, K.; Oh, T. H. Gel polymer electrolytes: advancing solid-state batteries for high-performance applications. Gels 2023, 9, 585-623.
22. Mendoza AE, Schmidt A, Zarbin AJG, Winnischofer H. Review of nanoscale approaches for tailoring electrode materials for advanced energy storage systems. ACS. Appl. Nano. Mater. 2024, 7, 23295-320.
23. Qiu, J.; Duan, Y.; Li, S.; et al. Insights into nano- and micro-structured scaffolds for advanced electrochemical energy storage. Nanomicro. Lett. 2024, 16, 130.
24. Sun, H. H.; Dolocan, A.; Weeks, J. A.; Heller, A.; Mullins, C. B. Stabilization of a highly Ni-rich layered oxide cathode through flower-petal grain arrays. ACS. Nano. 2020, 14, 17142-50.
25. Schomburg, F.; Heidrich, B.; Wennemar, S.; et al. Lithium-ion battery cell formation: status and future directions towards a knowledge-based process design. Energy. Environ. Sci. 2024, 17, 2686-733.
26. Albertus, P.; Anandan, V.; Ban, C.; et al. Challenges for and pathways toward Li-metal-based all-solid-state batteries. ACS. Energy. Lett. 2021, 6, 1399-404.
27. Frith, J. T.; Lacey, M. J.; Ulissi, U. A non-academic perspective on the future of lithium-based batteries. Nat. Commun. 2023, 14, 420.
28. Xiao, J.; Cao, X.; Gridley, B.; et al. From mining to manufacturing: scientific challenges and opportunities behind battery production. Chem. Rev. 2025, 125, 6397-431.
29. Zheng, F.; Li, C.; Li, Z.; et al. Advanced composite solid electrolytes for lithium batteries: filler dimensional design and ion path optimization. Small 2023, 19, e2206355.
30. Degen, F.; Winter, M.; Bendig, D.; Tübke, J. Energy consumption of current and future production of lithium-ion and post lithium-ion battery cells. Nat. Energy. 2023, 8, 1284-95.
31. Futscher, M. H.; Brinkman, L.; Müller, A.; Casella, J.; Aribia, A.; Romanyuk, Y. E. Monolithically-stacked thin-film solid-state batteries. Commun. Chem. 2023, 6, 110.
32. Zhang, L.; Al-mamun, M.; Wang, L.; et al. The typical structural evolution of silicon anode. Cell. Rep. Phys. Sci. 2022, 3, 100811.
33. Chen, Y.; Kang, Y.; Zhao, Y.; et al. A review of lithium-ion battery safety concerns: the issues, strategies, and testing standards. J. Energy. Chem. 2021, 59, 83-99.
34. Dave, A.; Mitchell, J.; Burke, S.; Lin, H.; Whitacre, J.; Viswanathan, V. Autonomous optimization of non-aqueous Li-ion battery electrolytes via robotic experimentation and machine learning coupling. Nat. Commun. 2022, 13, 5454.
35. MacLeod, B. P.; Parlane, F. G. L.; Rupnow, C. C.; et al. A self-driving laboratory advances the Pareto front for material properties. Nat. Commun. 2022, 13, 995.
36. Xiao, Y.; Wang, Y.; Bo, S.; Kim, J. C.; Miara, L. J.; Ceder, G. Understanding interface stability in solid-state batteries. Nat. Rev. Mater. 2019, 5, 105-26.
37. Zhang, Z.; Wang, X.; Li, X.; et al. Review on composite solid electrolytes for solid-state lithium-ion batteries. Mater. Today. Sustain. 2023, 21, 100316.
38. Lu, H.; Yang, C.; Wang, F.; et al. Interfacial high-concentration electrolyte for stable lithium metal anode: theory, design, and demonstration. Nano. Res. 2022, 16, 8321-8.
39. Grey, C. P.; Hall, D. S. Prospects for lithium-ion batteries and beyond-a 2030 vision. Nat. Commun. 2020, 11, 6279.
40. Rosolen, J. M.; Decker, F. Stress in carbon film electrodes during Li+ electrochemical intercalation. J. Electrochem. Soc. 2019, 143, 2417-21.
41. Liu, X.; Sun, L.; Zhai, F.; et al. Carbon dots induced supramolecular gel polymer electrolyte for high‐performance lithium metal batteries. Adv. Energy. Mater. 2025, 15, 2405433.
42. Larcher, D.; Tarascon, J. M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 2015, 7, 19-29.
43. Cheng, X. B.; Liu, H.; Yuan, H.; et al. A perspective on sustainable energy materials for lithium batteries. SusMat 2021, 1, 38-50.
45. Shi, P.; Li, T.; Zhang, R.; et al. Lithiophilic LiC6 layers on carbon hosts enabling stable Li metal anode in working batteries. Adv. Mater. 2019, 31, e1807131.
46. Yan, K.; Lee, H. W.; Gao, T.; et al. Ultrathin two-dimensional atomic crystals as stable interfacial layer for improvement of lithium metal anode. Nano. Lett. 2014, 14, 6016-22.
47. Sanchez, A. J.; Dasgupta, N. P. Lithium metal anodes: advancing our mechanistic understanding of cycling phenomena in liquid and solid electrolytes. J. Am. Chem. Soc. 2024, 146, 4282-300.
48. Yang, S.; Cheng, Y.; Xiao, X.; Pang, H. Development and application of carbon fiber in batteries. Chem. Eng. J. 2020, 384, 123294.
49. Xu, S.; Van, Der. Watt. J.; Laudal, D.; Zhang, R.; Ahmed, R.; Hou, X. Coal-derived carbon anodes for lithium-ion batteries: development, challenges, and prospects. J. Power. Sources. 2025, 628, 235858.
50. Wei, L.; Ji, D.; Zhao, F.; Tian, X.; Guo, Y.; Yan, J. A review of carbon nanofiber materials for dendrite-free lithium-metal anodes. Molecules. 2024, 29, 4096.
51. Chen, Z.; Zhang, Q.; Liang, Q. Carbon-coatings improve performance of Li-ion battery. Nanomaterials 2022, 12, 1936.
52. Wu, Z.; Sun, K.; Wang, Z. A review of the application of carbon materials for lithium metal batteries. Batteries. 2022, 8, 246.
53. Wang, B.; Waterhouse, G. I.; Lu, S. Carbon dots: mysterious past, vibrant present, and expansive future. Trends. Chem. 2023, 5, 76-87.
54. Shaker, M.; Ng, S.; Sadeghi, Ghazvini. A. A.; et al. Carbon/graphene quantum dots as electrolyte additives for batteries and supercapacitors: a review. J. Energy. Storage. 2024, 85, 111040.
55. Cheng, R.; Xiang, Y.; Guo, R.; et al. Structure and interface modification of carbon dots for electrochemical energy application. Small 2021, 17, e2102091.
56. Song, T. B.; Huang, Z. H.; Niu, X. Q.; et al. Applications of carbon dots in next‐generation lithium-ion batteries. ChemNanoMat 2020, 6, 1421-36.
57. Wang, Z.; Che, H.; Lu, W.; et al. Application of inorganic quantum dots in advanced lithium-sulfur batteries. Adv. Sci. 2023, 10, e2301355.
58. Sead, F. F.; Makasana, J.; Saraswat, S. K.; et al. Electrochemical behavior of carbon quantum dots as electrolyte additives for enhanced battery and supercapacitor performance. Mater. Technol. 2025, 40, 2500524.
59. Lasia, A. Electrochemical impedance spectroscopy and its applications. New York, NY: Springer New York; 2014.
60. Peled, E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems-the solid electrolyte interphase model. J. Electrochem. Soc. 2019, 126, 2047-51.
61. Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 2014, 114, 11503-618.
62. Evans, J.; Vincent, C. A.; Bruce, P. G. Electrochemical measurement of transference numbers in polymer electrolytes. Polymer 1987, 28, 2324-8.
63. Choo, Y.; Halat, D. M.; Villaluenga, I.; Timachova, K.; Balsara, N. P. Diffusion and migration in polymer electrolytes. Prog. Polym. Sci. 2020, 103, 101220.
64. Kasemchainan, J.; Zekoll, S.; Spencer, Jolly. D.; et al. Critical stripping current leads to dendrite formation on plating in lithium anode solid electrolyte cells. Nat. Mater. 2019, 18, 1105-11.
65. Flatscher, F.; Philipp, M.; Ganschow, S.; Wilkening, H. M. R.; Rettenwander, D. The natural critical current density limit for Li7La3Zr2O12 garnets. J. Mater. Chem. A. 2020, 8, 15782-8.
66. Lombardo, T.; Walther, F.; Kern, C.; et al. ToF-SIMS in battery research: advantages, limitations, and best practices. J. Vac. Sci. Technol. A. 2023, 41, 053207.
67. Mozhzhukhina, N.; Flores, E.; Lundström, R.; et al. Direct operando observation of double layer charging and early solid electrolyte interphase formation in Li-ion battery electrolytes. J. Phys. Chem. Lett. 2020, 11, 4119-23.
68. Baker, S. N.; Baker, G. A. Luminescent carbon nanodots: emergent nanolights. Angew. Chem. Int. Ed. 2010, 49, 6726-44.
69. Lim, S. Y.; Shen, W.; Gao, Z. Carbon quantum dots and their applications. Chem. Soc. Rev. 2015, 44, 362-81.
70. Li, H.; Kang, Z.; Liu, Y.; Lee, S. Carbon nanodots: synthesis, properties and applications. J. Mater. Chem. 2012, 22, 24230.
71. Cui, L.; Ren, X.; Sun, M.; Liu, H.; Xia, L. Carbon dots: synthesis, properties and applications. Nanomaterials 2021, 11, 3419.
72. Rocco, D.; Moldoveanu, V. G.; Feroci, M.; Bortolami, M.; Vetica, F. Electrochemical synthesis of carbon quantum dots. ChemElectroChem 2023, 10, e202201104.
73. Song, Y.; Zhu, S.; Zhang, S.; et al. Investigation from chemical structure to photoluminescent mechanism: a type of carbon dots from the pyrolysis of citric acid and an amine. J. Mater. Chem. C. 2015, 3, 5976-84.
74. Guo, H.; Lu, Y.; Lei, Z.; et al. Machine learning-guided realization of full-color high-quantum-yield carbon quantum dots. Nat. Commun. 2024, 15, 4843.
75. Li, L.; Li, Y.; Ye, Y.; et al. Kilogram-scale synthesis and functionalization of carbon dots for superior electrochemical potassium storage. ACS. Nano. 2021, 15, 6872-85.
76. Spotte-smith, E. W. C.; Petrocelli, T. B.; Patel, H. D.; Blau, S. M.; Persson, K. A. Elementary decomposition mechanisms of lithium hexafluorophosphate in battery electrolytes and interphases. ACS. Energy. Lett. 2022, 8, 347-55.
77. Yi, M.; Jing, M.; Yang, Y.; et al. Recent developments of carbon dots for advanced zinc‐based batteries: a review. Adv. Funct. Mater. 2024, 34, 2400001.
78. Yang, S.; Ye, X.; Shen, N.; et al. Carbon dots enabling high-performances sodium-ion batteries. Chemistry 2025, 31, e202402794.
79. Li, L.; Jia, X.; Zhang, Y.; et al. Li4Ti5O12 quantum dot decorated carbon frameworks from carbon dots for fast lithium ion storage. Mater. Chem. Front. 2019, 3, 1761-7.
