Stimuli-responsive hydrogels for ocular tumor therapy
Abstract
Ocular tumor therapy remains severely hindered by physiological barriers, notably the blood-ocular barrier, which limits drug penetration and specificity, resulting in suboptimal efficacy and potential systemic toxicity. In this context, stimuli-responsive hydrogels have emerged as versatile platforms that enable smart, controllable drug release in response to internal cues (e.g., pH, enzymatic activity, or redox gradients) or external triggers (e.g., temperature, light, or magnetic fields). By enabling targeted delivery, improved biocompatibility, and tunable spatiotemporal control of cargo release, these hydrogels can maximize therapeutic efficacy while minimizing off-target effects. This review systematically surveys recent advances in the design principles, mechanisms, and applications of stimuli-responsive hydrogels across ocular oncology, with emphasis on chemotherapy, immunotherapy, and combination strategies. Furthermore, we discuss translational challenges, including long-term biocompatibility, inter-patient variability in stimulus profiles, and scalable manufacturing processes. Emerging strategies, such as multi-stimuli-responsive platforms, theranostic designs, and personalized hydrogel systems, are highlighted as avenues to enhance safety and clinical translation. Collectively, the perspectives presented herein aim to guide the development of next-generation hydrogel-based therapies for ocular tumors.
Keywords
INTRODUCTION
Ocular tumors, such as uveal melanoma (UM) and retinoblastoma (Rb), pose substantial clinical challenges due to their prevalence and potential for severe visual impairment or metastasis in advanced cases[1]. The eye possesses intricate anatomical and physiological barriers, including the blood-aqueous barrier (BAB) and blood-retinal barrier (BRB), which maintain intraocular homeostasis by restricting the entry of harmful substances and macromolecular drugs from the systemic circulation[2,3]. However, these protective mechanisms also hinder the effective delivery of chemotherapeutic agents administered via systemic route such as intravenous or oral injection, preventing them from reaching therapeutic concentrations at ocular tumor sites[4]. Consequently, achieving efficacious drug levels in ocular tissues often requires substantially higher systemic doses. These elevated doses are associated with pronounced systemic toxicities, including bone marrow suppression and hepatic or renal impairment, and are poorly tolerated by patients.
Local delivery via eye drops exhibits poor bioavailability due to rapid tear turnover, blinking, and the corneal barrier[5,6]. The vitreous cavity, a constrained compartment of approximately 4 mL, accepts only 50-100 μL per intravitreal injection[7]. Additionally, repeated injections elevate risks of serious complications, such as endophthalmitis, hemorrhage, and retinal detachment[8,9]. Moreover, retinal photoreceptors and neurons are highly vulnerable to toxic insults from chemotherapeutics or radiation[10,11]. Thus, effective ocular tumor therapy should carefully balance potent tumor eradication with preservation of healthy retinal tissue to avoid irreversible vision loss.
Current treatments for ocular tumors, including surgery, radiotherapy, systemic chemotherapy, and intraocular injections, are constrained by substantial limitations in both efficacy and safety[12]. Surgical resection, though frequently required, risks irreversible vision impairment, structural damage to the eye, and possible tumor spread[13]. Radiotherapy, including brachytherapy and proton beam therapy, is frequently accompanied by serious adverse effects such as radiation retinopathy, cataract formation, dry eye syndrome, and iris neovascularization[14]. Furthermore, brachytherapy necessitates secondary surgical procedures for the placement and removal of radioactive sources, thereby elevating the risks of infection and operative complications[15-17]. Systemic chemotherapy is markedly hindered by nonspecific systemic toxicity, limited intraocular drug penetration imposed by the blood-ocular barrier, and the development of multidrug resistance upon repeated administration, often culminating in therapeutic failure and tumor relapse[18]. Although intraocular injections (e.g., intravitreal or ophthalmic artery chemoinfusion) can bypass biological barriers, they require repeated invasive procedures, which increase the incidence of infection, hemorrhage, and retinal detachment[19-21]. Additionally, inadequate drug distribution within the ocular compartments often results in subtherapeutic concentrations in certain tumor regions[22]. Consequently, these conventional approaches, often invasive and associated with functional impairment or cosmetic deficits, highlight the critical need for localized, sustained, and targeted treatment strategies that maximize antitumor efficacy while minimizing damage to healthy ocular tissues.
Stimuli-responsive hydrogels have emerged as a promising strategy to address the challenges associated with conventional therapies for ocular tumors, providing an advanced platform for targeted and controlled drug delivery[23,24]. These hydrogels consist of three-dimensional hydrophilic polymer networks that closely mimic the natural ocular environment, such as the vitreous humor and extracellular matrix, thereby providing excellent biocompatibility and structural integrity[25-27]. A major advantage of stimuli-responsive hydrogels is their tunable physicochemical properties, which enable responsiveness to endogenous signals within the tumor microenvironment (TME) or to external stimuli. Such responsiveness triggers controllable transformations, including swelling, degradation, or sol-gel transitions, ultimately enabling precise spatiotemporal regulation of therapeutic release[28,29]. This on-demand delivery capacity helps maintain therapeutically effective drug levels at the tumor site while reducing systemic exposure, off-target toxicity, and collateral damage to healthy ocular tissues[30,31]. Moreover, these hydrogels can be engineered to support sustained local drug release, accurate targeting, minimally invasive administration, and the co-delivery of multiple agents, highlighting their considerable potential to improve outcomes in ocular tumor therapy[32-34].
This review provides a systematic overview of recent advances and emerging directions in stimuli-responsive hydrogels for ocular tumor therapy, with particular emphasis on their design principles, material compositions, and mechanisms of controlled drug release. We first discuss how these “smart” hydrogels are engineered to overcome key clinical barriers, including the blood-ocular barrier, restricted orbital volume, non-specific toxicity, and the risk of vision impairment. We then summarize their current use across major therapeutic modalities, such as chemotherapy, surgery, immunotherapy, and combination regimens [Figure 1]. Finally, we outline the remaining translational hurdles and highlight promising strategies, ranging from multi-stimuli-responsive platforms and theranostic architectures to personalized delivery systems and more rigorous preclinical evaluation, to guide future research and accelerate clinical translation.
Figure 1. Schematic representation of stimuli-responsive hydrogels for ocular tumor therapy. (A) Classification of stimuli-responsive hydrogels according to internal (enzyme, pH, or redox) and external (temperature, light, ultrasound, or magnetic field) stimuli; (B) Therapeutic applications of stimuli-responsive hydrogels in ocular tumors, including chemotherapy, surgery, immunotherapy, and combinational therapy. TME, tumor microenvironment; PTT, photothermal therapy; CAR-T, chimeric antigen receptor T-cell immunotherapy. Created in BioRender. Li, H. (2026) https://BioRender.com/2ktdiv3. TME: Tumor microenvironment; PTT: photothermal therapy; CAR-T: chimeric antigen receptor T-cell; CTLA-4: cytotoxic T-lymphocyte-associated protein 4; PD-L1: programmed death ligand 1; CD8+: Cluster of Differentiation 8-positive.
CLASSIFICATION AND MECHANISMS OF STIMULI-RESPONSIVE HYDROGELS
Stimuli-responsive hydrogels are broadly classified into two categories based on the origin of their activation signals: internal and external stimuli-responsive types[35,36]. Internal stimuli-responsive hydrogels depend on specific physiological cues within the TME, such as pH variations, enzyme activities, or redox imbalances[37-39]. In contrast, external stimuli-responsive hydrogels undergo structural modifications in response to physical stimuli such as temperature, light, magnetic fields, or ultrasound, enabling controlled drug release [Figure 1].
Internal stimuli-responsive hydrogels
pH-responsive hydrogels
pH-responsive hydrogels exploit the acidic TME to achieve targeted drug delivery[40]. Intraocular tumors, including UM and Rb, exhibit an extracellular pH of 6.5-6.8 due to aerobic glycolysis (Warburg effect) and lactate accumulation, which is significantly more acidic than healthy intraocular tissues (pH 7.2-7.4)[41]. This gradient enables the design of hydrogels incorporating ionic groups, such as carboxyl or amino groups in chitosan or polyacrylic acid. Chitosan, with a pKa of approximately 6.5, undergoes protonation of amino groups when environmental pH falls below its pKa, generating electrostatic repulsion between cationic chains and increasing osmotic pressure (Donnan equilibrium), which induces network swelling and pore enlargement[42]. Conversely, poly(acrylic acid) (PAA) contains carboxylic groups (pKa ≈ 4.5-5.0) that deprotonate at neutral pH, enabling complementary pH-responsive swelling behavior[43]. For instance, Fan et al. reported a polypeptide hydrogel that disintegrates at pH 6.5 as a result of enhanced electrostatic repulsion, thereby promoting rapid release of doxorubicin (DOX) and conbercept[44].
pH responsiveness can also be achieved through acid-labile bonds: Schiff bases (imines) hydrolyze rapidly at pH 5.0-6.0; hydrazones at pH 5.0-6.5; acetals/ketals at pH < 6.0; and oxime bonds at pH 5.5-6.5[45]. These bonds remain stable at physiological pH 7.4 but undergo acid-catalyzed hydrolysis in the TME, triggering payload release through network disintegration (covalent bond scission) rather than mere physical swelling[46]. For instance, Zhang et al. employed Schiff base cleavage in a hydrogel to suppress immune cascades and promote corneal repair in acidic inflammatory microenvironments[47]. Furthermore, combining charged groups with acid-sensitive bonds in a nanocomposite hydrogel, as demonstrated by Zhou et al., enables synergistic drug release, achieving up to 70% release at pH 5.5[48].
Enzyme-responsive hydrogels
Enzyme-responsive hydrogels exploit tumor-associated enzymatic activities to achieve spatiotemporally controlled drug release. In intraocular malignancies, matrix metalloproteinases (MMPs) are critical biomarkers for tumor invasion and metastasis. Specifically, MMP-1, MMP-2, and MMP-9 are significantly overexpressed in UM tissues compared to normal uveal tissue, with high MMP-9 levels correlating with poor prognosis and increased metastatic risk[38]. Similarly, in Rb, MMP-2 and MMP-9 exhibit elevated activity in invasive tumor cells, providing a distinct enzymatic signature for targeted drug delivery[49]. MMPs can specifically cleave peptide substrates engineered into the hydrogel network as crosslinkers[50,51]. This enzymatic cleavage induces hydrogel degradation and drug release within tumor cells or peritumoral regions, thereby offering a non-invasive therapeutic strategy. MMP-sensitive systems have been extensively investigated. For instance, MMP-2-sensitive peptide-crosslinked hydrogels have been designed for cancer therapy, where they integrate photothermal, chemotherapeutic, and immunotherapeutic modalities to effectively inhibit both primary and metastatic lesions[52].
In the ocular field, enzyme-mediated hydrogels have shown particular promise for sustained intraocular delivery. For example, Lee et al. reported an injectable hydrogel formed via horseradish peroxidase- and hydrogen peroxide-mediated crosslinking, which enabled prolonged intravitreal release of bevacizumab and consequently reduced injection frequency in retinal vascular diseases[53]. In another study, Chen et al. utilized triglycerol monostearate (TG-18) to form MMP-9-responsive hydrogels via heat-cooling self-assembly for the treatment of inflammation-related eyelid disorders[54]. In vitro, the presence of MMP-9 accelerated hydrogel degradation, increasing the cumulative 30-day drug release from approximately 30% to 50%[54].
Redox-responsive hydrogels
Redox-responsive hydrogels exploit the abnormally elevated glutathione (GSH) levels in the TME, where intracellular concentration reaches 1-10 mM, approximately 100- to 1,000- fold higher than extracellular levels (2-20 μM) and normal tissues[55]. The rapid proliferation and hypoxic metabolism of tumor cells induce a pronounced redox imbalance, resulting in intracellular GSH concentrations that are markedly higher than those in normal tissues[56]. This steep redox gradient serves as the thermodynamic driving force for site-specific drug release[57]. To harness this feature, these hydrogels are commonly engineered with disulfide (-S-S-) linkages that undergo specific cleavage via a thiol-disulfide exchange mechanism; nucleophilic attack by GSH thiolate anions (GS-) on the electrophilic sulfur atoms of the disulfide bond results in disulfide scrambling and eventual bond scission, leading to network degradation and triggered intracellular drug release[58,59]. For example, Li et al. reported a methionine-containing copolymer hydrogel with dual redox and thermal responsiveness, enabling rapid degradation and co-delivery of DOX, resiquimod (R848), and antibody targeting programmed cell death protein 1 (αPD-1) within the TME[60]. This platform exhibited potent antitumor efficacy and favorable biocompatibility in combined chemotherapy-immunotherapy. In addition to disulfide linkages, a variety of redox-responsive chemistries have been developed to correspond with specific oxidative stress levels within the TME. These include diselenide (Se-Se) bonds, which are cleavable under milder oxidizing conditions[61], and thioether (-S-) groups that respond to hyperoxidation[62]. Furthermore, reactive oxygen species (ROS)-sensitive moieties, such as thioketals and boronate esters, have been designed to be selectively cleaved by H2O2 at pathological concentrations exceeding 50 μM[63,64].