80. Ge, G.; Li, L.; Wang, D.; et al. Carbon dots: synthesis, properties and biomedical applications. J. Mater. Chem. B. 2021, 9, 6553-75.
81. Ullal, N.; Mehta, R.; Sunil, D. Separation and purification of fluorescent carbon dots - an unmet challenge. Analyst 2024, 149, 1680-700.
82. Alafeef, M.; Srivastava, I.; Aditya, T.; Pan, D. Carbon dots: from synthesis to unraveling the fluorescence mechanism. Small 2024, 20, e2303937.
83. Arora, G.; Sabran, N. S.; Ng, C. Y.; Low, F. W.; Jun, H. K. Applications of carbon quantum dots in electrochemical energy storage devices. Heliyon 2024, 10, e35543.
84. Cai, D.; Zhong, X.; Xu, L.; et al. Biomass-derived carbon dots: synthesis, modification and applications. Chem. Sci. 2025, 16, 4937-70.
85. Crista, D. M. A.; Esteves, da. Silva. J. C. G.; Pinto, da. Silva. L. Evaluation of different bottom-up routes for the fabrication of carbon dots. Nanomaterials 2020, 10, 1316.
86. Rigodanza, F.; Burian, M.; Arcudi, F.; Đorđević, L.; Amenitsch, H.; Prato, M. Snapshots into carbon dots formation through a combined spectroscopic approach. Nat. Commun. 2021, 12, 2640.
87. Tu, H.; Liu, H.; Xu, L.; et al. Carbon dots from alcohol molecules: principles and the reaction mechanism. Chem. Sci. 2023, 14, 12194-204.
88. Choi, Y.; Choi, Y.; Kwon, O. H.; Kim, B. S. Carbon dots: bottom-up syntheses, properties, and light-harvesting applications. Chem. Asian. J. 2018, 13, 586-98.
89. Miao, S.; Liang, K.; Zhu, J.; Yang, B.; Zhao, D.; Kong, B. Hetero-atom-doped carbon dots: doping strategies, properties and applications. Nano. Today. 2020, 33, 100879.
90. Xia, C.; Zhong, J.; Han, X.; et al. The formation mechanism of carbonized polymer dots: crosslinking-induced nucleation and carbonization. Angew. Chem. Int. Ed. 2024, 63, e202410519.
91. Li, Z.; Liu, F.; Chen, S.; et al. Single Li ion conducting solid-state polymer electrolytes based on carbon quantum dots for Li-metal batteries. Nano. Energy. 2021, 82, 105698.
92. El-azazy, M.; Osman, A. I.; Nasr, M.; et al. The interface of machine learning and carbon quantum dots: from coordinated innovative synthesis to practical application in water control and electrochemistry. Coord. Chem. Rev. 2024, 517, 215976.
93. Hatimuria, M.; Phukan, P.; Bag, S.; et al. Green carbon dots: applications in development of electrochemical sensors, assessment of toxicity as well as anticancer properties. Catalysts 2023, 13, 537.
94. Tang, Y.; Xu, Q.; Zhang, X.; Zhu, R.; Zhao, N.; Wang, J. Expediting carbon dots synthesis by the active adaptive method with machine learning and applications in dental diagnosis and treatment. Nano. Res. 2024, 17, 10109-18.
95. Senanayake, R. D.; Yao, X.; Froehlich, C. E.; et al. Machine learning-assisted carbon dot synthesis: prediction of emission color and wavelength. J. Chem. Inf. Model. 2022, 62, 5918-28.
96. Burger, B.; Maffettone, P. M.; Gusev, V. V.; et al. A mobile robotic chemist. Nature 2020, 583, 237-41.
97. Szymanski, N. J.; Rendy, B.; Fei, Y.; et al. An autonomous laboratory for the accelerated synthesis of novel materials. Nature 2023, 624, 86-91.
98. Tom, G.; Schmid, S. P.; Baird, S. G.; et al. Self-driving laboratories for chemistry and materials science. Chem. Rev. 2024, 124, 9633-732.
99. Xu, Q.; Tang, Y.; Zhu, P.; et al. Machine learning guided microwave-assisted quantum dot synthesis and an indication of residual H2O2 in human teeth. Nanoscale 2022, 14, 13771-8.
100. Han, Y.; Tang, B.; Wang, L.; et al. Machine-learning-driven synthesis of carbon dots with enhanced quantum yields. ACS. Nano. 2020, 14, 14761-8.
101. Zhang, Y.; Yue, L.; Ding, H.; et al. Carbon dots promoting surface defect and interphase high anion concentration for sodium-ion battery carbon anodes. Nano. Energy. 2024, 127, 109696.
102. Dua, S.; Kumar, P.; Pani, B.; Kaur, A.; Khanna, M.; Bhatt, G. Stability of carbon quantum dots: a critical review. RSC. Adv. 2023, 13, 13845-61.
103. Tu, H.; Zhang, Y.; Wu, J.; et al. Revealing electrochemical process of functional carbon dots stabilized sodium metal anode: co-deposition and strengthened SEI films. Adv. Funct. Mater. 2024, 35, 2413488.
104. Chabu, J. M.; Zeng, K.; Jin, G.; Zhang, M.; Li, Y.; Liu, Y. Simple approach for the preparation of nitrogen and sulfur codoped carbon dots/reduced graphene oxide as host for high-rate lithium sulfur batteries. Mater. Chem. Phys. 2019, 229, 226-31.
105. Chao, D.; Zhu, C.; Xia, X.; et al. Graphene quantum dots coated VO2 arrays for highly durable electrodes for Li and Na ion batteries. Nano. Lett. 2015, 15, 565-73.
106. Wang, J.; Zhang, X.; He, W.; Yue, Y.; Wang, Y.; Zhang, C. Layered hybrid phase Li2NaV2(PO4)3/carbon dot nanocomposite cathodes for Li+/Na+ mixed-ion batteries. RSC. Adv. 2017, 7, 2658-66.
107. Kim, J.; Song, S.; Lee, C. S.; Lee, M.; Bae, J. Prominent enhancement of stability under high current density of LiFePO4-based multidimensional nanocarbon composite as cathode for lithium-ion batteries. J. Colloid. Interface. Sci. 2023, 650, 1958-65.
108. Zhang, Q.; Sun, C.; Fan, L.; Zhang, N.; Sun, K. Iron fluoride vertical nanosheets array modified with graphene quantum dots as long-life cathode for lithium ion batteries. Chem. Eng. J. 2019, 371, 245-51.
109. Balogun, M. S.; Luo, Y.; Lyu, F.; et al. Carbon quantum dot surface-engineered VO2 interwoven nanowires: a flexible cathode material for lithium and sodium ion batteries. ACS. Appl. Mater. Interfaces. 2016, 8, 9733-44.
110. Fu, L.; Liu, D.; Zuo, X.; Qiu, Y.; You, T.; Zhang, Y. The key role of N/S codoped carbon dots in efficient capture and conversion of lithium polysulfides. Small 2025, 21, e2406714.
111. Xu, J.; Zhou, P.; Yuan, L.; Liu, X.; Ma, J.; Zhang, C. Dual lignin valorization enabled by carbon quantum dots and lithium-sulfur cathode. Ind. Crops. Prod. 2021, 170, 113801.
112. Yang, Y.; Zhu, J.; Wang, P.; et al. NH2-MIL-125 (Ti) derived flower-like fine TiO2 nanoparticles implanted in N-doped porous carbon as an anode with high activity and long cycle life for lithium-ion batteries. Acta. Phys. Chim. Sin. 2021, 38, 2106002.
113. Lijuan, K.; Yongqiang, Y.; Ruiyi, L.; Zaijun, L. Phenylalanine-functionalized graphene quantum dot-silicon nanoparticle composite as an anode material for lithium ion batteries with largely enhanced electrochemical performance. Electrochim. Acta. 2016, 198, 144-55.
114. Nan, H.; Zhang, Y.; Wei, H.; et al. Low-cost and environmentally friendly synthesis of an Al3+ and Mn4+ co-doped Li4Ti5O12 composite with carbon quantum dots as an anode for lithium-ion batteries. RSC. Adv. 2019, 9, 22101-5.
115. Sun, X.; Gao, X.; Li, Z.; et al. Nanowires framework supported porous lotus-carbon anode boosts lithium-ion and sodium-ion batteries. Small. Methods. 2024, 8, e2300746.
116. Kim, K. H.; Ahn, H. J. Surface functional group‐tailored B and N co‐doped carbon quantum dot anode for lithium‐ion batteries. Intl. J. Energy. Res. 2022, 46, 8367-75.
117. Khan, F.; Oh, M.; Kim, J. H. N-functionalized graphene quantum dots: charge transporting layer for high-rate and durable Li4Ti5O12-based Li-ion battery. Chem. Eng. J. 2019, 369, 1024-33.
118. Xiang, Y.; Xu, L.; Yang, L.; et al. Natural stibnite for lithium-/sodium-ion batteries: carbon dots evoked high initial coulombic efficiency. Nanomicro. Lett. 2022, 14, 136.
119. Kim, J.; Jang, W.; Kim, J. H.; Yang, C. Synthesis of graphene quantum dots-coated hierarchical CuO microspheres composite for use as binder-free anode for lithium-ion batteries. Compos. Part. B. Eng. 2021, 222, 109083.
120. Lee, H. R.; Kim, Y. S.; Lee, S. Y.; Son, U. H.; Lee, S.; Joh, H. Bifunctional effects of nitrogen-doped carbon quantum dots on CoS2/mesoporous carbon composites for high-performance lithium-ion batteries. Appl. Surf. Sci. 2024, 664, 160228.
121. Gottam, R.; Zhang, X.; Chen, J.; Xue, X.; Zhang, Y. Sulfur‐implanted carbon dots‐embedded graphene as ultrastable anode for Li‐ion batteries. Energy. Technol. 2021, 9, 2000899.
122. Wang, A.; Hong, W.; Li, L.; et al. Hierarchical bismuth composite for fast lithium storage: carbon dots tuned interfacial interaction. Energy. Storage. Mater. 2022, 44, 145-55.
123. Kong, L.; Zhou, X.; Fan, S.; Li, Z.; Gu, Z. Study on the synthesis and electrochemical performance of histidine-functionalized graphene quantum dots@silicon composite anode material. Acta. Chim. Sin. 2016, 74, 620.
124. Du, Q.; Wu, Q.; Wang, H.; et al. Carbon dot-modified silicon nanoparticles for lithium-ion batteries. Int. J. Miner. Metall. Mater. 2021, 28, 1603-10.
125. Hwang, S. W. SiOx/C composite anode for lithium-ion battery with improved performance using graphene quantum dots and carbon nanoparticles. Molecules 2024, 29, 2578.
126. Feng, G.; Shi, Y.; Jia, H.; et al. Progressive and instantaneous nature of lithium nucleation discovered by dynamic and operando imaging. Sci. Adv. 2023, 9, eadg6813.
127. Shen, H.; Song, C.; Wang, F.; Li, G.; Li, Y. Lithiated graphdiyne quantum dots for stable lithium metal anodes. CCS. Chem. 2024, 6, 1300-11.
128. Cheng, X. B.; Zhang, R.; Zhao, C. Z.; Zhang, Q. Toward safe lithium metal anode in rechargeable batteries: a review. Chem. Rev. 2017, 117, 10403-73.