External stimuli-responsive hydrogels
Thermo-responsive hydrogels
Thermo-responsive hydrogels are among the most widely investigated and clinically promising systems. They undergo reversible changes in volume, shape, hydrophilic-hydrophobic balance, and mechanical properties in response to temperature variations[65]. Representative polymers, including poly(N-isopropylacrylamide) (pNIPAM) and Pluronic®, possess a lower critical solution temperature (LCST) that facilitates temperature-triggered gelation[66,67]. Specifically, pNIPAM contains hydrophilic amide (-CONH-) groups and hydrophobic isopropyl [-CH(CH3)2] side chains. Below the LCST (~ 32 °C), the amide groups form extensive hydrogen bonds with water, maintaining the polymer in an extended, hydrated state. Heating above the LCST disrupts these hydrogen bonds and promotes hydrophobic collapse of the isopropyl groups. This results in a coil-to-globule transition, ultimately driving gel formation[68,69]. Similarly, Pluronic F127 undergoes temperature-induced micellization via progressive dehydration of its hydrophobic poly(propylene oxide) blocks. Subsequent entanglement of the hydrophilic poly(ethylene oxide) coronae from adjacent micelles leads to a percolated gel network at the critical gelation temperature[70,71]. At room temperature, these materials typically exist as injectable sols. Following administration into the body (approximately 37 °C), they rapidly transform into gels without the need for chemical crosslinking, which favors biocompatibility and supports sustained drug release[72]. This physical gelation results from supramolecular self-assembly rather than covalent crosslinking. It eliminates the need for potentially toxic crosslinking agents and enables reversible phase transitions that dynamically respond to temperature changes[73,74]. This behavior has been leveraged to design efficient ocular drug delivery platforms. For example, Luo et al. synthesized thermosensitive poly(ethylene glycol) (PEG)-poly(ɛ-caprolactone) (PCL)-PEG (PECE) block copolymers by coupling methoxy polyethylene glycol (MPEG)-PCL copolymers using 3-Isocyanatomethyl-3, 5, 5-trimethylcyclohexyl isocyanate (IPDI) reagent[75]. In vitro, the resulting PECE hydrogel enabled sustained release of loaded diclofenac sodium for up to 7 days[75]. Thermo-responsive hydrogels have also shown promise in cancer therapy[76]. For instance, Yang et al. developed a thermosensitive nanogel that remains liquid at 4 °C to facilitate administration, while undergoing gelation at body temperature[76]. This formulation has been applied in combined photodynamic therapy and immunotherapy for breast cancer, demonstrating effective temperature-regulated drug release.
Light-responsive hydrogels
Light-responsive hydrogels are a class of smart materials that undergo reversible or irreversible physical and chemical changes upon exposure to light of specific wavelengths, including ultraviolet (UV), visible, or near-infrared (NIR) regions[77,78]. As an external stimulus, light is attractive because it is non-invasive and allows precise spatiotemporal regulation, while its effects can be tuned by adjusting the intensity, wavelength, and irradiation duration[79]. Depending on the nature of the photosensitizer, light-triggered responses can be reversible or irreversible. Specifically, light irradiation induces processes such as cleavage, isomerization, or dimerization of photosensitive groups, thereby leading to network de-crosslinking, degradation, swelling, or contraction of the hydrogel[80-82].
A widely used strategy is to incorporate photothermal nanomaterials, such as gold nanorods (GNRs), graphene, black phosphorus, polydopamine nanoparticles, or melanin nanoparticles, into the hydrogel matrix[83-86]. Upon NIR irradiation, these nanomaterials convert light into heat, trigger phase transitions within the gel, and promote on-demand drug release, thus enabling photothermal therapy (PTT)[87,88]. For example, Hu et al. reported a light-responsive nanostructured hydrogel, which was formed by indocyanine green (ICG), gemcitabine (GEM), anti-lipopolysaccharide (LPS) antibody, Poly A nucleotides, mesoporous silica nanoparticle (MSN), and methacrylated hyaluronic acid (HAMA) (I/G-LPMSN@HAMA), capable of eliminating intra-tumoral bacteria and activating the immune system, thus achieving synergistic antibacterial, antitumor, and immunostimulatory effects[89]. Under 785 nm NIR irradiation, indocyanine green (ICG) embedded in the hydrogel converts light energy into heat, elevating local temperatures to induce photothermal ablation of tumor cells and bacteria. Simultaneously, the generated heat disrupts A-T base pairing, allowing spatiotemporally controlled drug release that enhances chemotherapeutic efficacy while reducing systemic toxicity. This thermally triggered release is mediated by the temperature-induced dehybridization of DNA duplexes or dissociation of thermolabile covalent bonds, which destabilize the hydrogel network and facilitate payload diffusion[90].
In addition, the photothermal effect can induce immunogenic cell death (ICD), thereby supporting photoimmunotherapy[91]. Beyond tumor ablation, light-responsive hydrogels have also been applied to ocular tissue repair. For example, Xu et al. developed a light-responsive exosome-loaded hydrogel (3D-Exo-gelatin methacrylate (GelMA)) for corneal wound healing[92]. Upon exposure to 405 nm light, the 3D-Exo-GelMA hydrogel undergoes rapid gelation within 30 s, enabling convenient in situ formation and controlled exosome delivery. This rapid gelation is mediated by a Type I photoinitiation mechanism. Specifically, lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) absorbs 405 nm photons and undergoes α-cleavage, generating benzoyl and phosphinoyl radicals[93]. These radicals initiate chain-growth polymerization by attacking the methacrylate vinyl groups (C=C) of GelMA. This leads to covalent crosslinking between gelatin chains via carbon-carbon bonds, rapidly establishing a stable three-dimensional network[94].
Magnetic-responsive hydrogels
Magnetic-responsive hydrogels incorporate superparamagnetic iron oxide nanoparticles (SPIONs), which generate heat under an alternating magnetic field (AMF)[95,96]. This magnetothermal effect enables spatiotemporally controlled drug release and magnetic hyperthermia (MHT)[97,98]. For example, Gong et al. developed an injectable magnetic nanocomposite hydrogel (CG-IM) for multidisciplinary treatment of hepatocellular carcinoma[99]. In a liver resection model, postoperative injection of CG-IM followed by AMF irradiation resulted in a 0% recurrence rate[99]. In an unresectable subcutaneous tumor model, intra-tumoral CG-IM combined with AMF increased the local temperature to approximately 46 °C, markedly reducing tumor volume and weight[99]. This treatment induced extensive tumor cell death and significantly elevated tumor necrosis factor-alpha (TNF-α) expression. Moreover, in an in situ VX2 liver carcinoma model in rabbits, ultrasound-guided percutaneous administration of CG-IM followed by 15 min of AMF irradiation achieved complete tumor eradication[99]. These outcomes highlight the efficacy of magnetothermal ablation mediated by embedded magnetic nanoparticles. Similarly, Ma et al. designed an injectable, multi-stimuli-responsive artesunate nanoparticles hydrogel (ART-NP-hydrogel) that integrates chemotherapy, PTT, and chemo-dynamic therapy for multimodal liver cancer treatment[100]. In a murine hepatocellular carcinoma model, the combination of an external magnetic field, NIR irradiation, and ART-NP-hydrogel almost completely suppressed tumor growth with excellent biosafety[100]. In addition, Cyanine7 (Cy7)-labeled drugs loaded in the hydrogel exhibited markedly prolonged retention at tumor sites compared with free drugs, underscoring the potential of magnetic-responsive hydrogels for sustained, localized cancer therapy[100].
Ultrasound-responsive hydrogels
Ultrasound-responsive hydrogels exploit the deep tissue penetration and noninvasive characteristics of ultrasound, using thermal and cavitation effects to induce network restructuring and spatiotemporally controlled drug release[101,102]. For instance, Meng et al. developed an ultrasound-responsive, self-healing nanoclay hydrogel (NC gel) system for remotely regulated nanovaccine delivery, enabling a single-injection, multiple-stimulation cancer immunotherapy strategy[103]. In this system, ultrasonic mechanical vibrations transiently disrupt weak physical interactions, including hydrogen bonds between polymer chains and clay platelets, converting the gel into a sol state and promoting rapid nanovaccine release. Once ultrasound is discontinued, the hydrogel network rapidly re-forms, allowing repeated, on-demand dosing. Beyond drug delivery, ultrasound-responsive hydrogels have also been investigated for shape-memory applications, underscoring their versatility in biomedical engineering[104]. In these systems, acoustic energy induces localized heating or mechanical deformation that temporarily fixes a temporary shape; upon cessation of the ultrasonic stimulus, the hydrogel recovers its permanent shape through entropy elasticity and the reformation of physical crosslinks, enabling minimally invasive deployment and actuation within deep tissues[105].
PREPARATION METHODS OF STIMULI-RESPONSIVE HYDROGELS
Stimuli-responsive hydrogels can be fabricated through various methods, with selection depending on response mechanisms, application requirements, and desired material properties. Primary strategies include chemical crosslinking, physical crosslinking, radiation-induced crosslinking, and graft crosslinking [Figure 2A].
Figure 2. Design and characterization of stimuli-responsive hydrogels for ocular tumor therapy. (A) Representative crosslinking strategies used to construct stimuli-responsive hydrogels, including chemical, physical, photoinduced, and grafting-based crosslinking; (B) Main characterization approaches for stimuli-responsive hydrogels, including rheological behavior, morphology, swelling and degradation profiles, and drug-release kinetics. Created in BioRender. Li, H. (2026) https://BioRender.com/p3im2n2.
Chemically crosslinked hydrogels
Chemically crosslinked hydrogels form permanent three-dimensional networks through covalent bonds, offering precise control over structure and enhanced mechanical robustness. Representative strategies include radical polymerization, photo-crosslinking, and click chemistry. In radical polymerization, vinyl monomers such as acrylamide or N-isopropylacrylamide (NIPAM) are polymerized in the presence of initiators (e.g., ammonium persulfate, APS) and catalysts (e.g., N,N,N’, N’-tetramethylethylenediamine, TEMED), while bifunctional crosslinkers such as N, N-methylenebisacrylamide (MBA) integrate the network[106]. This well-established method is highly tunable and is frequently employed for thermosensitive and pH-responsive hydrogels. Its suitability for ocular application is enhanced by the LCST of NIPAM, which is approximately 32 °C. This property allows an injectable solution to undergo a liquid-to-gel transition upon intravitreal administration, as it warms from room temperature (~ 25 °C) to physiological temperature (37 °C). Such in situ gelation enables minimally invasive delivery to the posterior segment of the eye, thereby avoiding the need for surgical implantation[107]. For instance, Zhu et al. reported an acid- and GSH-dual-responsive hydrogel formed via a Schiff base reaction between polyethyleneimine (PEI) and oxidized sodium alginate (OSA), which gelled within 40 s without additional catalysts and exhibited excellent injectability and self-healing properties[108].
Photo-crosslinking, a subset of chemical crosslinking, employs photo-initiators to induce polymerization under UV or visible light[109]. This method is particularly advantageous for ocular tumor therapy owing to the optical transparency of the eye, which allows non-invasive, external modulation of gelation and drug release kinetics. Moreover, photo-crosslinking can be seamlessly integrated with photodynamic therapy, a first-line treatment for choroidal melanoma. In such systems, the same light source used for hydrogel formation can simultaneously activate photosensitizers (e.g., chlorin e6 or verteporfin), generating cytotoxic ROS and thereby enabling synergistic chemo-photodynamic outcomes[110]. For example, Hu et al. developed a UV-crosslinked hydrogel (I/G-LPMSN@HAMA) for combined antibacterial and antitumor therapy, harnessing the spatial precision of light to confine gel formation specifically to the tumor site[89].