129. Wang, A.; Kadam, S.; Li, H.; Shi, S.; Qi, Y. Review on modeling of the anode solid electrolyte interphase (SEI) for lithium-ion batteries. NPJ. Comput. Mater. 2018, 4, 15.
130. Zhu, Y.; Ge, M.; Ma, F.; Wang, Q.; Huang, P.; Lai, C. Multifunctional electrolyte additives for better metal batteries. Adv. Funct. Mater. 2023, 34, 2301964.
131. Wang, G.; Xiong, X.; Xie, D.; et al. Suppressing dendrite growth by a functional electrolyte additive for robust Li metal anodes. Energy. Storage. Mater. 2019, 23, 701-6.
132. Hong, D.; Choi, Y.; Ryu, J.; et al. Homogeneous Li deposition through the control of carbon dot-assisted Li-dendrite morphology for high-performance Li-metal batteries. J. Mater. Chem. A. 2019, 7, 20325-34.
133. Li, S.; Luo, Z.; Tu, H.; et al. N,S-codoped carbon dots as deposition regulating electrolyte additive for stable lithium metal anode. Energy. Storage. Mater. 2021, 42, 679-86.
134. Wang, W. C.; Song, Y. H.; Yang, G. D.; et al. Carbonized polymer dots with controllable N, O functional groups as electrolyte additives to achieve stable Li metal batteries. Small 2023, 19, e2206597.
135. Kushima, A.; So, K. P.; Su, C.; et al. Liquid cell transmission electron microscopy observation of lithium metal growth and dissolution: root growth, dead lithium and lithium flotsams. Nano. Energy. 2017, 32, 271-9.
136. Kar, D. K.; V, P.; Si, S.; Panigrahi, H.; Mishra, S. Carbon dots and their polymeric nanocomposites: insight into their synthesis, photoluminescence mechanisms, and recent trends in sensing applications. ACS. Omega. 2024, 9, 11050-80.