Alternatively, click chemistry provides an alternative route to covalent crosslinking through highly efficient, chemo-selective reactions under mild conditions, thereby facilitating complex and multifunctional hydrogel architectures[111,112]. For ocular tumor therapy, this bioorthogonal strategy is particularly valuable when delivering sensitive biologics such as monoclonal antibodies or immune checkpoint inhibitors. Compared to conventional radical polymerization, click reactions (e.g., thiol-ene and Diels-Alder) proceed under physiological conditions without requiring cytotoxic catalysts, UV irradiation, or generating excessive heat. This gentle process is crucial for preserving the conformational integrity and bioactivity of protein therapeutics[113]. Furthermore, hydrogels formed via click chemistry exhibit robust, covalently crosslinked networks that resist rapid dissolution in the highly fluid vitreous humor. This stability enables sustained drug release over extended periods, effectively overcoming the short intravitreal half-life of injected antibodies[114]. For instance, Ail et al. fabricated a reduction-responsive carboxymethyl cellulose (CMC) hydrogel via inverse electron demand Diels-Alder (IEDDA) click chemistry, achieving high biocompatibility and tumor-targeting capability[115]. In another study, Ilochonwu et al. further developed a thermo-responsive hydrogel based on a Diels-Alder reaction for drug delivery to the posterior segment of the eye[116].
Physically crosslinked hydrogels
Physically crosslinked hydrogels are formed through reversible, noncovalent interactions, including hydrogen bonding, electrostatic attraction, hydrophobic association, and ionic coordination, thus generating biocompatible, injectable networks without the need for chemical crosslinkers[117]. Thermosensitive polymers, such as agarose and poly(lactic-co-glycolic acid) (PLGA)-PEG-PLGA triblock copolymers, undergo a sol-gel transition in the vicinity of body temperature, which enables minimally invasive administration, in situ gelation, and sustained drug release[118,119].
In ionically crosslinked systems, multivalent cations (e.g., Ca2+ or Fe3+) bridge anionic polymers such as alginate or carboxymethyl chitosan (CMCS), frequently affording hydrogels with intrinsic pH responsiveness[120]. Alginate, for example, forms stable “egg-box” structures upon coordination with Ca2+, which markedly improves the mechanical robustness of the gel network[121]. Recently, Guan et al. developed an injectable immune-activating hydrogel by mixing Mn2+ with an antitumor peptide (N‑Pep)[122]. Coordination between Mn2+ and functional groups (-COO-, Tyrosine, or Phenylalanine), combined with π-π stacking and hydrogen bonding, prompted peptide self-assembly into β-sheet nanofibers and eventual hydrogelation.
Additional crosslinking strategies
Irradiation crosslinking uses high-energy radiation (e.g., γ-rays or electron beams) to generate radicals along polymer chains, thereby initiating network formation without added chemical initiators. The crosslinking density can be tuned by adjusting the radiation dose, dose rate, and oxygen content[123]. For example, Elshahawy et al. prepared a poly(vinyl alcohol)/tragacanth gum/graphene oxide (PVA/TG/GO) composite hydrogel via electron beam irradiation[124]. Irradiation at 25-45 kiloGray (kGy) increased crosslinking density, gel fraction, and porosity, while reducing swelling capacity, thus yielding an optimized matrix for wound dressing applications. In parallel, graft crosslinking introduces functional moieties onto polymer backbones to improve mechanical and/or biological performance. For instance, Cheng et al. grafted 3-aminophenylboronic acid (PBA) onto CMC through amide coupling, producing a mucoadhesive PBA-CMC hydrogel for GSH delivery in the treatment of dry eye syndrome[125].
Overall, each crosslinking strategy provides distinct advantages. The selection should be guided by the desired function (e.g., stimulus responsiveness, drug release profile, or mechanical robustness), application scenario (e.g., injectability or target tissue), and biocompatibility requirements. Chemical crosslinking affords highly stable networks but may leave residual toxic reagents. In contrast, physical crosslinking offers superior biosafety at the cost of long-term stability. Irradiation and click chemistry are well-suited to high-performance or specialized settings, whereas hybrid strategies combine multiple mechanisms to broaden the design space for multifunctional hydrogel systems.
CHARACTERIZATION OF STIMULI-RESPONSIVE HYDROGELS
Systematic characterization of physicochemical properties, network microstructure, stimuli-responsive behavior, and drug-release profiles is essential to ensure reliable performance of stimuli-responsive hydrogels in ocular tumor therapy. To this end, rheometry, electron microscopy, swelling and degradation assays, and in vitro drug-release studies are commonly employed to quantify mechanical behavior, structural features, stability, and release kinetics under physiologically relevant conditions [Figure 2B].
Rheological properties
Rheological analysis is a primary tool for characterizing the mechanical behavior and phase transition of hydrogels, including sol-gel transition temperature, shear-thinning behavior, self-healing capacity, and injectability[126]. For example, Duan et al. employed a rheometer to assess the thermosensitive gel comprising of arsenic trioxide (As2O3)-loaded polyethylene glycol-dipalmitoylphosphatidylethanolamine (mPEG-DPPE) calcium phosphate nanoparticles encapsulated within an injectable thermoresponsive hydrogel (ANPs-Gel)[127]. At low temperatures (< 25 °C), the storage modulus (G”) exceeded the loss modulus (G’), indicating a liquid state[127]. As temperature increased to approximately 27.1 °C, the crossover of G’ and G” signaled the sol-gel transition. At physiological temperature (37 °C), G’ became higher than G”, confirming the formation of a solid-like gel.
Frequency sweep tests revealed nearly linear responses of both G’ and G” over the viscoelastic range, demonstrating stable rheological properties. Under high strain, G’ decreased sharply, reflecting a fluid-like state. However, when the strain was reduced to 0.1%, G’ rapidly recovered within 10 s and remained consistent over multiple cycles, demonstrating efficient self-healing[127]. Similarly, P[N-isopropylacrylamide (NIPAM)-co-N-acryloxysuccinimide (NAS)-co-methacrylated dexamethasone (mDEX)]-PEG-P(NIPAM-co-NAS-co-mDEX)-cystamine (PNADEX-CA), an injectable thermosensitive hydrogel developed for chronic ocular inflammation, exhibited stable gelation and self-healing properties[128]. After injection through a 30 G needle, the hydrogel maintained its structural integrity, further confirming its shear-thinning behavior and self-healing properties, which are advantageous for localized ocular delivery.
Morphology
Morphological characterization, primarily by scanning electron microscopy (SEM), enables detailed observation of the microporous architecture of hydrogels, including porosity, pore size, and interconnectivity[129]. These structural parameters directly influence drug-loading capacity, cell permeability, nutrient diffusion, and tissue integration. To preserve the native 3D architecture, hydrogel samples are typically freeze-dried prior to imaging. For instance, Li et al. reported a mussel-inspired “plug-and-play” hydrogel (BP-Ag@HA-DA-Plu; black phosphorus-Ag nanocomposites-loaded dopamine-modified hyaluronic acid-Pluronic® F127) that displayed a uniform, interconnected porous network under SEM, with pore sizes of 10-20 μm, which was favorable for cell ingrowth and tissue penetration[130]. High-magnification SEM further revealed a homogeneous distribution of black phosphorus-Ag (BP-Ag) nanosheets within the matrix, confirming efficient nanofiller loading and structural stability.
In addition, transmission electron microscopy (TEM) is widely employed to resolve the dispersion, morphology, and size distribution of nanoscale components (e.g., nanoparticles, liposomes, or nanosheets) embedded in composite hydrogels. Such high-resolution imaging provides critical insight into nano-structural uniformity and the degree of integration between the nanomaterials and the hydrogel network, which in turn underpins the reproducibility and performance of stimuli-responsive ocular drug delivery systems.
Swelling and degradation
Swelling and degradation behaviors are critical for assessing hydrogel stability and responsiveness under physiological environments[131]. Swelling capacity, typically determined by periodic weighing of dry gels immersed in buffers with different pH values or ionic strengths, reflects both water uptake and network integrity. Although pronounced swelling can facilitate drug diffusion, excessive swelling may compromise mechanical stability and induce burst release.
Degradation profiles are commonly assessed by monitoring mass loss or molecular weight changes under simulated physiological conditions or in the presence of specific triggers (e.g., enzymes, ROS, or GSH), with the ideal degradation rate coordinated with the desired drug release and treatment schedule[132,133]. For instance, Wang et al. developed an ROS-responsive hydrogel [N1-(4-boronobenzyl)-N3-(4-boronophenyl)-N1,N1,N3,N3-tetramethylpropane-1,3-diaminium (TSPBA)-poly(vinyl alcohol) (PVA)] that remained largely intact for 6 days under normal conditions but underwent rapid degradation in a simulated TME containing 0.5 mmol L-1 H2O2. This indicated high ROS sensitivity and site-selective breakdown[134]. Consistently, in vivo the hydrogel persisted for more than 2 weeks in normal tissue yet degraded completely within 2 weeks in tumor tissue. In another study, EKGel, which was prepared by the reaction between chemically modified cellulose nanocrystals and gelatin, showed negligible swelling and almost no volume change in phosphate buffered saline (PBS), indicating robust structural stability and resistance to expansion[135]. Under flow conditions mimicking physiological shear stress, EKGel exhibited only ~ 14% volume reduction over 5 days, in contrast to ~ 60% for basement membrane extract (BME), highlighting its superior shear resistance for dynamic settings such as microfluidic systems[135].
Drug release kinetics
Drug release kinetics are a key indicator of therapeutic efficacy and safety in hydrogel-based delivery systems. Typically, drug-loaded hydrogels are incubated in simulated body fluids (e.g., PBS) or stimulus-specific environments (e.g., acidic pH, ROS, or enzymes-containing solutions). Aliquots collected at predefined time points are then analyzed by high-performance liquid chromatography (HPLC), UV-Visible, or fluorescence spectroscopy to construct release profiles[136]. These profiles are commonly fitted with kinetic models such as Higuchi or Korsmeyer-Peppas equations to elucidate the dominant release mechanisms.
Recent studies underscore that TME-associated stimuli (e.g., ROS overproduction or local pH shifts) can be exploited to program drug release, thereby improving intra-tumoral retention and penetration[137,138]. For example, Liang et al. co-loaded R848 and anti-OX40 (aOX40) into a ROS-responsive hydrogel (abbreviated as PHOENIX) and evaluated release in PBS, 1 mM H2O2, and 10 mM H2O2 at 37 °C under agitation[137]. Cumulative drug release was significantly accelerated in H2O2-containing media relative to PBS, with the 10 mM H2O2 group exhibiting the fastest release, confirming ROS-triggered bond cleavage as the principal mechanism[137]. R848 displayed a rapid initial burst (~ 60% within 10 h), followed by a sustained phase, whereas aOX40 showed a delayed onset and reached ~ 50% cumulative release at approximately 20 h. Similarly, Luo et al. reported a pH-responsive nanocomposite hydrogel (denoted as PDO gel) formed via dynamic Schiff base linkages[138]. The network remained stable at physiological pH, but drug release was markedly accelerated under mildly acidic conditions (pH 6.5) characteristic of tumor tissues, due to acid-induced Schiff base cleavage[138]. Oxaliplatin (OXA) release was substantially higher at pH 6.5 than at pH 7.4 and further increased with decreasing crosslinking density, validating an acid-sensitive, tunable release profile[138].
Beyond environmental triggers, the intrinsic network architecture of hydrogels, particularly mesh size (ξ) and crosslinking density (ν), plays a central role in regulating drug diffusion kinetics. As described by the Flory-Rehner model, mesh size decreases with increasing crosslinking density (ν ≈ 1/ξ3), directly shaping the effective diffusion coefficient of therapeutic molecules within the polymer network[139]. For ocular tumor therapy, where drugs need to navigate the vitreous humor and cross the BRB, tight control over these structural parameters is essential[140,141]. High crosslinking densities (> 5 mol/m3, mesh size < 20 nm) create restrictive and tortuous diffusion paths, slowing the release of small-molecule chemotherapeutics such as OXA and cisplatin over extended periods. This sustained release could maintain drug levels within the therapeutic window in the posterior segment[142,143]. In contrast, lower crosslinking densities generate larger mesh sizes that promote faster molecular transport and a more pronounced initial burst release, which may be advantageous when rapid cytotoxic activity is needed for highly aggressive intraocular tumors[144,145].
Overall, comprehensive system characterization is fundamental to the functional optimization and clinical translation of stimuli-responsive hydrogels. Rheological analysis defines their mechanical properties and injectability, while electron microscopy reveals the underlying microstructure. Swelling and degradation assays are used to assess stability and stimulus responsiveness, and drug-release studies directly inform therapeutic performance. Looking ahead, advances in characterization methodologies, including in situ real-time monitoring and multimodal platforms, are expected to further accelerate innovation in hydrogel research.
APPLICATION OF STIMULI-RESPONSIVE HYDROGELS IN OCULAR TUMOR THERAPY
Stimuli-responsive hydrogels show great promise for ocular tumor therapy due to their excellent local retention, on-demand drug release, and favorable biocompatibility. By enabling spatially and temporally controlled delivery, these systems can enhance radio-sensitization, improve the precision of chemotherapy, and support effective postoperative adjuvant treatment. Consequently, stimuli-responsive hydrogels are increasingly viewed not only as powerful adjuncts to existing regimens but also as potential alternatives to conventional ocular tumor therapies.
Chemotherapy
Conventional chemotherapy for intraocular tumors is severely limited by the blood-ocular barrier and systemic toxicity. Local administration strategies, such as intravitreal injection, are further compromised by rapid drug clearance and the need for frequent dosing. Stimuli-responsive hydrogels offer a promising alternative by enabling sustained intraocular drug retention and tumor-selective release via in situ gelation and environment-triggered payload delivery. This strategy is particularly relevant for Rb, the most common pediatric intraocular malignancy, in which standard regimens are constrained by suboptimal pharmacokinetics and dose-limiting toxicity. To address these challenges, diverse stimuli-responsive hydrogel platforms have been designed to achieve prolonged, localized release of chemotherapeutics, thereby enhancing antitumor efficacy while reducing systemic and ocular adverse effects.
pH-responsive hydrogels for chemotherapy
pH-responsive hydrogels have been extensively explored for enhanced intraocular drug delivery. For instance, Guo et al. developed an injectable nanocomposite hydrogel (hyaluronic acid (HA)-DOX/lapatinib (LAP) gel) for combined therapy of UM[114]. This system incorporates an alginate-dopamine (ALG-DPA) network crosslinked by endogenous Ca2+ ions and loaded with HA-modified pH-sensitive nanoparticles (HA-DOX/LAP NPs)[114]. Under the mildly acidic conditions of the TME, the hydrogel undergoes stepwise degradation and promotes cascade release of DOX and LAP, thereby enhancing intra-tumoral penetration and cellular uptake and ultimately achieving synergistic cytotoxicity[114]. Upon intravitreal injection, the formulation first forms an in situ gel, which gradually degrades to liberate the encapsulated nanoparticles. The HA shell mediates active targeting to CD44 receptors on tumor cells, and subsequent acid-triggered drug release in endo/lysosomal compartments (pH ≈ 5.0) enables concurrent intracellular action of DOX and LAP. In vivo, a single administration led to marked inhibition of tumor growth (76.1% suppression), extended survival, and reduced local recurrence and distant metastasis, while maintaining good ocular biocompatibility and low systemic exposure. This system demonstrated cascade release in acidic environments in vitro. The reliance on endogenous Ca2+ for gelation assumes stable intraocular calcium concentrations; however, vitreal Ca2+ levels fluctuate significantly with age and pathological states, potentially compromising gelation reproducibility[146].
Similarly, Cooper et al. engineered an injectable, pH-responsive dendrimer hydrogel (DH) for localized melphalan delivery in Rb[147]. Based on fifth-generation Polyamidoamine (PAMAM) dendrimers, this system enables dual drug loading via acid-labile acetal linkages for covalent drug conjugation, together with physical encapsulation. The conjugated melphalan is rapidly liberated under acidic conditions, while at physiological pH (7.4) drug release is slower and sustained, thereby achieving context-dependent release profiles [Figure 3A]. In another approach, Fan et al. developed a pH-responsive peptide hydrogel (DDIIIOH) for co-delivery of conbercept and DOX[44]. The hydrogel forms a stable, injectable 3D fibrous network at pH 7.4 with adequate mechanical integrity, yet undergoes protonation-induced disassembly at tumor-relevant pH (6.5), triggering accelerated drug release. In vitro, this platform effectively inhibited Y79 Rb cell proliferation and suppressed VEGF-driven angiogenesis in human retinal endothelial cells (HRECs)[44]. In vivo, it produced synergistic antitumor and antiangiogenic effects, leading to a marked reduction in tumor volume and microvessel density[44].
Figure 3. Stimuli-responsive hydrogels for ocular tumor chemotherapy. (A) Schematic illustration of the synthesis of a dendrimer hydrogel carrying melphalan in both conjugated and free forms and its application for retinoblastoma. Figure 3A is quoted with permission from Cooper et al.[147]; (B) Application of RENP-ICG@PNIPAM: Dox-FA in choroidal melanoma therapy. Figure 3B is quoted with permission from Li et al.[150]; (C) Fabrication process of a bilayer hydrogel microrobot; (D) Scheme of the therapeutic action of the bilayer hydrogel microrobot against Y79 retinoblastoma cells. Figure 3C and D is quoted with permission from Kim et al.[151]. DH: Dendrimer hydrogel; GSH: glutathione; TME: tumor microenvironment; NIR: near infrared; RENP: rare-earth nanoparticle; ICG: indocyanine green; PNIPAM: poly(N-isopropylacrylamide); PEGDA: poly(ethylene glycol) diacrylate; FA: folic acid; PLGA: poly(lactic-co-glycolic acid); PVA: polyvinyl alcohol; MNP: magnetic nanoparticle; AMF: alternating magnetic field.
Thermo-responsive hydrogels for chemotherapy
Thermo-responsive hydrogels represent another promising strategy that exploits physiological temperature as an endogenous trigger. For example, Huo et al. formulated a topotecan (TPT)-loaded thermosensitive hydrogel based on Poloxamer 407, which remains a sol at room temperature but rapidly gels at near-physiological temperature (~ 37 °C), thereby forming a local drug depot[148]. In a Y79 Rb xenograft model, a single injection markedly prolonged intraocular TPT retention (t1/2 > 94 h) and sustained release for > 400 h. Compared with oral TPT, the hydrogel achieved comparable antitumor efficacy while significantly reducing systemic toxicity, including hematological, hepatic, and renal damage, and simultaneously improving safety and formulation stability[148]. This hydrogel offers a potential paradigm shift from repetitive intravitreal injections to single-administration sustained-release platforms.
Building on this concept, Xie et al. developed a composite system in which curcumin nanoparticles were embedded in an in situ thermosensitive PLGA-PEG-PLGA hydrogel for localized Rb treatment[149]. The formulation is injectable as a low-viscosity liquid and undergoes rapid gelation in the vitreous, enabling prolonged intraocular drug retention and controlled release. In vitro, it potently inhibited Y79 cell proliferation and suppressed inflammatory cytokine expression, indicating combined chemotherapeutic and anti-inflammatory effects[149].
Beyond single-stimulus responsiveness, multi-stimuli-responsive systems have also been engineered. For example, Li et al. designed a dual temperature/GSH-responsive hydrogel nanocomposite composed of rare-earth-doped nanoparticles (RENP) loaded with ICG and poly(N-isopropylacrylamide) (PNIPAM) carrying doxorubicin (DOX) and folic acid (FA), RENPICG@PNIPAM: DOX-FA, for near-infrared second region (NIRII) imaging-guided photothermal-chemotherapy of choroidal melanoma[150]. Drug release was accelerated under elevated temperature induced by laser irradiation and in a high-GSH microenvironment, with release rates positively correlated with both laser power and GSH concentration [Figure 3B]. In ocular choroidal melanoma-1 (OCM-1) cells, this system induced over 95% cell death, and in vivo it elicited pronounced tumor regression, outperforming the corresponding monotherapies[150]. Collectively, these thermo-responsive hydrogel platforms enable minimally invasive, sustained intraocular chemotherapy with integrated imaging or photothermal capabilities, offering enhanced antitumor efficacy, reduced systemic toxicity, and improved local pharmacokinetics for the treatment of ocular malignancies.
Magnetically responsive hydrogels for chemotherapy
Magnetically responsive hydrogels provide a compelling platform for ocular tumor chemotherapy by enabling externally regulated drug release while reducing off-target toxicity. For instance, Kim et al. reported a bilayer hydrogel microrobot designed to overcome the challenge of residual magnetic nanoparticle accumulation after treatment[151]. In this system, the therapeutic layer consists of a gelatin/PVA hydrogel loaded with PLGA-DOX particles for drug encapsulation and release, whereas the magnetic layer is formed from a poly(ethylene glycol) diacrylate (PEGDA) hydrogel incorporating Fe3O4 nanoparticles to enable magnetic guidance and subsequent retrieval [Figure 3C]. Under an AMF, the magnetic layer generates heat, leading to dissolution of the therapeutic layer and thereby achieving on-demand, controlled drug release [Figure 3D]. Compared with free DOX, this microrobot affords sustained drug elution and significantly reduces Y79 Rb cell viability to approximately 42% after 48 h, indicating robust and prolonged antitumor activity[151]. Overall, such magnetically responsive hydrogel microrobots exemplify a versatile platform for spatiotemporally controlled ocular chemotherapy. Moreover, they combine targeted navigation, on-demand drug release, and post-treatment retrieval to enhance therapeutic precision and safety.
Physical stimuli-responsive hydrogels for chemotherapy
Physical stimuli-responsive hydrogels harness intrinsic material properties to enable localized, sustained, and targeted drug release, offering a non-classical yet effective strategy for ocular tumor therapy. For example, Taich et al. designed a non-thermosensitive sustained-release hydrogel based on a PCL-PEG-PCL triblock copolymer for intravitreal delivery of TPT[152]. By finely tuning the hydrophilic-hydrophobic balance, this system achieved prolonged TPT release (approximately 70% over 7 days) while maintaining consistent cytotoxicity against Wills Eye Research Institute-Retinoblastoma 1 (WERI-RB1) cells in vitro. In rabbit models, therapeutic TPT concentrations in the vitreous were sustained for more than 24 h[152]. Although a fourfold higher dose was required to match the area-under-the-curve (AUC) of conventional solution formulations, the hydrogel provided a gradual release profile without an initial burst. Notably, both blank and TPT-loaded hydrogels induced no detectable retinal or systemic toxicity, confirming their favorable biocompatibility.
In a related approach, Cocarta et al. developed a dual-layer methacrylate hydrogel implant for sustained local delivery of TPT and vincristine (VCR) via scleral diffusion[153]. The device comprises a hydrophilic 2-hydroxyethyl methacrylate polymer (pHEMA) inner layer serving as the drug reservoir and a hydrophobic 2-ethoxyethyl methacrylate polymer (pEOEMA) outer layer acting as a barrier to outward diffusion. Through this asymmetric architecture, the implant enables directional release, illustrating controlled delivery governed by physical structure. Similarly, Hobzova et al. reported a bilayer implant with a pHEMA inner core and a pEOEMA outer layer for directional TPT delivery[154]. In rabbit eyes, this implant exhibited excellent biocompatibility and favorable surgical handling, reaching therapeutic TPT levels (> 10 ng mL-1) within 8 h and maintaining them for up to 48 h, thereby markedly mitigating the rapid drug clearance associated with intravitreal injections[154]. Furthermore, plasma drug concentrations remained minimal, highlighting the system’s capacity to limit systemic exposure and associated toxicities.
Collectively, physically modulated hydrogels, particularly multilayer and triblock copolymer systems, can provide directional, sustained intravitreal or transscleral delivery of chemotherapeutics for ocular tumors. By prolonging intraocular drug exposure while minimizing systemic levels, such platforms effectively mitigate burst release and clearance-related limitations of conventional injections, thereby enhancing local efficacy and reducing off-target toxicity.