137. Qian, Y.; Liang, Y.; Zhang, W.; Xi, B.; Lin, N. Thermal polymerization of ion-modified carbon dots into multi-functional LiF-carbon interface for stabilizing SiO anode. Energy. Storage. Mater. 2023, 63, 102996.
138. Wang, Z.; Liang, S.; Kang, Y.; et al. Manipulating interfacial polymerization for polymeric nanofilms of composite separation membranes. Prog. Polym. Sci. 2021, 122, 101450.
139. Najafloo, M.; Naji, L. Sustainable self-healing gel polymer electrolyte based on water-in-deep eutectic solvent for flexible supercapacitors. ACS. Appl. Polym. Mater. 2024, 6, 11706-21.
140. Gannoun, R.; Durán-olivencia, F.; Pérez, A.; Valverde, J. Titania coatings: a mechanical shield for cohesive granular media at high temperatures. Chem. Eng. J. 2022, 450, 138123.
141. Liu, H.; Xu, L.; Zhu, F.; et al. Unveiling the effect law of carbon dots with polyfunctional groups on interface structure and ion migration in polymer electrolytes for solid lithium battery. Nano. Energy. 2024, 126, 109623.
142. Huang, Z. H.; Wei, J. S.; Song, T. B.; Ni, J. W.; Wang, F.; Xiong, H. M. Carbon dots crosslinked gel polymer electrolytes for dendrite‐free and long‐cycle lithium metal batteries. SmartMat 2022, 3, 323-36.
143. Xu, L.; Li, S.; Tu, H.; et al. Molecular engineering of highly fluorinated carbon dots: tailoring Li+ dynamics and interfacial fluorination for stable solid lithium batteries. ACS. Nano. 2023, 17, 22082-94.
144. Zhang, Z.; Li, Y.; Xu, R.; et al. Capturing the swelling of solid-electrolyte interphase in lithium metal batteries. Science 2022, 375, 66-70.