Surgical applications
Stimuli-responsive hydrogels are not only used for direct tumor ablation, but also serve as versatile platforms for postoperative adjuvant therapy and tissue regeneration following ocular surgery. For example, after tumor resection procedures such as local sclerectomy, hydrogels loaded with chemotherapeutic agents or immunosuppressants can be injected into the surgical cavity to eradicate residual micro-metastases and reduce the risk of recurrence. In 2023, Chen et al. reported a tetra-arm polyethylene glycol (Tetra-PEG)-based immunotherapeutic artificial vitreous body (AVB) hydrogel co-loaded with melphalan (Mel) and anti-programmed death ligand 1 (αPDL1) antibodies [Figure 4A and B][155]. In this design, melphalan was released rapidly to induce ICD, whereas αPDL1 antibody was delivered in a sustained manner to block immunosuppressive signaling, thereby significantly suppressing residual tumor growth in a choroidal melanoma surgery model. In murine models, complete tumor eradication was achieved without compromising intraocular pressure or retinal function, highlighting the dual advantages of localized immune activation and concomitant systemic antitumor responses [Figure 4C][155].
Figure 4. Stimuli-responsive hydrogels for postsurgical and local immunotherapy of ocular tumors. (A) Schematic illustration of an immunotherapeutic AVB hydrogel constructed from highly branched polymers for postsurgical management of choroidal melanoma, enabling localized tumor control and preservation of visual function; (B) Synthetic route of AVB hydrogel by a two-step process; (C) Representative photographs (upper panel) and corresponding hematoxylin and eosin (H&E)-stained histopathological sections (lower panel) of mouse eyes from each experimental group. Scale bar 1 mm. Figure 4A-C is quoted with permission from Chen et al.[155]. AVB: Artificial vitreous body; Mel: melphalan; MA: maleimide groups; SH: sulfhydryl groups; αPDL1: anti-programmed death ligand 1; IFN-g: interferon-g; TNF-α: tumor necrosis factor-α; DAMP: damage-associated molecular pattern; CTL: cytotoxic T lymphocyte; DC: dendritic cell; PEG: poly(ethylene glycol).
Beyond tumor control, stimuli-responsive hydrogels can act as transient scaffolds to support tissue repair and modulate postoperative wound healing. Gelatin-based and HA-based matrices are particularly attractive due to their biocompatibility and tunable degradation. Lin et al. recently developed a photopolymerizable hydrogel (GelDex-S58) to prevent scarring after glaucoma filtration surgery[156]. The GelDex network is formed via Schiff base crosslinking between GelMA and oxidized dextran (ODex), with S58 aptamers encapsulated within the hydrogel matrix. S58 exhibited sustained in vitro release for approximately 20 days, while the hydrogel degraded in vivo over roughly 35 days, which matches the critical postoperative healing window. In a rabbit model of glaucoma filtration surgery, GelDex-S58 significantly reduced scar formation, prolonged the functional lifetime of filtration blebs, and improved overall surgical success rates[156]. Together, these advances highlight stimuli-responsive hydrogels as multifunctional intraocular depots that couple spatiotemporally controlled drug release with structural support. Such a system could enhance tumor eradication, modulate postoperative immunity, limit scarring, and ultimately improve long-term ocular surgical outcomes.
Local immunotherapy via stimuli-responsive hydrogels
Ocular tumors, particularly UM, are characterized by a highly immunosuppressive microenvironment that limits the efficacy of systemic immunotherapies, largely due to the blood-ocular barrier and poor T-cell infiltration. To overcome these challenges, stimuli-responsive hydrogels have emerged as promising local delivery platforms capable of providing sustained intraocular release and retention of immunomodulatory agents or cellular therapies. These systems establish a new paradigm for treating ocular tumors by enabling precise spatiotemporal control over immune activation. For instance, hydrogels can be loaded with immune checkpoint inhibitors such as αPD-1/programmed death ligand 1 (PD-L1) and anti-cytotoxic T-lymphocyte-associated protein 4 (αCTLA-4) antibodies, facilitating prolonged local release while minimizing systemic immune-related adverse effects such as dermatitis, colitis, and endocrine disorders[157,158]. A notable example is the Tetra-PEG AVB hydrogel developed by Chen et al., which enables programmed release of αPDL1, resulting in robust activation of Cluster of Differentiation 8-positive (CD8+) T cells, significant suppression of postoperative tumor recurrence, induction of systemic immune memory, and inhibition of contralateral metastasis[155].
Chimeric antigen receptor T-cell (CAR-T) immunotherapy represents another promising immunotherapeutic approach for Rb, though its application is hindered by short intravitreal persistence, immunosuppressive conditions, and T-cell dysfunction[159]. To address these limitations, Wang et al. designed a thermosensitive chitosan-PEG hydrogel for co-encapsulation of disialoganglioside (GD2)-specific CAR-T cells and IL-15[160]. Upon injection and in situ gelation, this system forms a three-dimensional matrix that supports T-cell viability and functionality by retaining autocrine IL-15 and shielding cells from rapid clearance. Notably, while standalone GD2. CAR-T cells only transiently suppressed tumor growth, hydrogel-encapsulated GD2. CAR-Ts releasing IL-15 (GD2. CAR.15-T) cells achieved complete tumor control, prevented recurrence, and significantly preserved retinal structure and visual function. The hydrogel played a critical role in enhancing T-cell migration, sustaining IL-15 release, and reducing intraocular inflammation and retinal damage, underscoring its potential as a versatile platform for local cellular immunotherapy. This supports clinical translation given the established safety of chitosan-PEG hydrogels and regulatory familiarity with CAR-T platforms.
Multimodal therapeutic strategies via stimuli-responsive hydrogels
Multimodal therapy leveraging stimuli-responsive hydrogels has emerged as a promising strategy to overcome the limitations of single-modality treatments, including tumor heterogeneity, drug resistance, and immune evasion. These hydrogels act as versatile co-delivery platforms that integrate chemotherapy, PTT, gene therapy, and immunotherapy, enabling spatiotemporally controlled and mechanistically complementary interventions. For example, Wang et al. developed an injectable thermosensitive hydrogel (collagen-based hydrogel with cyclodextrin aldehyde, CCA) formed via Schiff-base crosslinking of collagen with aldehyde-modified cyclodextrin [Figure 5A][161]. This CCA system co-delivered oncolytic adenovirus and melphalan, providing sustained local release and thereby maintaining prolonged intraocular therapy and preserving vision in Rb.
Figure 5. Stimuli-responsive hydrogels for combinational therapy of ocular tumors. (A) Schematic of an injectable composite hydrogel co-encapsulating an oncolytic adenovirus and melphalan for retinoblastoma treatment. Figure 5A is quoted with permission from Wang et al.[161] under the CC BY-NC-ND license without modification; (B) Fabrication and near-infrared light-induced gel-sol transition of the CP@Au@DC_AC50 hydrogel for uveal melanoma therapy; (C) Multifunctional properties of the CP@Au@DC_AC50 hydrogel, integrating gene-targeted therapy, mild photothermal therapy, and antibacterial activity. Figure 5B and C are quoted with permission from Wang et al.[164]. Col: Collagen; Mel: melphalan; CDA: β-cyclodextrin aldehyde; OA: oncolytic adenovirus; RB: retinoblastoma; CP: chitosan@puerarin; GNR: gold nanorod; ATOX1: antioxidant 1 Copper Chaperone; NIR: near-infrared; PTT: photothermal therapy; DC_AC50: a small-molecule inhibitor of ATOX1; CCA: collagen-based hydrogel with cyclodextrin aldehyde
In addition, incorporation of photothermal agents further enables precise tumor ablation under NIR laser irradiation, offering a minimally invasive alternative or adjunct to surgery while synergizing with other therapeutic modalities[162]. A representative example is the NIR-responsive liposome/hydrogel hybrid system (QD Lipo/ICG/LAgel) developed by Lin et al., which co-delivered quercetin (QUE), DOX, and the photosensitizer ICG for synergistic chemotherapy and mild PTT in Rb[163]. Upon NIR exposure, ICG-generated heat induces a gel-sol transition, facilitating the release of QD Lipo. QUE further enhances therapeutic efficacy by inhibiting HSP70 and epithelial-mesenchymal transition (EMT) proteins, thereby increasing thermal sensitivity and suppressing metastasis. In vivo, this system achieved prolonged intravitreal drug retention, marked tumor growth inhibition, minimal retinal toxicity, and a favorable safety profile, underscoring its potential for pediatric Rb treatment[163].
Similarly, Wang et al. constructed an injectable thermosensitive hydrogel (CP@Au@DC_AC50) based on chitosan-puerarin, loaded with GNRs and the ATOX1 (antioxidant 1 Copper Chaperone) inhibitor DC_AC50, for combined photothermal and gene therapy in UM[164]. NIR irradiation triggered gel dissolution, DC_AC50 release, and simultaneous PTT activation, resulting in effective gene silencing and hyperthermia [Figure 5B and C]. In vivo studies showed rapid suppression of tumor progression within two weeks, improved ocular parameters, and no evidence of intraocular infection or structural damage, confirming its antitumor efficacy and biosafety[164]. In another study, Xie et al. reported a manganese-coordinated polymer hydrogel composed of HA and CMCS coordinated polymer nanoparticles (CP2) loaded with DOX (HA/CMCS-CP2@DOX) for controlled DOX delivery[165]. NIR irradiation induced the localized heating, promoting drug release and enhancing cytotoxicity against OCM-1 UM cells, with notable tumor-targeting capability. Drug release was further accelerated in the acidic TME (pH 6.5), reaching approximately 75% within 6 h[165].
Tumor-specific design considerations for ocular tumors
Across the preceding sections, stimuli-responsive hydrogels have been discussed in a largely modality-oriented manner. For clinical translation, however, it is essential to recognize that UM and Rb differ markedly in microenvironmental features, anatomical niches, and therapeutic goals. These distinctions demand tumor-specific engineering principles rather than a uniform hydrogel design.
Anatomical localization and delivery route considerations
UM arises from melanocytes within the vascular choroid or ciliary body, located outside the BRB but in close continuity with the scleral vasculature[166]. This positioning favors transscleral or suprachoroidal hydrogel delivery, which permits direct drug deposition at the tumor site while limiting exposure of the neurosensory retina[167,168]. Hydrogels for UM therefore should be engineered to optimize transport across the sclera and to distribute efficiently within the retro-equatorial space. In practice, this requires tunable mechanical properties that allow the material to traverse tissue planes and conform to the globe curvature without inducing retinal detachment or choroidal effusion.
By contrast, Rb arises from the developing neural retina and is often confined to the intravitreal compartment, thus making intravitreal injection the primary delivery route for hydrogel systems[169]. The small vitreous volume in pediatric eyes (approximately 4.0 mL) imposes stringent restrictions on injectable volume, typically below 100 μL per dose, to avoid acute intraocular pressure elevation[7]. In addition, Rb commonly manifests vitreous seeding, where tumor cell clusters float within the vitreous gel. Thus, hydrogels intended for Rb should either exhibit anti-floating or anchoring behavior or enable controlled dispersion. This feature allows therapeutic agents to coat and eradicate seed nodules without aggregating, obscuring the visual axis, or perturbing vitreoretinal integrity[170,171].
Tumor microenvironment-guided stimulus selection
Differences in tumor metabolism and stromal remodeling between UM and Rb inform the choice of stimuli and the design of responsive motifs. UM is characterized by a distinctly acidic microenvironment (pH 5.4-6.5) and high expression of MMP-2/9, which correlates with invasive behavior and metastatic risk[172,173]. Consequently, pH-responsive hydrogels incorporating acid-labile Schiff-base linkages, together with MMP-cleavable peptide crosslinkers, are therefore particularly suitable for UM[114]. Such systems can undergo preferential degradation within the tumor bed, enhancing intra-tumoral drug release while reducing off-target diffusion and systemic leakage.
In contrast, Rb typically exhibits profound hypoxia, especially within avascular vitreous seeds, with marked overexpression of hypoxia-inducible factor 1-alpha (HIF-1α) and metabolic adaptation to low oxygen tension[174,175]. At the same time, Rb is generally an immunologically “cold” tumor, with minimal baseline CD8+ T-cell infiltration and low PD-L1 expression, converting to a more “hot” phenotype only after cytotoxic chemotherapy[176]. These features suggest that hypoxia-responsive hydrogels, particularly when loaded with immune-stimulatory agents or adjuvants, may be more rational for Rb than pH-responsive systems, given the near-physiological pH of the vitreous cavity. In this setting, hypoxia-triggered release could be leveraged to remodel the immune landscape and potentiate local immunochemotherapy while preserving surrounding healthy ocular tissues.