145. Liu, H.; Zhu, F.; Zhang, Y.; et al. Synergistic regulation of multi-interface chemistry by functional carbon dots for high-performance composite solid electrolytes. Angew. Chem. Int. Ed. 2025, 64, e202505230.
146. Shadike, Z.; Lee, H.; Borodin, O.; et al. Identification of LiH and nanocrystalline LiF in the solid-electrolyte interphase of lithium metal anodes. Nat. Nanotechnol. 2021, 16, 549-54.
147. Zhu, F.; Xu, L.; Hu, X.; et al. Trace fluorinated carbon dots driven Li-garnet solid-state batteries. Angew. Chem. Int. Ed. 2024, 63, e202410016.
148. Zhu, F.; Liu, H.; Zhang, B.; et al. Tailoring electron-blocking and ionic-transport interface via functional carbon dots for durable garnet-based solid-state batteries. Adv. Funct. Mater. 2025, 35, 2507998.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






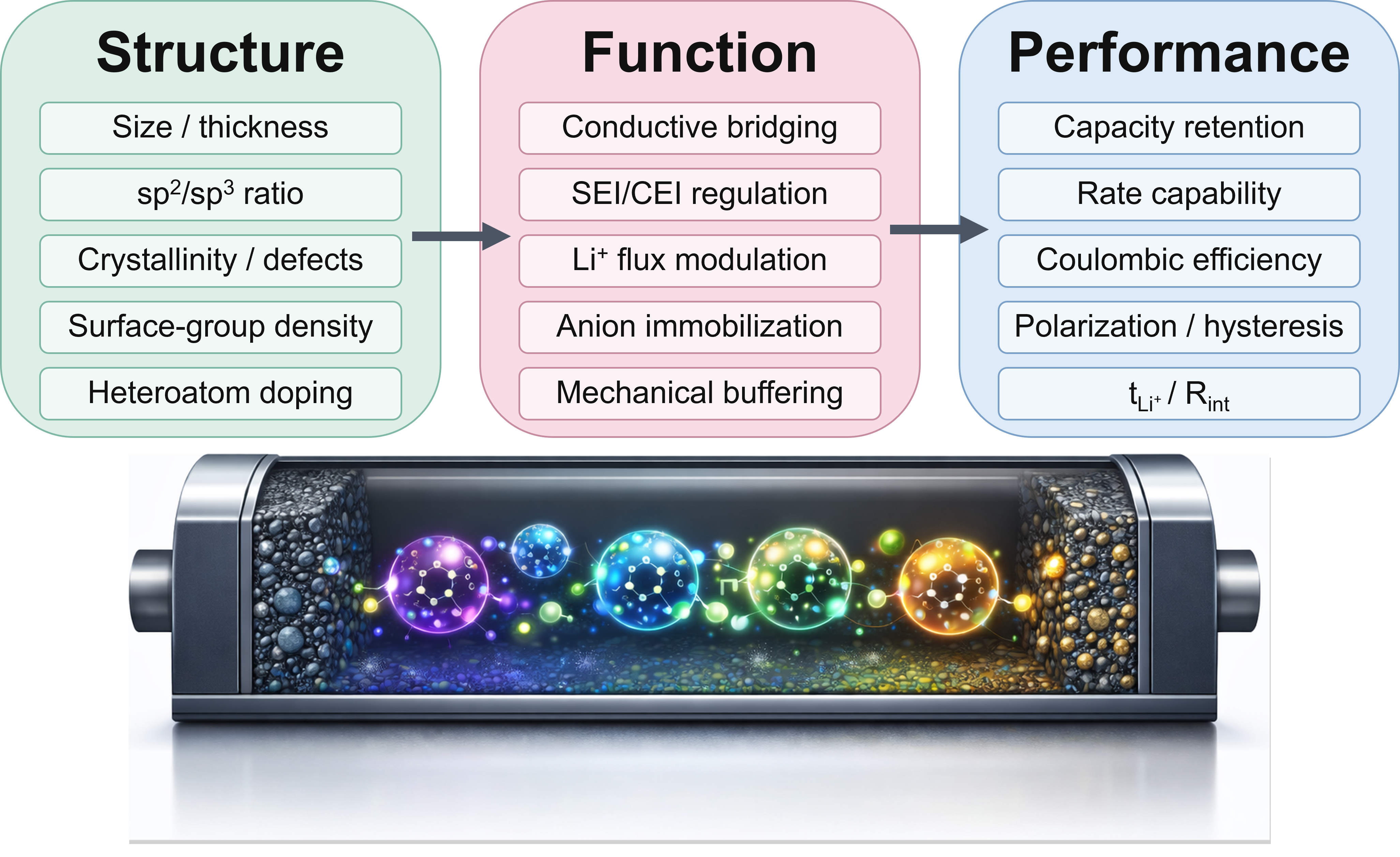
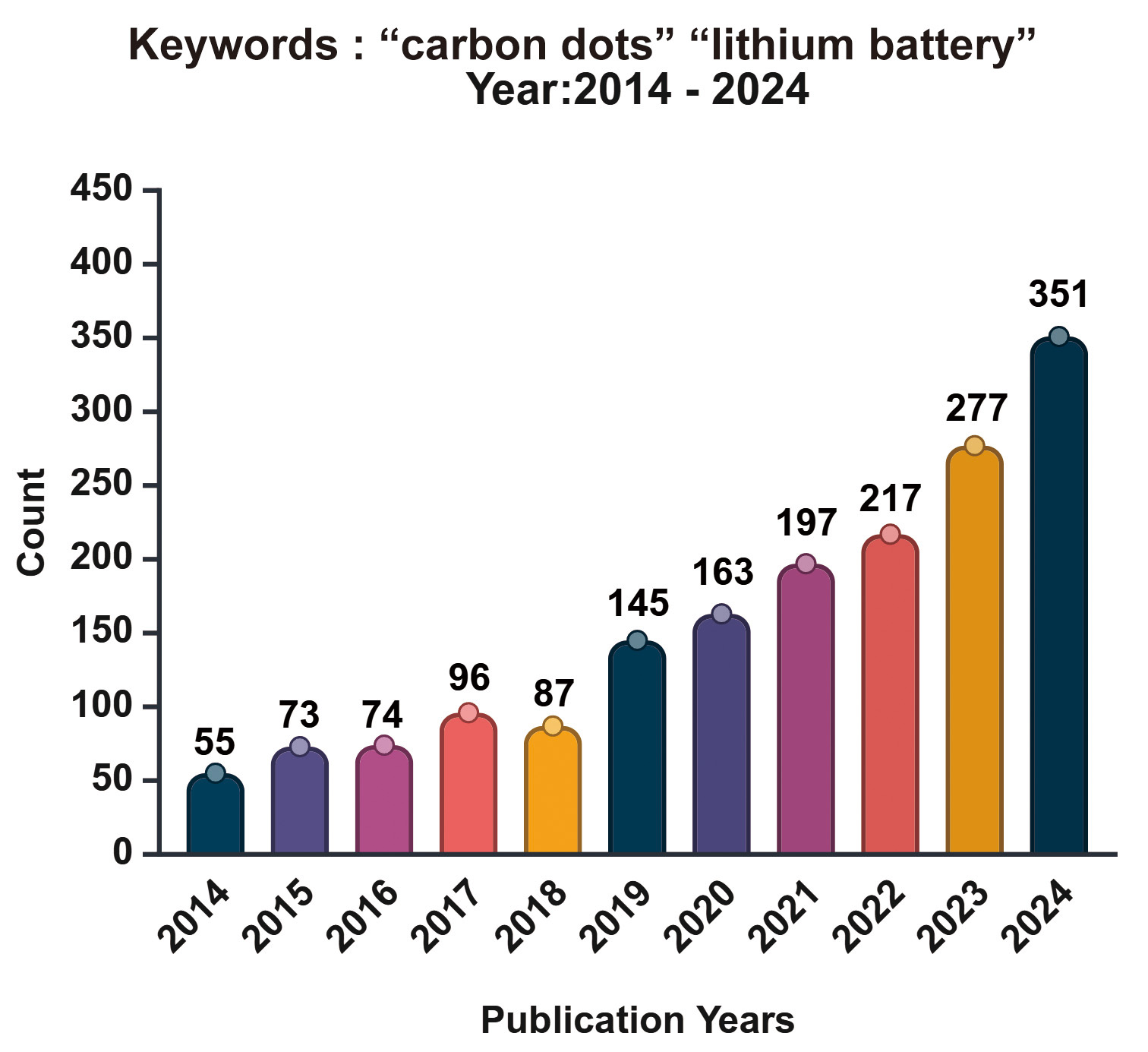
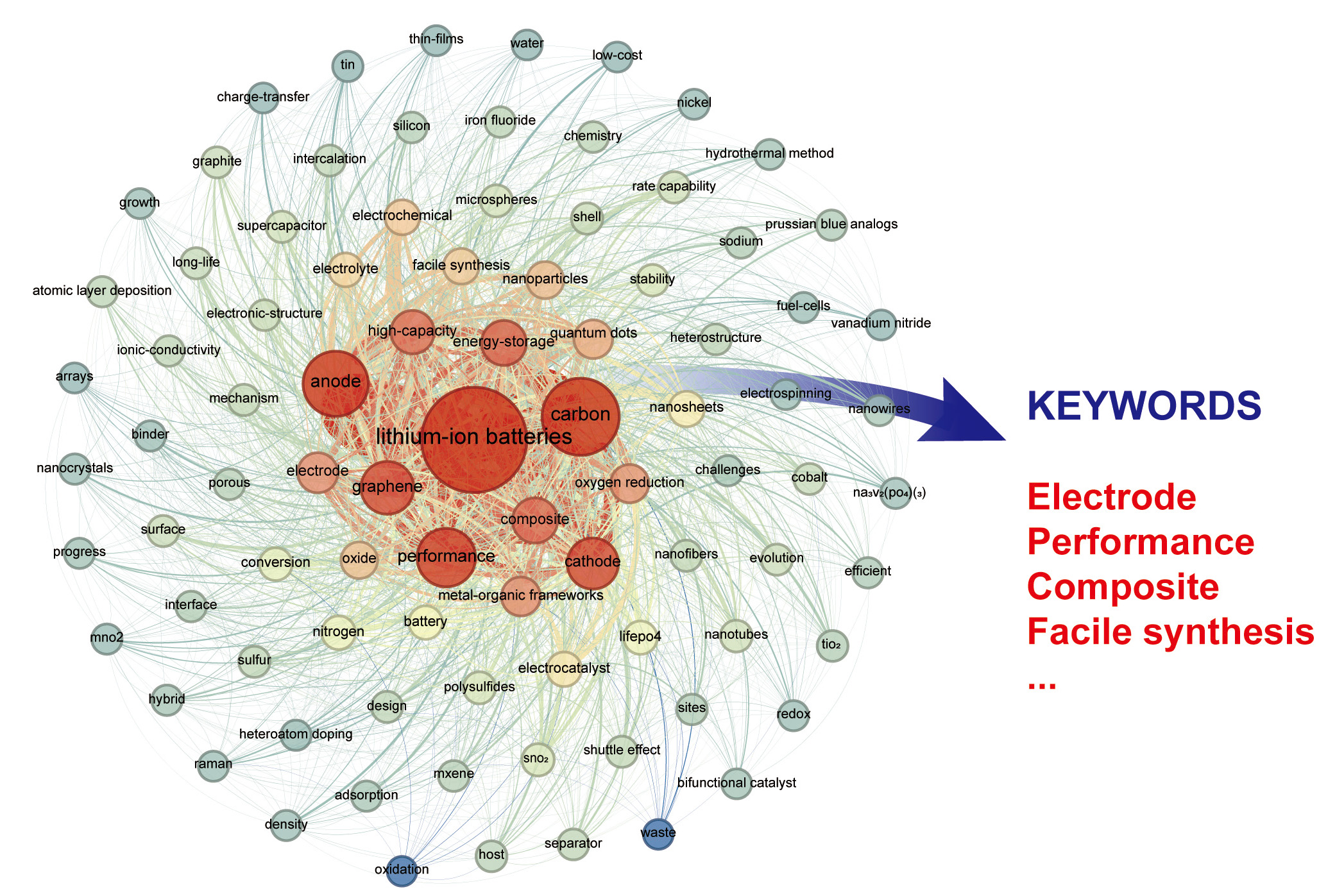
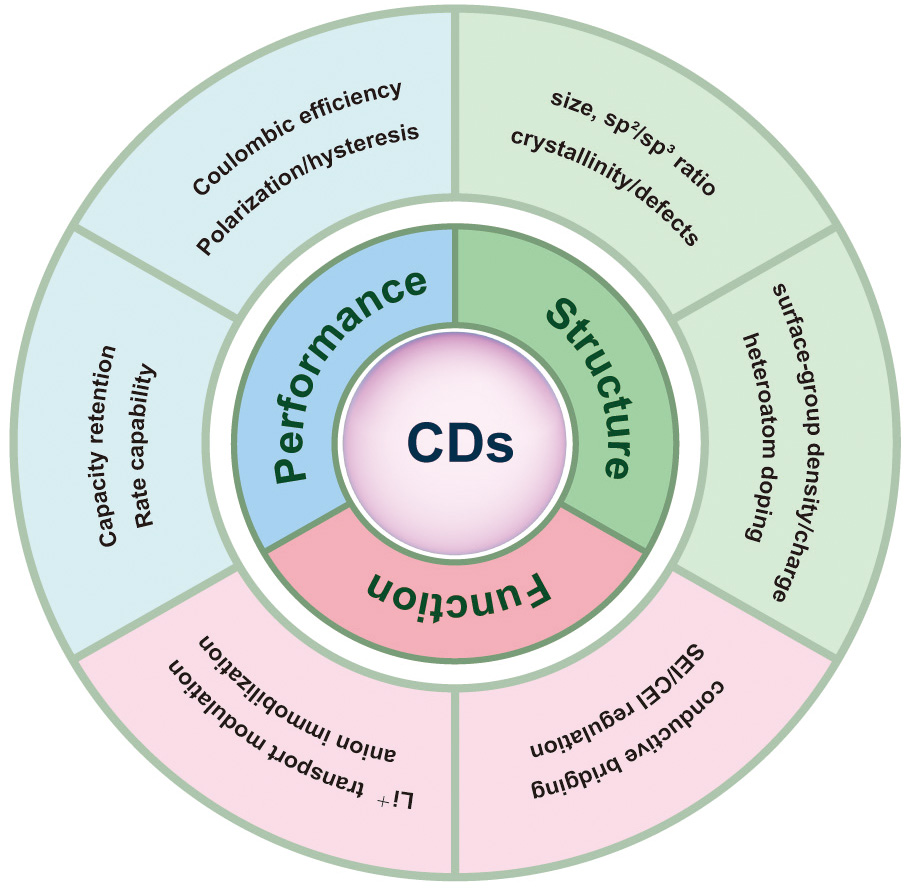
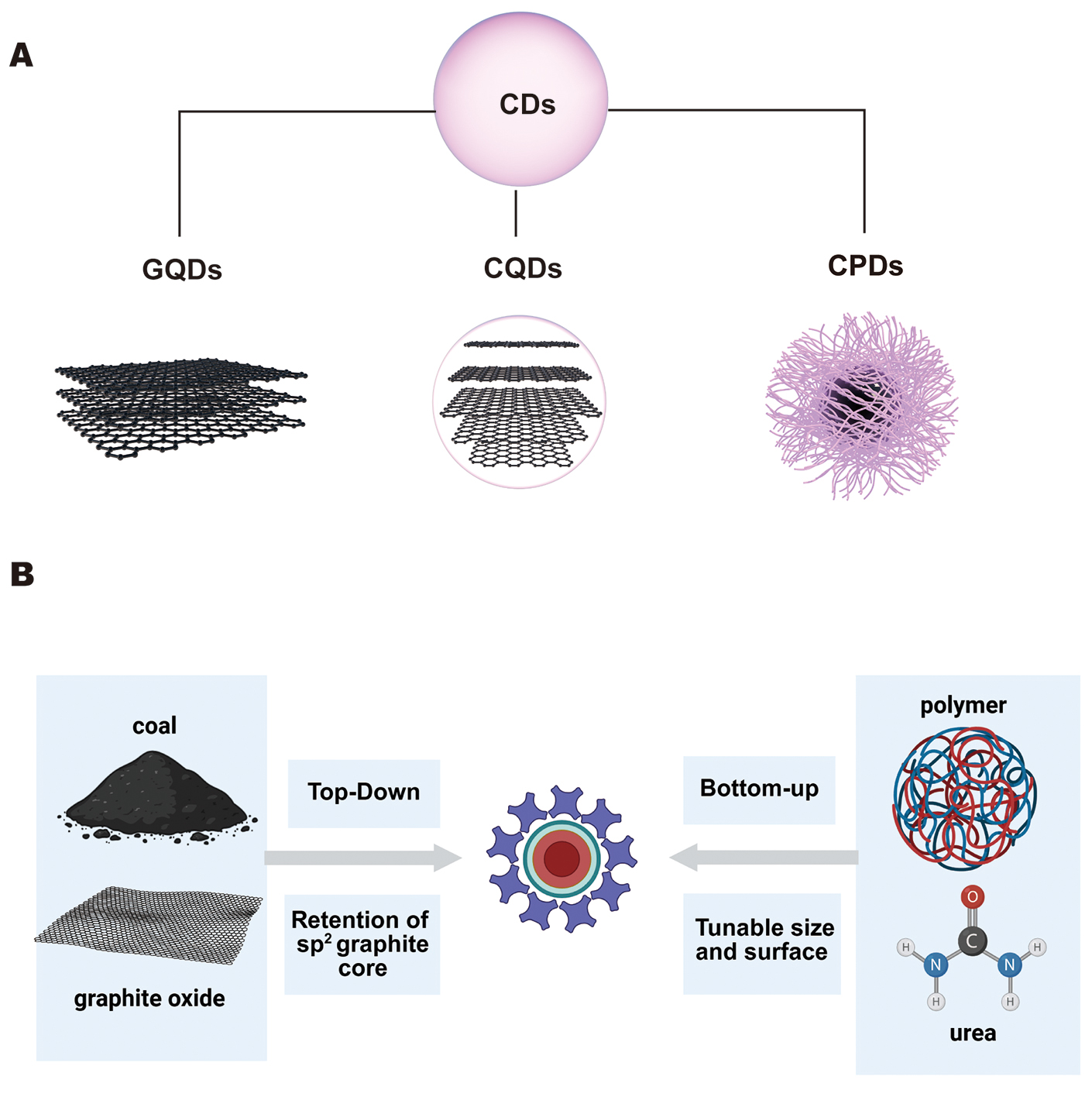
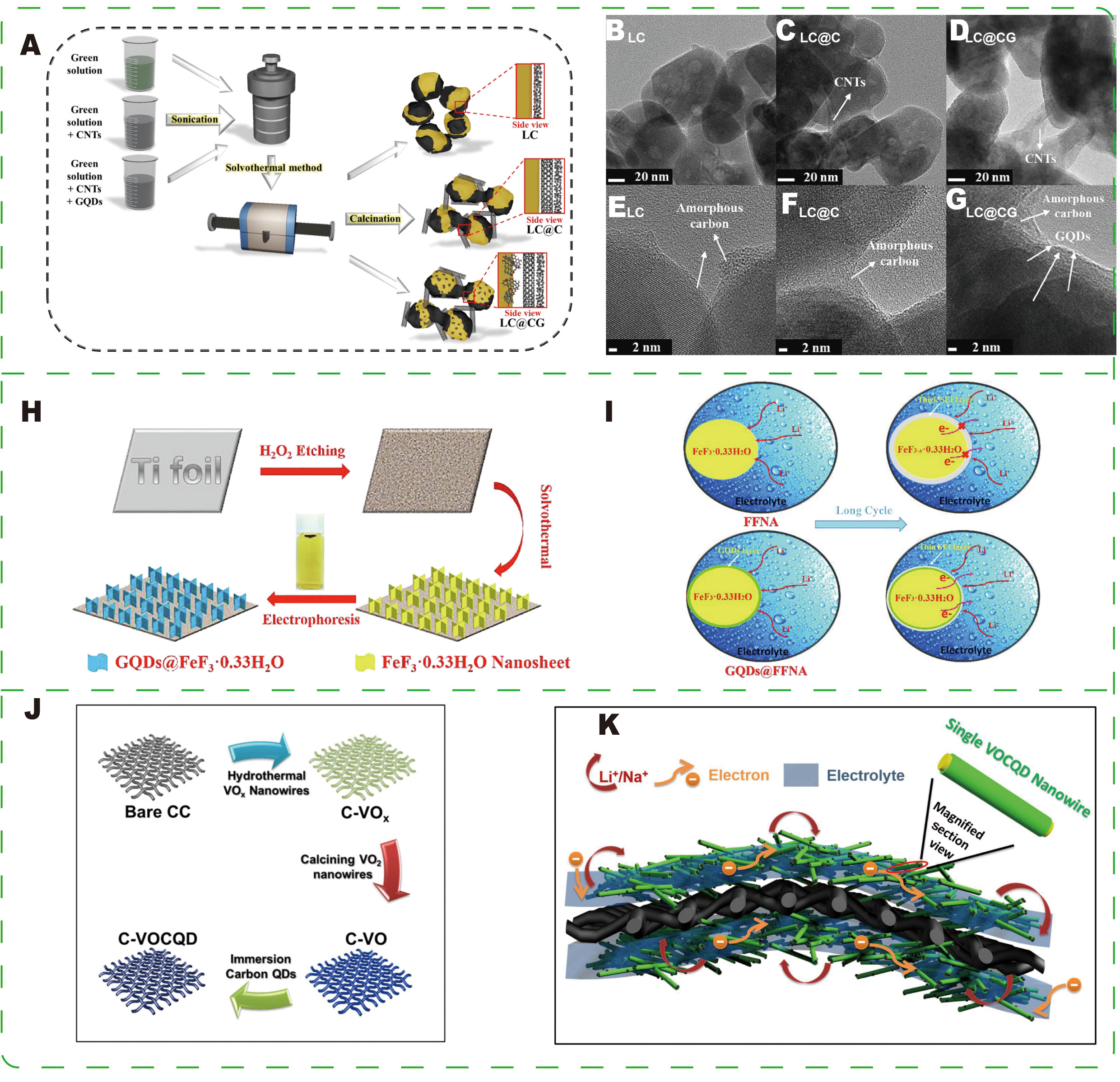
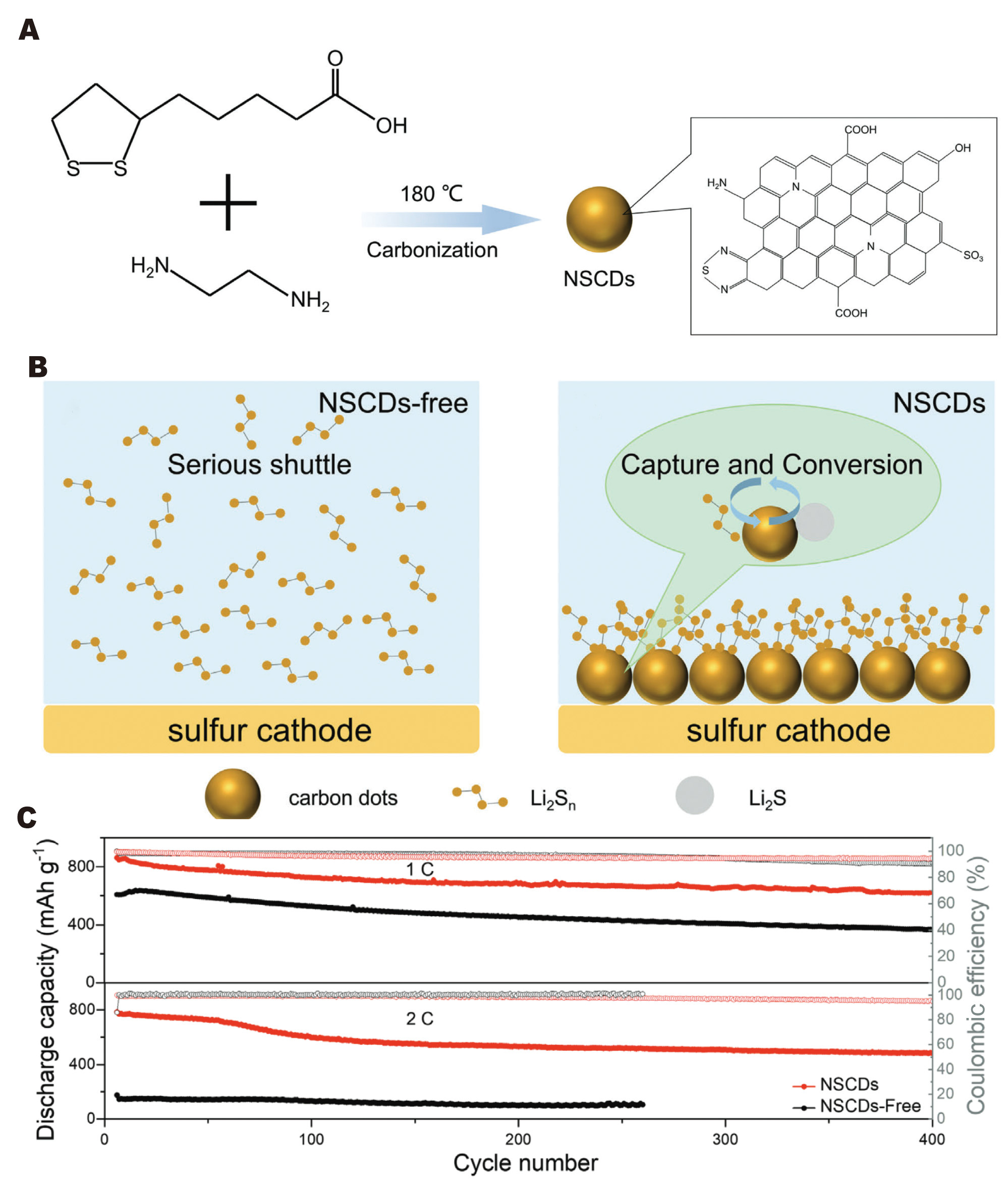
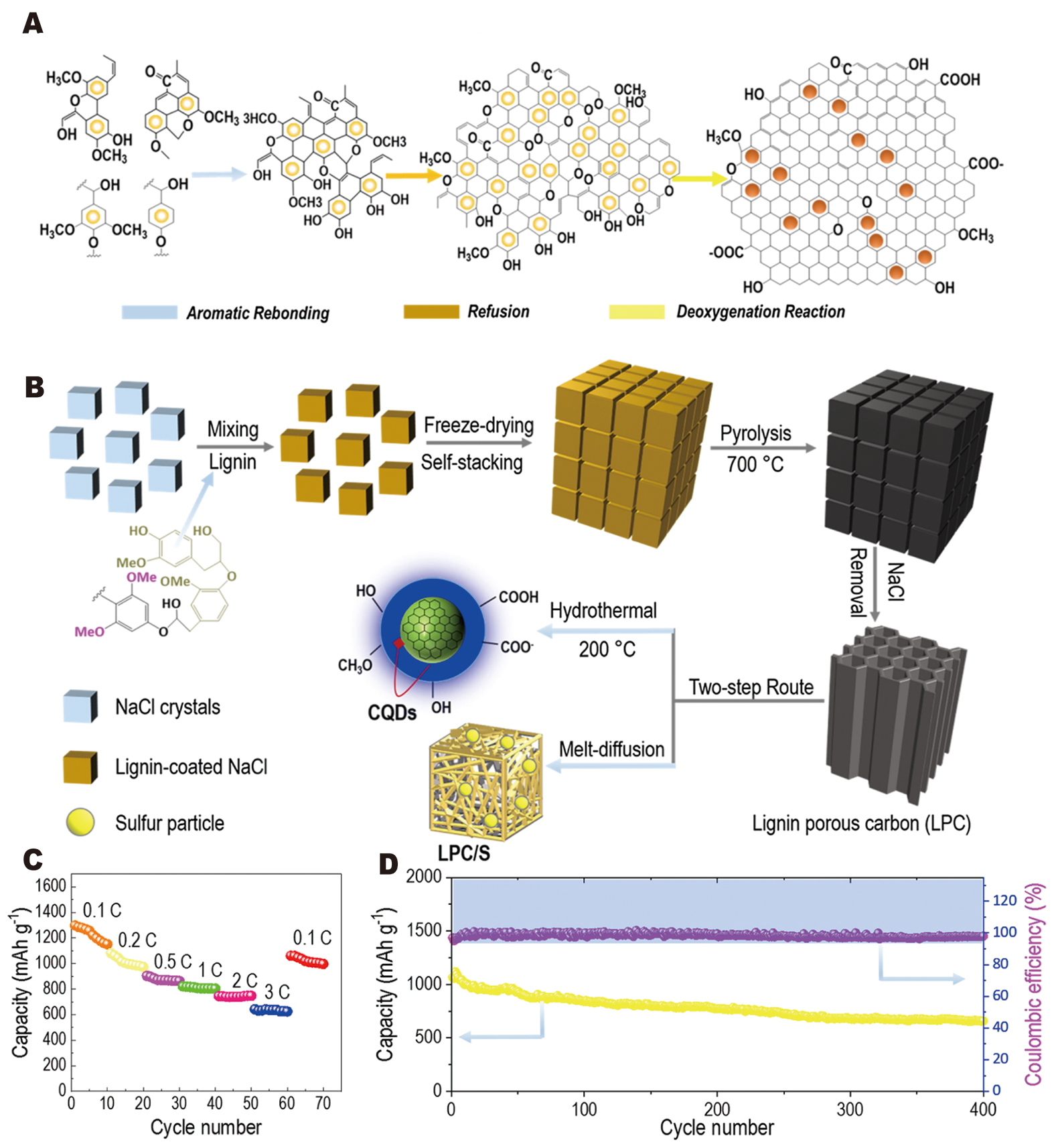
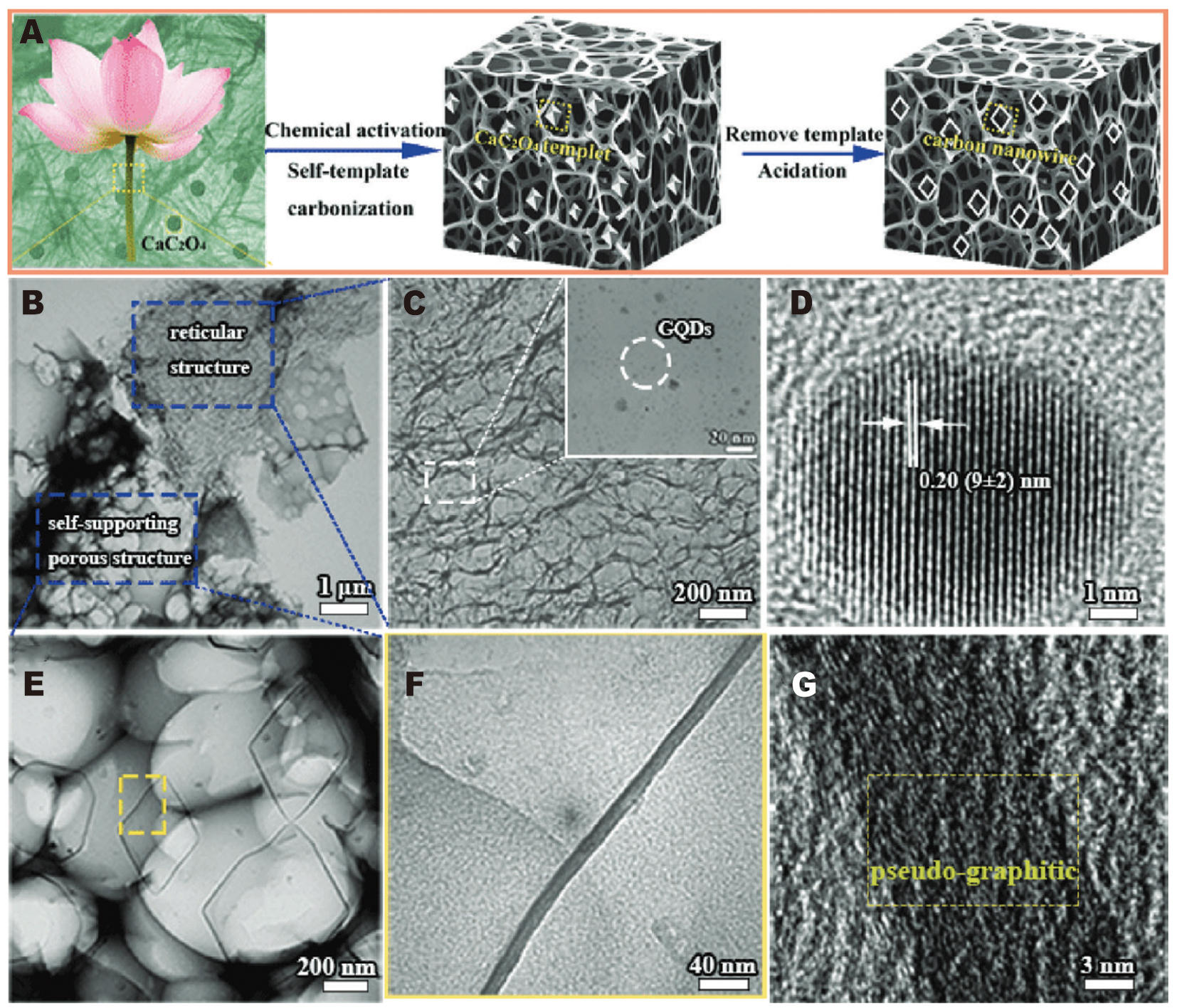
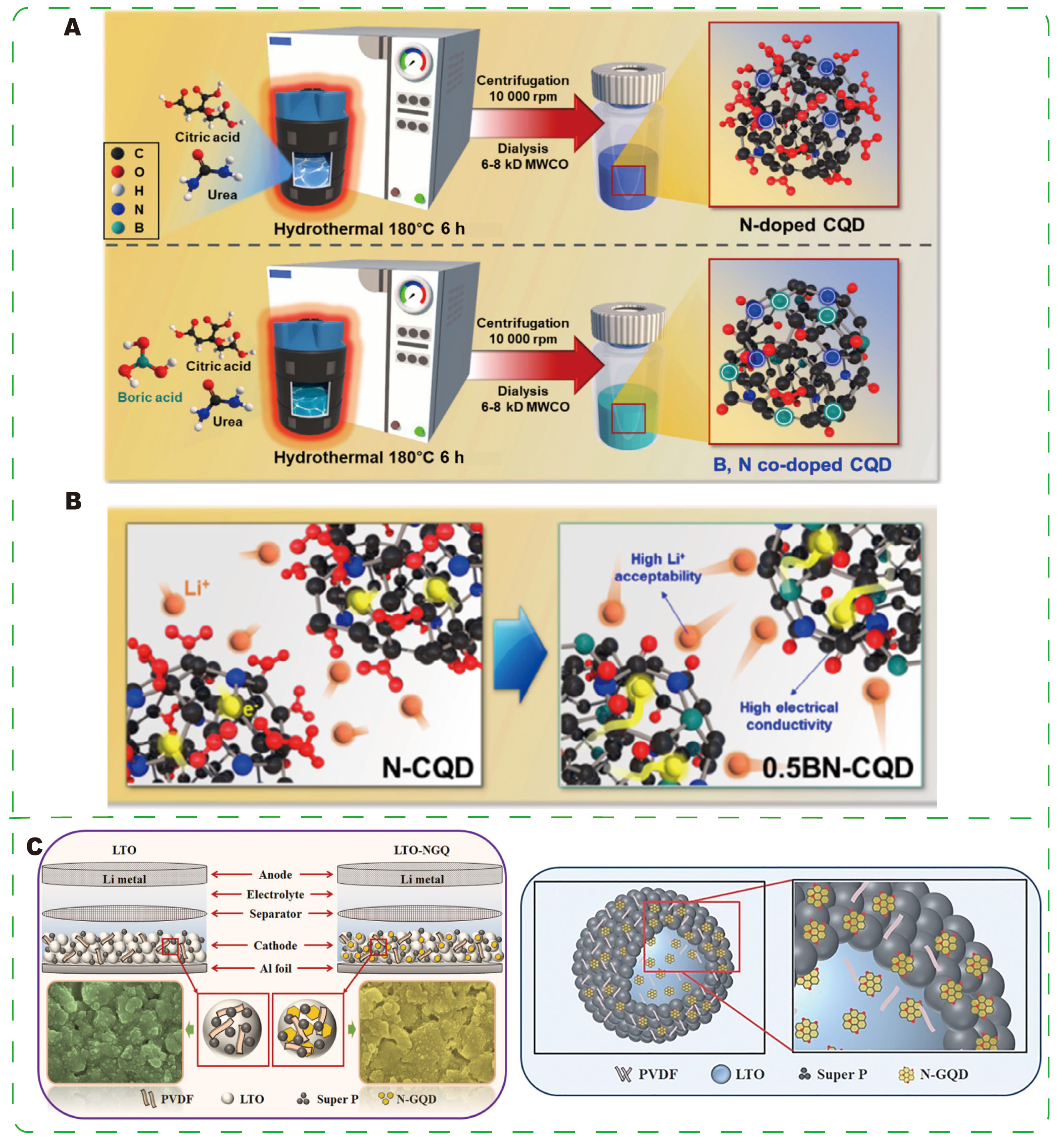
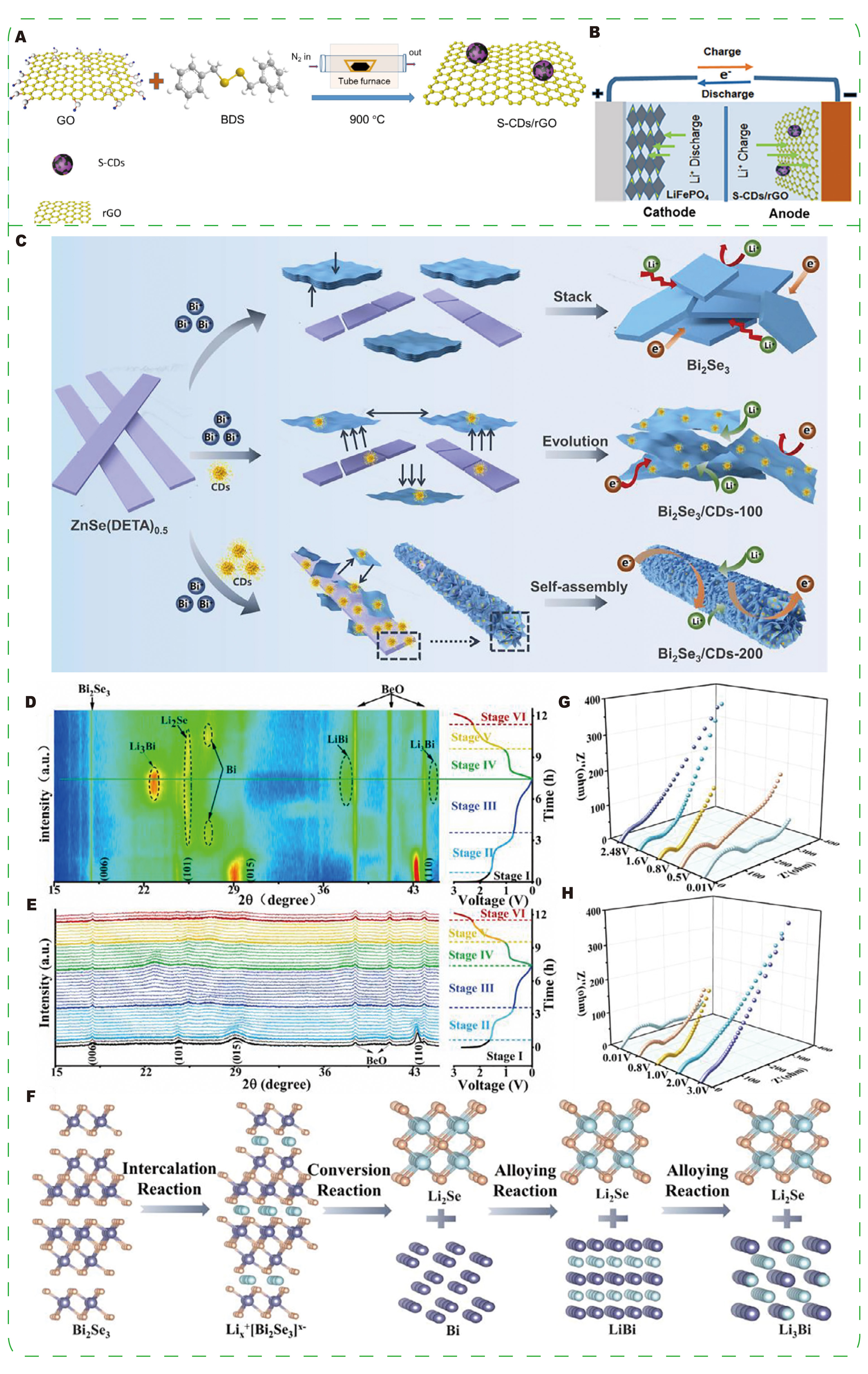

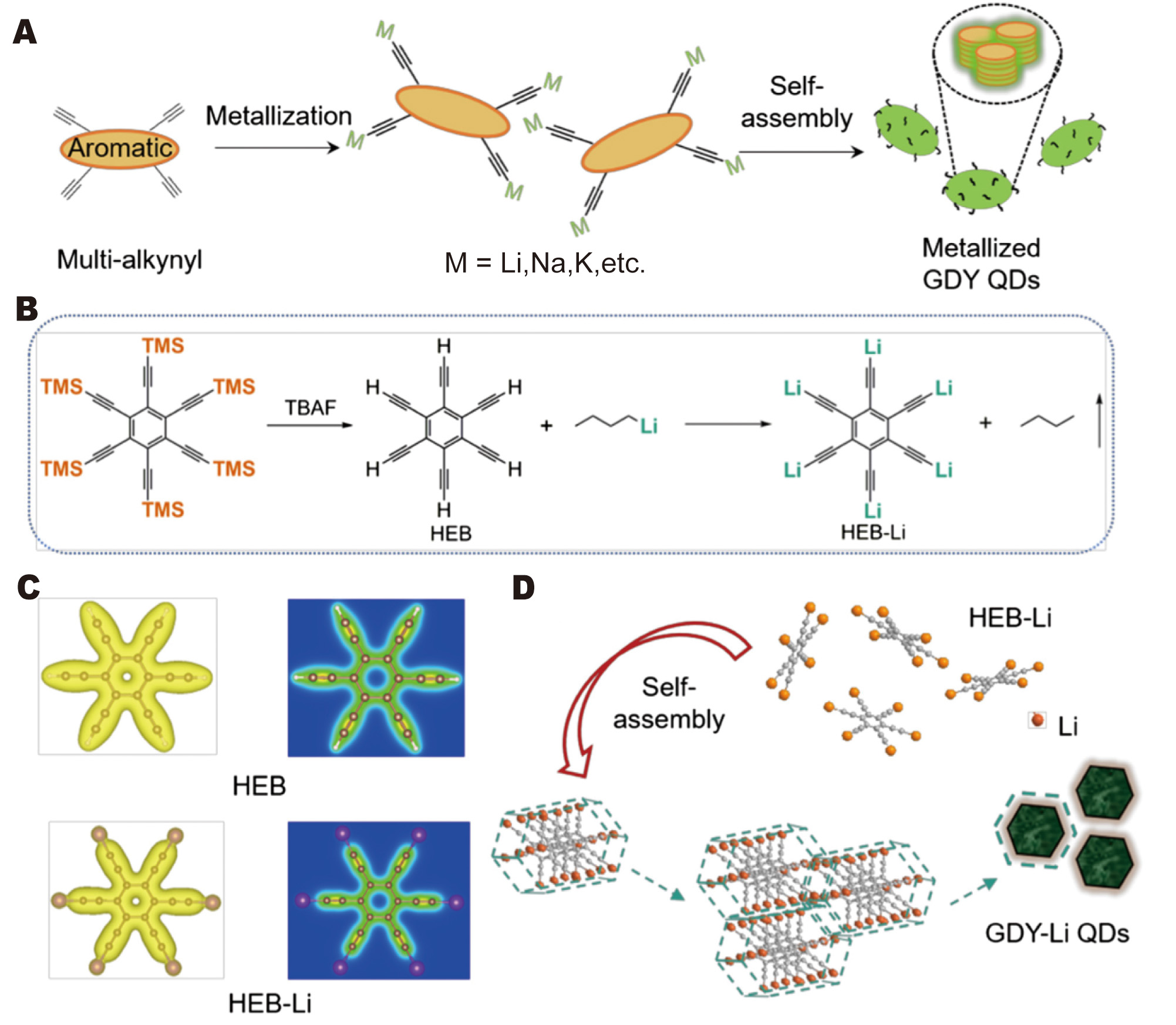
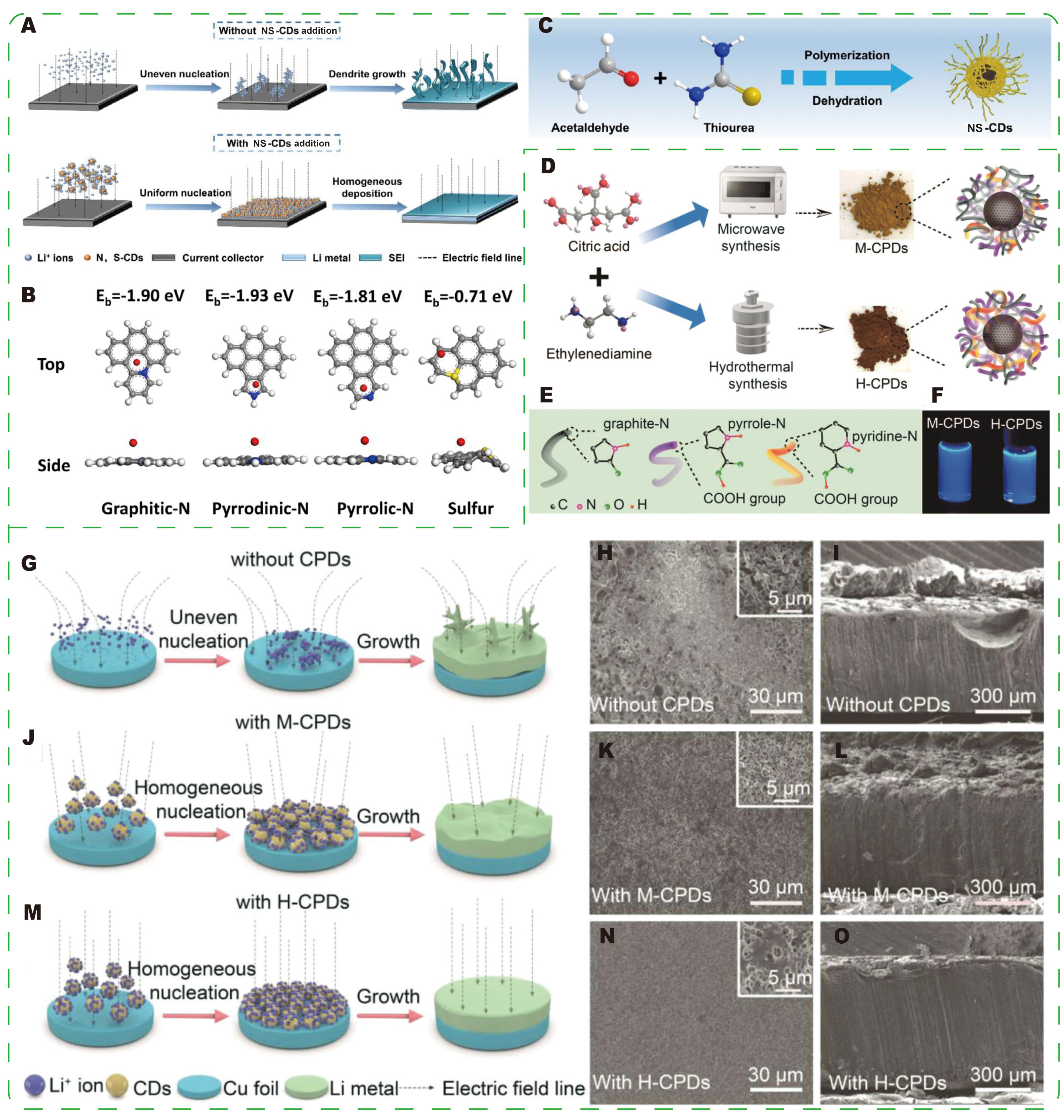
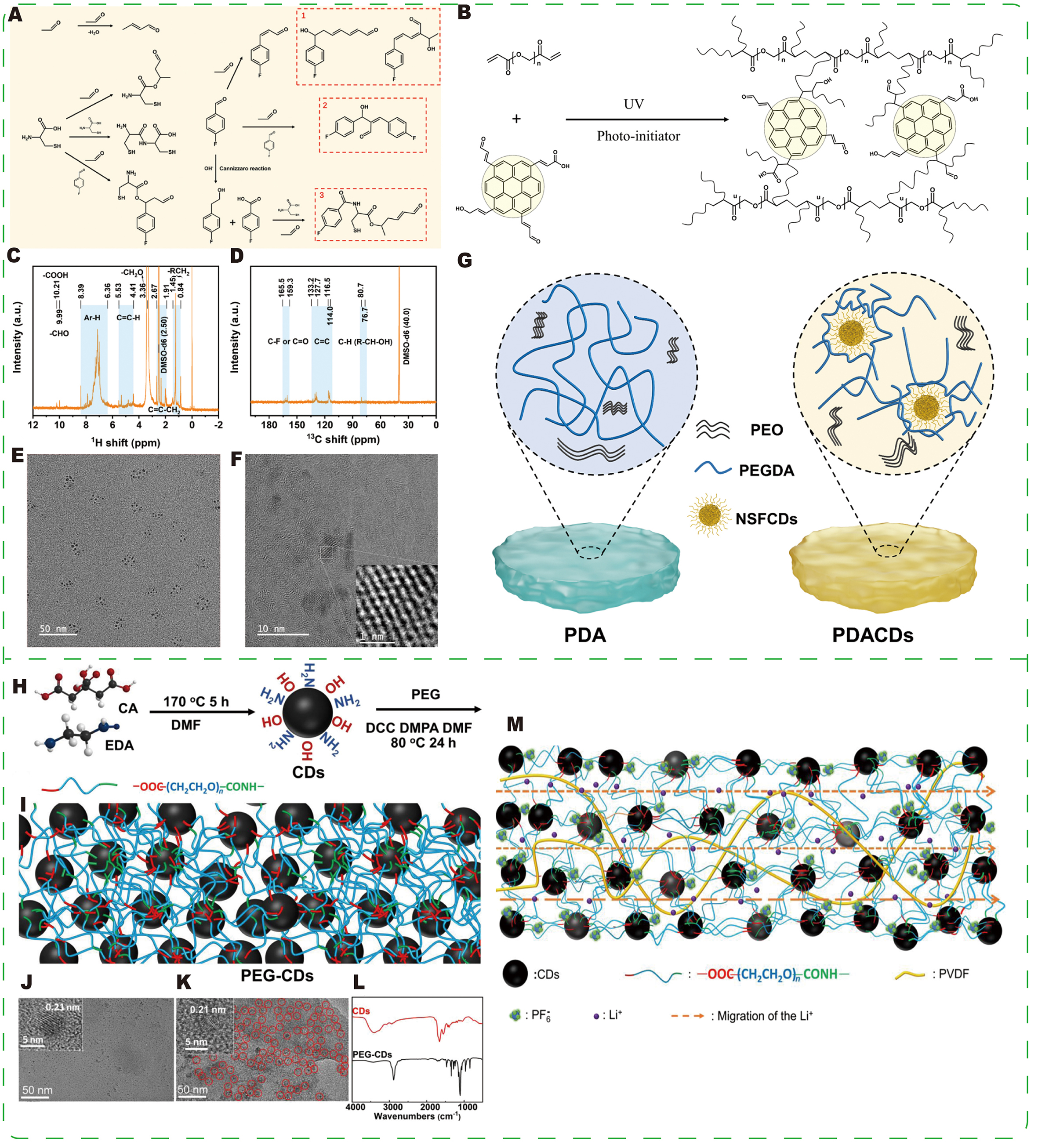
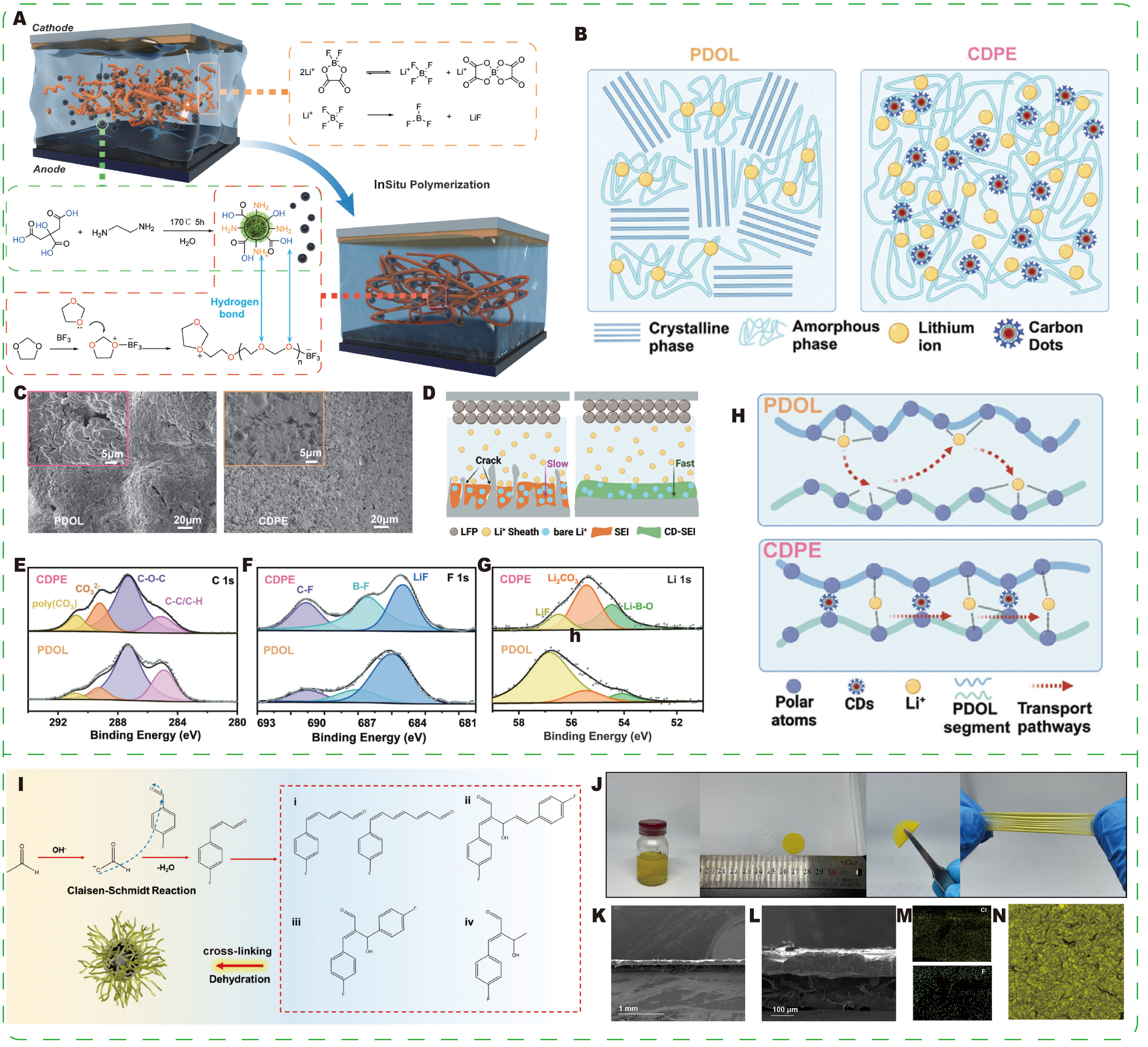
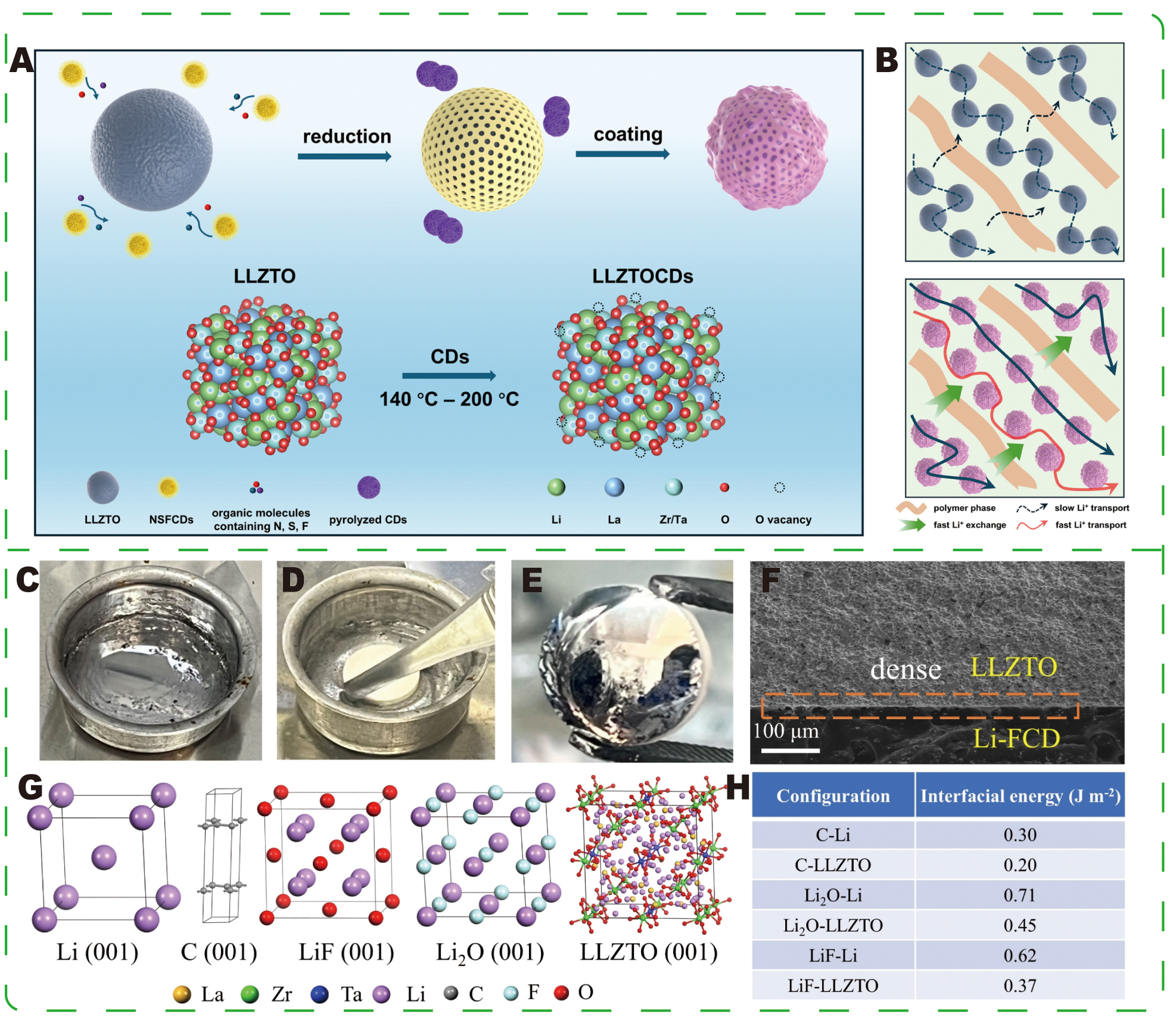
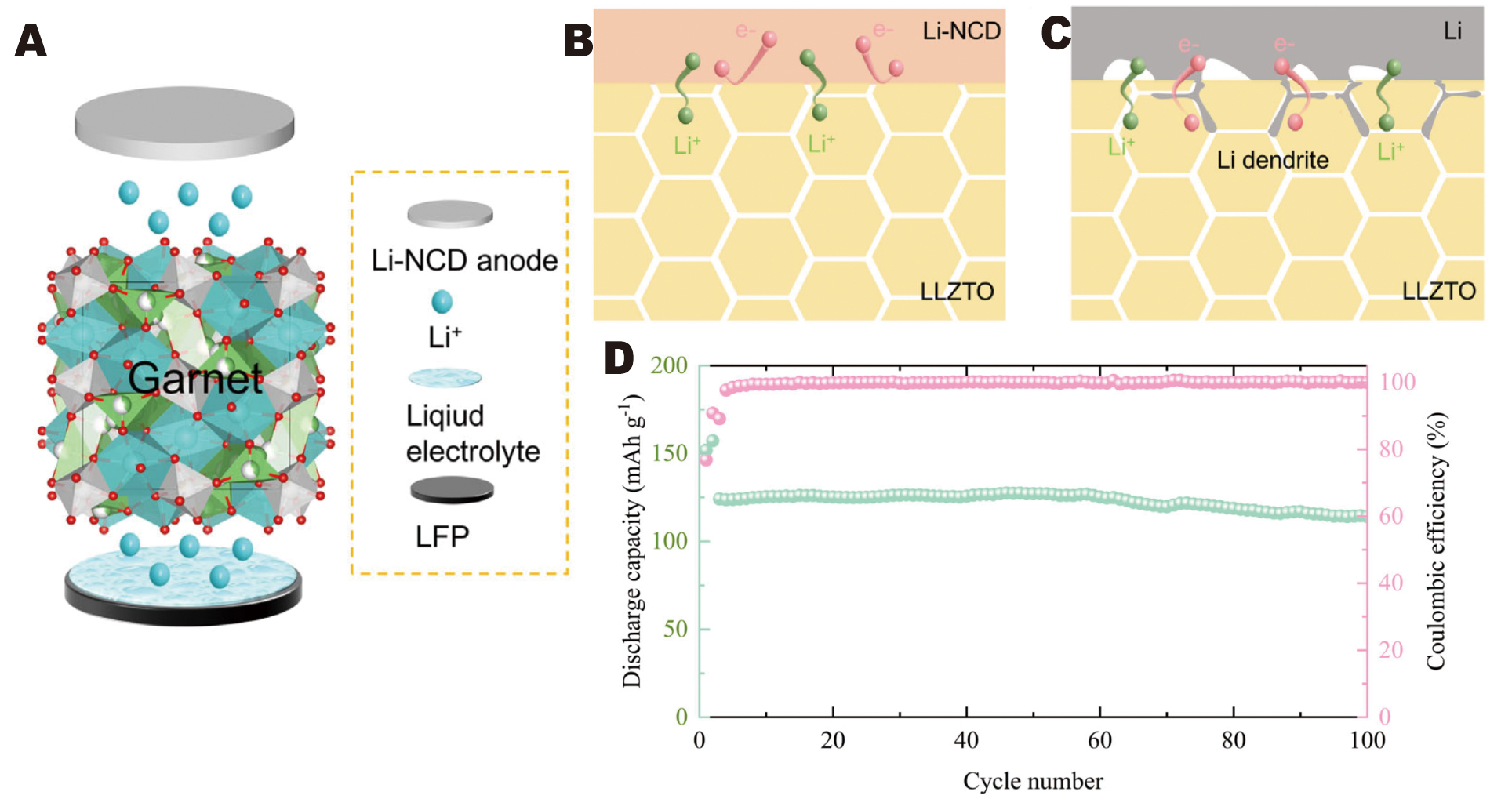












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.