Patient demographics and biocompatibility requirements
UM predominantly arises in middle-aged to elderly adults (mean age 55-60 years), which permits greater flexibility in material selection[177]. Semi-permanent hydrogel implants or long-acting depots that persist for several months (e.g., thermosensitive PNIPAM-based systems) may be advantageous for sustaining therapeutic intraocular drug levels[178]. Nevertheless, adult UM patients typically need to preserve high-quality vision for driving and professional activities. Accordingly, hydrogels should exhibit excellent optical clarity, low haze, and minimal light-scattering to avoid glare, reduced contrast sensitivity, or other visual disturbances.
In contrast, Rb occurs mainly in infants and young children (median age at diagnosis ~ 18 months), whose ocular tissues and visual pathways are still undergoing rapid development[179]. This demographic imposes stricter biocompatibility criteria on hydrogel design. First, complete and timely biodegradation is essential to prevent chronic foreign body reactions that could disrupt ocular growth, refractive development, or retinal maturation. Second, material composition should be stringently controlled to exclude developmental neurotoxins (e.g., residual acrylamide monomers, or heavy-metal-based crosslinkers) that may impair neurocognitive outcomes in germline RB1 mutation carriers. Third, sterilization protocols should ensure sterility without compromising network integrity, mechanical stability, or drug release characteristics, while providing a high safety margin for immunologically immature pediatric patients[180,181].
Divergent therapeutic objectives and risk profiles
In UM, therapeutic objectives extend beyond local tumor eradication to the prevention of hepatic metastasis, which develops in up to 50% of patients even after successful globe-sparing treatment[182]. Thus, hydrogel systems for UM should be engineered to support local-systemic crosstalk, for example by enabling sustained release of immune checkpoint inhibitors (e.g., αPD-1, or αCTLA-4 antibody) or adjuvants that promote tumor antigen presentation and systemic antitumor immunity targeting micro-metastatic liver disease[155]. In addition, these formulations should be compatible with standard radiotherapy regimens, such as iodine-125 plaque brachytherapy. Under irradiation, they should retain structural stability and controlled release, while avoiding radiation-induced premature degradation or aggravated inflammatory responses.
For Rb, the principal goals are globe preservation, functional vision retention, and reduction of the lifetime risk of secondary malignancies, particularly osteosarcoma in patients with germline RB1 mutations[18,183]. Thus, hydrogel-based chemotherapy facilitates metronomic dosing by providing sustained low-dose drug release over several weeks. This approach maintains effective intraocular cytotoxic levels while reducing systemic peak concentrations, thereby minimizing risks of myelosuppression and secondary tumor formation. Additionally, special caution is warranted when incorporating DNA-damaging agents (e.g., DOX, or alkylating drugs), as hereditary Rb is characterized by intrinsic defects in DNA damage surveillance and repair[184,185]. In this setting, hydrogel platforms should allow precise control over dose intensity, and cumulative exposure to balance effective tumor control with long-term safety.
In summary, stimuli-responsive hydrogels have extended their role in ocular oncology beyond sustained drug delivery to encompass multimodal therapy, immunomodulation, postoperative reconstruction, and functional preservation. The convergence of materials science with immune-engineering is enabling the development of personalized, targeted, and multifunctional intraocular therapeutic platforms. Collectively, these systems offer clinically feasible, vision-sparing strategies that reshape conventional paradigms, radiotherapy, chemotherapy, surgery, and immunotherapy, and lay a robust foundation for efficient tumor control while preserving ocular structure and function [Table 1].
Stimuli-responsive hydrogels for ocular tumor treatment
| Stimulus type | Composition | Gelation | Injection route | Ocular tumor | Ref |
| pH (6.5) -responsive | DDIIIOH-NH2; Conbercept; DOX | Physical crosslinking | Peritumoral injection | Retinoblastoma | [44] |
| pH (5.0), enzyme (hyaluronidase)-responsive | HA-DOX/LAP NPs; ALG-DPA; Ca2+ | Physical crosslinking | Peritumoral injection | Uveal melanoma | [114] |
| pH (5.3) -responsive | G5-acetal-melphalan; PEG-DA | Chemical crosslinking | Intratumoral injection | Retinoblastoma | [147] |
| Thermo (37 °C) -responsive | Topotecan; Poloxamer 407 | Physical crosslinking | Intratumoral injection | Retinoblastoma | [148] |
| Thermo (37 °C)-responsive | Collagen II; HA; 8- arm PEG succinimidyl glutarate; Curcumin-loaded nanoparticles; Sucrose | Physical crosslinking | Intravitreal injection | Uveal melanoma | [149] |
| Redox (GSH), thermo (32 °C)-responsive | RENPs; ICG; PNIPAM; BAC; DOX; FA | Chemical crosslinking | Intravenous injection | Choroidal melanoma | [150] |
| Thermo (40 °C)-responsive | Gelatin; PVA; PLGA-DOX; PEGDA; Fe3O4 | Chemical crosslinking | Intravitreal injection | Retinoblastoma | [151] |
| Thermo (37 °C)-responsive | Chitosan; PEG; NHS; β-glycerophosphate | Physical crosslinking | Subretinal injection | Retinoblastoma | [160] |
| Thermo (37 °C), pH-responsive | Collagen; CDA; OA; Melphalan | Chemical crosslinking | Intravitreal injection | Retinoblastoma | [161] |
| Redox (GSH ≥ 1 mM), thermo (56 °C)-responsive | NIPAM; BAC; MCNs/Ln/GD; FA; R8 | Chemical crosslinking | Intravitreal injection | Uveal melanoma | [162] |
| Light (808 nm), thermo (45-46 °C)-responsive | LAgel; ICG; QUE/DOX lipo | Physical crosslinking | Intravitreal injection | Retinoblastoma | [163] |
| Light (808 nm), thermo (42-47 °C)-responsive | Chitosan; Puerarin; Gold nanorods; DC_AC50 | Physical crosslinking | Intravitreal injection | Uveal melanoma | [164] |
| pH (6.5), photothermal (480 nm; 50°C)-responsive | CP2; HA; CMCS; DOX | Physical crosslinking | Intratumoral injection | Uveal melanoma | [165] |
CONCLUSION AND OUTLOOK
Stimuli-responsive hydrogels represent a transformative platform for the treatment of ocular tumors, offering targeted and controllable drug delivery. These “smart” biomaterials respond to defined internal or external cues to achieve precise spatiotemporal release of therapeutic agents. By bypassing or modulating physiological barriers such as the blood-ocular barrier, they enhance local drug concentration, reduce systemic exposure, and minimize off-target toxicity. Their application spans multiple therapeutic modalities, including chemotherapy, immunotherapy, and perioperative adjuncts, positioning them as promising platforms for vision-sparing ocular tumor therapy. Collectively, these features position stimuli-responsive hydrogels as promising platforms for vision-sparing ocular tumor therapy.
Recent preclinical studies highlight the strong translational potential of stimuli-responsive hydrogel systems for ocular tumor therapy. Platforms integrating CAR-T cell therapy, oncolytic adenoviruses, or thermosensitive chemotherapeutic formulations have achieved effective tumor suppression, extended intraocular retention, and preservation of retinal architecture in orthotopic models. Moreover, single-administration sustained-release hydrogels provide antitumor efficacy comparable to that of repeated conventional injections while markedly reducing systemic toxicity. These findings underscore the ability of hydrogel-based delivery systems to redefine current treatment strategies.
Translation challenges
Despite this promise, the clinical translation of stimuli-responsive hydrogels faces several critical challenges. Long-term biocompatibility and safety remain paramount, necessitating a thorough examination of potential risks within the closed, fragile, and non-regenerative intraocular milieu. Specific concerns include the chronic toxicity and immunogenicity of polymer components and their degradation products, which could adversely affect sensitive structures such as retinal ganglion cells, photoreceptors, and the lens. The intravitreal injection of hydrogel formulations creates a space-occupying lesion in a compartment with limited compliance, risking mechanical compression that may compromise choroidal blood flow and retinal function[186,187]. Furthermore, prolonged hydrogel residence could instigate foreign body reactions, leading to granulomatous inflammation, fibrous encapsulation, and progressive neurodegeneration. Documented sequelae include severe retinal degeneration and epiretinal membrane formation, driven by chronic macrophage activation and pro-inflammatory cytokine release[180,188]. The health of the retina, terminally differentiated neural tissue with minimal regenerative capacity, is particularly vulnerable to these sustained insults, including oxidative stress from hydrogel components[189]. Additionally, the presence or degradation of hydrogels may disrupt aqueous humor dynamics, posing a potential risk to intraocular pressure regulation. Achieving precise implantation and controlled localization within specific ocular compartments presents additional technical hurdles, while significant interpatient variability in endogenous stimulus profiles complicates the development of universally effective and standardized formulations.
Future perspectives
Looking ahead, stimuli-responsive hydrogels have the potential to reshape ocular oncology by enabling intelligent, spatially controlled, and adaptive drug delivery. Future work should focus on several directions. First, the design of multi-stimuli-responsive systems (for example, dual pH/enzyme- or light/temperature-responsive hydrogels) may improve targeting precision, reduce premature activation, and increase disease specificity. Second, rationally engineered combination strategies, such as chemo-photothermal, radio-chemo, or immune-hydrogel approaches, could produce synergistic antitumor effects and promote durable local and systemic immune responses, while enabling dynamic treatment adaptation. Third, robust preclinical evaluation in physiologically relevant models, including large animal models and patient-derived xenograft (PDX) systems incorporating ocular tumor organoids, will be essential to improve the predictive value of efficacy and safety data. In parallel, systematic optimization of production scalability, sterilization protocols, degradation kinetics, and long-term intraocular biocompatibility will be required to support regulatory approval and clinical adoption.
In conclusion, stimuli-responsive hydrogels are driving a shift in ocular tumor management from traditional excision-based strategies toward precision therapies. Although significant barriers remain, these systems offer substantial potential to improve outcomes for patients with ocular malignancies by enabling intelligent, spatially controlled, and adaptive drug delivery.
DECLARATIONS
Acknowledgments
Figures 1 (https://BioRender.com/2ktdiv3) and 2 (https://BioRender.com/p3im2n2) are created with the assistance of Biorender. com. All reproduced figures have been used under the necessary permissions from the respective copyright holders.
Authors’ contributions
Writing-original draft: Han, D.
Visualization: Han, D.; Peng, H.
Conceptualization: Han, D.; Qian, J.; Zhou, X.; Luo, Z.
Supervision: Chan, S. Y.; Qian, J.; Zhou, X.; Luo, Z.
Writing-review & editing: Luo, Z.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Availability of data and materials
During the preparation of this manuscript, the AI tool DeepSeek was used exclusively for language polishing. The tool did not contribute to the scientific content of the study. All authors accept full responsibility for the integrity of the research and the final text.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant No. 82572418), the EYE & ENT Hospital of Fudan University Talents Program (Grant no. yn23039 and yn25002).
Conflicts of interest
Luo Z. is a Senior Editorial Board Member of the journal Micro Nano Science. Luo Z. was not involved in any steps of the editorial process, including reviewers’ selection, manuscript handling, and decision-making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
2. Ahmed, S.; Amin, M. M.; Sayed, S. Ocular drug delivery: a comprehensive review. AAPS. PharmSciTech. 2023, 24, 66.
3. Khan, S.; Do, C. W.; Ho, E. A. Recent updates on drug delivery approaches for improved ocular delivery with an insight into nanostructured drug delivery carriers for anterior and posterior segment disorders. Drug. Deliv. Transl. Res. 2025, 15, 1828-76.
4. Han, H.; Li, S.; Xu, M.; et al. Polymer- and lipid-based nanocarriers for ocular drug delivery: current status and future perspectives. Adv. Drug. Delivery. Rev. 2023, 196, 114770.
5. Subrizi, A.; Del Amo, E. M.; Korzhikov-Vlakh, V.; Tennikova, T.; Ruponen, M.; Urtti, A. Design principles of ocular drug delivery systems: importance of drug payload, release rate, and material properties. Drug. Discov. Today. 2019, 24, 1446-57.
6. Onugwu, A. L.; Nwagwu, C. S.; Onugwu, O. S.; et al. Nanotechnology based drug delivery systems for the treatment of anterior segment eye diseases. J. Control. Release. 2023, 354, 465-88.
7. Bhandari, A. Ocular fluid mechanics and drug delivery: a review of mathematical and computational models. Pharm. Res. 2021, 38, 2003-33.
8. Wen, J. C.; Reina-Torres, E.; Sherwood, J. M.; et al. Intravitreal anti-VEGF injections reduce aqueous outflow facility in patients with neovascular age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 2017, 58, 1893-8.
9. Alshaikh, R. A.; Waeber, C.; Ryan, K. B. Polymer based sustained drug delivery to the ocular posterior segment: barriers and future opportunities for the treatment of neovascular pathologies. Adv. Drug. Delivery. Rev. 2022, 187, 114342.
10. Cerna-Chavez, R.; Rozanska, A.; Poretti, G. L.; Benvenisty, N.; Parulekar, M.; Lako, M. Retinal pigment epithelium exhibits gene expression and phagocytic activity alterations when exposed to retinoblastoma chemotherapeutics. Exp. Eye. Res. 2023, 233, 109542.
11. Kim, H.; Roh, H.; Kim, S. H.; Lee, K.; Im, M.; Oh, S. J. Effective protection of photoreceptors using an inflammation-responsive hydrogel to attenuate outer retinal degeneration. NPJ. Regen. Med. 2023, 8, 68.
12. Williams BK, J. R.; Di Nicola, M. Ocular oncology-primary and metastatic malignancies. Med. Clin. North. Am. 2021, 105, 531-50.
13. Nishath, T.; Stacey, A. W.; Steinberg, D.; et al. Global retinoblastoma study group. retinoblastoma survival and enucleation outcomes in 41 countries from the African continent. Br. J. Ophthalmol. 2024, 109, 64-9.
14. Yilmaz, M. T.; Sari, S. Y.; Zorlu, F.; Yazici, G. External beam radiotherapy in the management of uveal melanoma. Curr. Treat. Options. Oncol. 2024, 25, 932-51.
15. Kaliki, S.; Shields, C. L.; Rojanaporn, D.; et al. Scleral necrosis after plaque radiotherapy of uveal melanoma: a case-control study. Ophthalmology 2013, 120, 1004-11.
16. Kovács, G. SP-0035 The role of interstitial radiotherapy in orbital tumors or extrascleral uveal melanoma. Radiother. Oncol. 2021, 158, S21.
17. Shields, C. L.; Suribhatla, R.; Romero, M.; et al. Tissue glue-assisted plaque radiotherapy for choroidal tumors with scleral thinning. Retina 2025, 45, 675-82.
18. Grümme, L.; Biewald, E.; Reschke, M.; et al. Comparing efficacy and side effects of two systemic chemotherapy regimens for eye-preserving therapy in children with retinoblastoma. Pediatr. Blood. Cancer. 2022, 69, e29362.
19. Grzybowski, A.; Told, R.; Sacu, S.; et al. ; Euretina Board. 2018 Update on intravitreal injections: Euretina expert consensus recommendations. Ophthalmologica. 2018, 239, 181-93.
20. Biewald, E. M.; Bornfeld, N.; Metz, K. A.; et al. Histopathology of retinoblastoma eyes enucleated after intra-arterial chemotherapy. Br. J. Ophthalmol. 2020, 104, 1171-5.
21. Ramos, M. S.; Xu, L. T.; Singuri, S.; et al. Patient-reported complications after intravitreal injection and their predictive factors. Ophthalmol. Retina. 2021, 5, 625-32.
22. Da, M.; Li, K. K.; Chan, K. C.; Wu, E. X.; Wong, D. S. Distribution of triamcinolone acetonide after intravitreal injection into silicone oil-filled eye. Biomed. Res. Int. 2016, 2016, 5485467.
23. Kasiński, A.; Zielińska-Pisklak, M.; Oledzka, E.; Sobczak, M. Smart hydrogels - synthetic stimuli-responsive antitumor drug release systems. Int. J. Nanomedicine. 2020, 15, 4541-72.
24. Li, S.; Chen, L.; Fu, Y. Nanotechnology-based ocular drug delivery systems: recent advances and future prospects. J. Nanobiotechnol. 2023, 21, 232.
25. Rumon, M. M. H.; Akib, A. A.; Sarkar, S. D.; et al. Polysaccharide-based hydrogels for advanced biomedical engineering applications. ACS. Polym. Au. 2024, 4, 463-86.
26. Feng, Y.; Zhang, Z.; Tang, W.; Dai, Y. Gel/hydrogel-based in situ biomaterial platforms for cancer postoperative treatment and recovery. Exploration. (Beijing). 2023, 3, 20220173.
27. Yu, C.; Xu, J.; Heidari, G.; et al. Injectable hydrogels based on biopolymers for the treatment of ocular diseases. Int. J. Biol. Macromol. 2024, 269, 132086.
28. Shao, H.; Yin, K.; Xu, N.; et al. Adaptive surfaces with stimuli-responsive wettability: from tailoring to applications. ACS. Nano. 2025, 19, 6729-47.
29. Solanki, R.; Bhatia, D. Stimulus-responsive hydrogels for targeted cancer therapy. Gels 2024, 10, 440.
30. Sabbagh, F.; Zargarian, S. S.; Kosik-Kozioł, A.; Nakielski, P.; Pierini, F. Hydrogel-based ocular drug delivery systems. J. Mater. Chem. B. 2025, 13, 14982-5006.
31. Torres-Luna, C.; Fan, X.; Domszy, R.; Hu, N.; Wang, N. S.; Yang, A. Hydrogel-based ocular drug delivery systems for hydrophobic drugs. Eur. J. Pharm. Sci. 2020, 154, 105503.
32. Kalairaj, M. S.; Pradhan, R.; Saleem, W.; Smith, M. M.; Gaharwar, A. K. Intra-articular injectable biomaterials for cartilage repair and regeneration. Adv. Healthc. Mater. 2024, 13, e2303794.
33. Kaur, H.; Gogoi, B.; Sharma, I.; et al. Hydrogels as a potential biomaterial for multimodal therapeutic applications. Mol. Pharm. 2024, 21, 4827-48.
34. Lin, D.; Lei, L.; Shi, S.; Li, X. Stimulus-responsive hydrogel for ophthalmic drug delivery. Macromol. Biosci. 2019, 19, e1900001.
35. Khattak, S.; Ullah, I.; Sohail, M.; et al. Endogenous/exogenous stimuli‐responsive smart hydrogels for diabetic wound healing. Aggregate 2024, 6, e688.
36. Guo, L.; Fu, Z.; Li, H.; et al. Smart hydrogel: a new platform for cancer therapy. Adv. Colloid. Interface. Sci. 2025, 340, 103470.
37. Manjeri, A.; George, S. D. Hydrogel-embedded polydimethylsiloxane contact lens for ocular drug delivery. ACS. Appl. Bio. Mater. 2024, 7, 7324-31.
38. Chen, Y.; Yan, Y.; Wei, W. Research advances of matrix metalloproteinases family in uveal melanoma. Eur. J. Med. Res. 2025, 30, 609.
39. Cheng, X.; Xu, H. D.; Ran, H. H.; Liang, G.; Wu, F. G. Glutathione-depleting nanomedicines for synergistic cancer therapy. ACS. Nano. 2021, 15, 8039-68.
40. Luo, Z.; Wu, Q.; Yang, C.; et al. A powerful CD8+ T-Cell stimulating D-tetra-peptide hydrogel as a very promising vaccine adjuvant. Adv. Mater. 2017, 29.
41. Anemone, A.; Consolino, L.; Arena, F.; Capozza, M.; Longo, D. L. Imaging tumor acidosis: a survey of the available techniques for mapping in vivo tumor pH. Cancer. Metastasis. Rev. 2019, 38, 25-49.
42. Alizadeh, H.; Ahmadi, M.; Heydari Shayesteh, O. On chip synthesis of a pH sensitive gefitinib anticancer drug nanocarrier based on chitosan/alginate natural polymers. Sci. Rep. 2024, 14, 772.
43. Wang, Y.; Wang, J.; Yuan, Z.; et al. Chitosan cross-linked poly(acrylic acid) hydrogels: drug release control and mechanism. Colloids. Surf. B. Biointerfaces. 2017, 152, 252-9.
44. Fan, W.; Chen, M.; Raza, F.; et al. pH-sensitive peptide hydrogel encapsulating the anti-angiogenesis drug conbercept and chemotherapeutic drug dox as a combination therapy for retinoblastoma. Mater. Adv. 2024, 5, 2175-84.
45. Chu, S.; Shi, X.; Tian, Y.; Gao, F. pH-responsive polymer nanomaterials for tumor therapy. Front. Oncol. 2022, 12, 855019.
46. Peng, S.; Xiao, F.; Chen, M.; Gao, H. Tumor-microenvironment-responsive nanomedicine for enhanced cancer immunotherapy. Adv. Sci. (Weinh). 2022, 9, e2103836.
47. Zhang, J.; Su, L.; Liu, Z.; et al. A responsive hydrogel modulates innate immune cascade fibrosis to promote ocular surface reconstruction after chemical injury. J. Control. Release. 2024, 365, 1124-38.
48. Zhou, J.; Wang, H.; Chen, H.; et al. pH-responsive nanocomposite hydrogel for simultaneous prevention of postoperative adhesion and tumor recurrence. Acta. Biomater. 2023, 158, 228-38.
49. Zhu, J.; Zhang, X.; Ai, L.; Yuan, R.; Ye, J. Clinicohistopathological implications of MMP/VEGF expression in retinoblastoma: a combined meta-analysis and bioinformatics analysis. J. Transl. Med. 2019, 17, 226.
50. Cui, Q.; Wang, X.; Zhang, Y.; Shen, Y.; Qian, Y. Macrophage-derived MMP-9 and MMP-2 are closely related to the rupture of the fibrous capsule of hepatocellular carcinoma leading to tumor invasion. Biol. Proced. Online. 2023, 25, 8.
51. Sobczak, M. Enzyme-responsive hydrogels as potential drug delivery systems-state of knowledge and future prospects. Int. J. Mol. Sci. 2022, 23.
52. Zhang, Q.; Hu, W.; Guo, M.; et al. MMP-2 responsive peptide hydrogel-based nanoplatform for multimodal tumor therapy. Int. J. Nanomedicine. 2024, 19, 53-71.
53. Lee, S.; Hong, H. K.; Song, J. S.; et al. Intravitreal injectable hydrogel rods with long-acting bevacizumab delivery to the retina. Acta. Biomater. 2023, 171, 273-88.
54. Chen, L.; Yan, D.; Wu, N.; et al. Injectable bio-responsive hydrogel for therapy of inflammation related eyelid diseases. Bioact. Mater. 2021, 6, 3062-73.
55. Estrela, J. M.; Ortega, A.; Obrador, E. Glutathione in cancer biology and therapy. Crit. Rev. Clin. Lab. Sci. 2006, 43, 143-81.
56. Liu, H.; Deng, Z.; Li, T.; et al. Fabrication, GSH-responsive drug release, and anticancer properties of thioctic acid-based intelligent hydrogels. Colloids. Surf. B. Biointerfaces. 2022, 217, 112703.
57. Hsu, P. H.; Almutairi, A. Recent progress of redox-responsive polymeric nanomaterials for controlled release. J. Mater. Chem. B. 2021, 9, 2179-88.
58. Xu, Z.; Yang, W.; Zhang, R.; et al. Microenvironment-programmed siRNA-based hydrogel for spatiotemporal gene silencing in wound healing. Adv. Mater. 2025, 37, e09558.
59. Zhang, R.; Nie, T.; Fang, Y.; Huang, H.; Wu, J. Poly(disulfide)s: from synthesis to drug delivery. Biomacromolecules 2022, 23, 1-19.
60. Li, F.; Ding, J.; Li, Z.; Rong, Y.; He, C.; Chen, X. ROS-responsive thermosensitive polypeptide hydrogels for localized drug delivery and improved tumor chemoimmunotherapy. Biomater. Sci. 2024, 12, 3100-11.
61. Xu, H.; Cao, W.; Zhang, X. Selenium-containing polymers: promising biomaterials for controlled release and enzyme mimics. Acc. Chem. Res. 2013, 46, 1647-58.
62. Abed, H. F.; Abuwatfa, W. H.; Husseini, G. A. Redox-responsive drug delivery systems: a chemical perspective. Nanomaterials. (Basel). 2022, 12, 3183.
63. Zhou, J.; Fang, C.; Rong, C.; Luo, T.; Liu, J.; Zhang, K. Reactive oxygen species-sensitive materials: a promising strategy for regulating inflammation and favoring tissue regeneration. Smart. Mater. Med. 2023, 4, 427-46.
64. Mathew, J. Ru-hung, .; Le, T.; et al. ROS-responsive camptothecin-linked thioketal drug delivery system based on ring-closing polymerization. Eur. Polym. J. 2024, 202, 112646.
65. Yang, M.; Chen, X.; Chen, Z.; et al. Thermoresponsive antioxidant metal-free carbon nanodot hydrogel: An effective therapeutic approach for ocular surface disease. Sci. Adv. 2025, 11, eadt8775.
66. Chang, R.; Wang, X.; Li, X.; An, H.; Qin, J. Self-activated healable hydrogels with reversible temperature responsiveness. ACS. Appl. Mater. Interfaces. 2016, 8, 25544-51.
67. Tuszynska, M.; Skopinska-Wisniewska, J.; Bartniak, M.; Bajek, A. Conceptualization and preliminary characterization of poloxamer-based hydrogels for biomedical applications. Bioconjugate. Chem. 2025, 36, 1169-79.
68. Levin, M.; Cohen, N. Thermo-mechanics of PNIPAM gels: from a single chain to a network response. Macromolecules 2025, 58, 5187-200.
69. Kopač, T. Mathematical model for characterization of temperature-responsive polymers: A study on the rheological behavior of gelatin and poly(N-isopropylacrylamide). Polym. Test. 2024, 133, 108402.
70. Balan, G. A.; Precupas, A.; Matei, I. Gelation behaviour of Pluronic F127/polysaccharide systems revealed via thioflavin T fluorescence. Gels 2023, 9, 939.
71. Shriky, B.; Mahmoudi, N.; Kelly, A.; Isreb, M.; Gough, T. The effect of PEO homopolymers on the behaviours and structural evolution of Pluronic F127 smart hydrogels for controlled drug delivery systems. Colloids. Surf. A. Physicochem. Eng. Asp. 2022, 645, 128842.
72. Wang, Q.; Zhao, X.; Yu, F.; et al. Photocurable and temperature-sensitive bioadhesive hydrogels for sutureless sealing of full-thickness corneal wounds. Small. Methods. 2024, 8, e2300996.
73. Luo, J.; Zhao, X.; Guo, B.; Han, Y. Preparation, thermal response mechanisms and biomedical applications of thermosensitive hydrogels for drug delivery. Expert. Opin. Drug. Deliv. 2023, 20, 641-72.
74. Annala, A.; Sadeghi, A.; Toropainen, E.; et al. Intravitreal sustained release of dexamethasone from a self-healing injectable hydrogel: an in vivo safety and release study. Mol. Pharm. 2025, 22, 6920-31.
75. Luo, Z.; Jin, L.; Xu, L.; et al. Thermosensitive PEG-PCL-PEG (PECE) hydrogel as an in situ gelling system for ocular drug delivery of diclofenac sodium. Drug. Delivery. 2016, 23, 63-8.
76. Yang, C.; Liao, X.; Zhou, K.; et al. Multifunctional nanoparticles and collagenase dual loaded thermosensitive hydrogel system for enhanced tumor-penetration, reversed immune suppression and photodynamic-immunotherapy. Bioact. Mater. 2025, 48, 1-17.
77. Li, L.; Scheiger, J. M.; Levkin, P. A. Design and applications of photoresponsive hydrogels. Adv. Mater. 2019, 31, e1807333.
78. Zhao, Y.; Ran, B.; Lee, D.; Liao, J. Photo-controllable smart hydrogels for biomedical application: a review. Small. Methods. 2024, 8, e2301095.
79. Pandey, T.; Pandey, V. A mechanistic understanding to photophysical phenomenon in development of near-infrared (NIR) responsive hydrogels: advancements in precision drug delivery. J. Drug. Deliv. Sci. Technol. 2025, 106, 106682.
80. Xing, Y.; Zeng, B.; Yang, W. Light responsive hydrogels for controlled drug delivery. Front. Bioeng. Biotechnol. 2022, 10, 1075670.
81. Ansari, A.; Bhowmik, S.; Zhang, K.; et al. A visible light-responsive hydrogel to study the effect of dynamic tissue stiffness on cellular mechanosensing. Adv. Funct. Mater. 2025, 35, 2501585.
82. Hao, Z. W.; Zhang, Z. Y.; Wang, Z. P.; et al. Bioactive peptides and proteins for tissue repair: microenvironment modulation, rational delivery, and clinical potential. Mil. Med. Res. 2024, 11, 75.
83. Pal, K.; Singh, S.; Itakura, S.; Hashimoto, M.; Kusamori, K.; Nishikawa, M. Reactive oxygen species augmented polydopamine-chlorin e6 nanosystem for enhanced chemo/photothermal/photodynamic therapy: a synergistic trimodal combination approach in vitro & in vivo. Int. J. Biol. Macromol. 2024, 283, 137236.
84. Liao, S.; Yue, W.; Cai, S.; et al. Improvement of gold nanorods in photothermal therapy: recent progress and perspective. Front. Pharmacol. 2021, 12, 664123.
85. Shuai, C.; Pan, G.; He, T.; Shuai, X.; Wu, P.; Zhong, Q. Photothermal effect of graphene oxide accelerate singlet oxygen release from artesunate to endow the scaffold with antibacterial properties. Mater. Today. Commun. 2024, 39, 109058.
86. Kumar, R.; Singh, S. K.; Srivastava, R. K.; Mallick, S.; Vankayala, R. NIR-responsive black phosphorus nanosheet-integrated niosomes for combinatorial chemo-phototherapy of cancers. ACS. Bio. Med. Chem. Au. 2025, 5, 143-53.
87. Wang, Z. J.; Li, C. Y.; Zhao, X. Y.; Wu, Z. L.; Zheng, Q. Thermo- and photo-responsive composite hydrogels with programmed deformations. J. Mater. Chem. B. 2019, 7, 1674-8.
88. Bordbar-Khiabani, A.; Gasik, M. Smart hydrogels for advanced drug delivery systems. Int. J. Mol. Sci. 2022, 23, 3665.
89. Hu, H.; Xu, Z.; Liu, J.; et al. Light-responsive hydrogel with nano-microstructures for bacteria eradication and tumor therapy. ACS. Appl. Nano. Mater. 2025, 8, 6242-54.
90. Wang, Z.; Chen, R.; Yang, S.; Li, S.; Gao, Z. Design and application of stimuli-responsive DNA hydrogels: A review. Mater. Today. Bio. 2022, 16, 100430.
91. Aria, H.; Rezaei, M. Immunogenic cell death inducer peptides: a new approach for cancer therapy, current status and future perspectives. Biomed. Pharmacother. 2023, 161, 114503.
92. Xu, Y.; Wei, C.; Ma, L.; et al. 3D mesenchymal stem cell exosome-functionalized hydrogels for corneal wound healing. J. Control. Release. 2025, 380, 630-46.
93. Duymaz, D. Karaoğlu, İ. C.; Kizilel, S. Effect of photoinitiation process on photo-crosslinking of gelatin methacryloyl hydrogel networks. Macromol. Rapid. Commun. 2025, 46, e00376.
94. Fan, X.; Shang, H.; Ji, L.; Ma, S.; Liao, M. Preparation and biomedical application of light-responsive hydrogels based on natural products. Front. Pharmacol. 2025, 16, 1714907.
95. Kwizera, E. A.; Stewart, S.; Mahmud, M. M.; He, X. Magnetic nanoparticle-mediated heating for biomedical applications. J. Heat. Transfer. 2022, 144, 030801.
96. Kang, M. A.; Fang, J.; Paragodaarachchi, A.; et al. Magnetically induced Brownian motion of iron oxide nanocages in alternating magnetic fields and their application for efficient siRNA delivery. Nano. Lett. 2022, 22, 8852-9.
97. Li, Z.; Li, Y.; Chen, C.; Cheng, Y. Magnetic-responsive hydrogels: from strategic design to biomedical applications. J. Control. Release. 2021, 335, 541-56.
98. Suhail, M. Alamgir; Wahab, A.; et al. Magnetically responsive hydrogel systems: fundamental features, emerging applications, and future horizons. Coord. Chem. Rev. 2025, 543, 216916.
99. Gong, J.; Hu, J.; Yan, X.; et al. Injectable hydrogels including magnetic nanosheets for multidisciplinary treatment of hepatocellular carcinoma via magnetic hyperthermia. Small 2024, 20, e2300733.
100. Ma, H.; Yu, G.; Cheng, J.; et al. Design of an injectable magnetic hydrogel based on the tumor microenvironment for multimodal synergistic cancer therapy. Biomacromolecules 2023, 24, 868-85.
101. Zhou, Y.; Liu, G.; Guo, S. Advances in ultrasound-responsive hydrogels for biomedical applications. J. Mater. Chem. B. 2022, 10, 3947-58.
102. Xiao, H.; Aliabouzar, M.; Fabiilli, M. L. Acoustically responsive scaffolds: unraveling release kinetics and mechanisms for sustained, steady drug delivery. J. Control. Release. 2024, 374, 205-18.
103. Meng, Z.; Zhang, Y.; She, J.; et al. Ultrasound-mediated remotely controlled nanovaccine delivery for tumor vaccination and individualized cancer immunotherapy. Nano. Lett. 2021, 21, 1228-37.
104. Naeem, A.; Yu, C.; Zhou, L.; et al. Shape memory hydrogels in tissue engineering: Recent advances and challenges. Bioact. Mater. 2025, 54, 215-47.
105. Gerayeli, F.; Khalef, N.; Bakri, A.; Benech, P.; Martin, D. K. Ultrasound-stimulated Brownian ratchet enhances diffusion of molecules retained in hydrogels. Nanomedicine 2021, 31, 102308.
106. Huang, H.; Dong, Z.; Ren, X.; et al. High-strength hydrogels: Fabrication, reinforcement mechanisms, and applications. Nano. Res. 2023, 16, 3475-515.
107. Kang Derwent, J. J.; Mieler, W. F. Thermoresponsive hydrogels as a new ocular drug delivery platform to the posterior segment of the eye. Trans. Am. Ophthalmol. Soc. 2008, 106, 206-13.
108. Zhu, B.; Zong, T.; Zheng, R.; et al. Acid and glutathione dual-responsive, injectable and self-healing hydrogels for controlled drug delivery. Biomacromolecules 2024, 25, 1838-49.
109. Zhang, Q.; Yan, K.; Zheng, X.; Liu, Q.; Han, Y.; Liu, Z. Research progress of photo-crosslink hydrogels in ophthalmology: a comprehensive review focus on the applications. Mater. Today. Bio. 2024, 26, 101082.
110. Abdelmohsen, H. A. M.; Copeland, N. A.; Hardy, J. G. Light-responsive biomaterials for ocular drug delivery. Drug. Deliv. Transl. Res. 2023, 13, 2159-82.
111. Yan, L.; Zhao, Z.; Liu, Y.; et al. The inverse electron demand Diels-alder (IEDDA): a facile bioorthogonal click reaction for development of injectable polysaccharide-based hydrogels for biomedical applications. Carbohydr. Polym. 2025, 352, 123142.
112. Morozova, S. M. Recent advances in hydrogels via Diels-alder crosslinking: design and applications. Gels 2023, 9, 102.
113. Sojdeh, S.; Panjipour, A.; Yaghmour, A.; Arabpour, Z.; Djalilian, A. R. Click chemistry-based hydrogels for tissue engineering. Gels 2025, 11, 724.
114. Guo, Z.; Xiu, L.; Li, Y.; et al. Injectable nanocomposite hydrogel with cascade drug release for treatment of uveal melanoma. J. Control. Release. 2024, 376, 1086-99.
115. Ali, I.; Gulfam, M.; Jo, S. H.; et al. Reduction-responsive and bioorthogonal carboxymethyl cellulose based soft hydrogels cross-linked via IEDDA click chemistry for cancer therapy application. Int. J. Biol. Macromol. 2022, 219, 109-20.
116. Ilochonwu, B. C.; van der Lugt, S. A.; Annala, A.; et al. Thermo-responsive Diels-Alder stabilized hydrogels for ocular drug delivery of a corticosteroid and an anti-VEGF fab fragment. J. Control. Release. 2023, 361, 334-49.
117. Yang, J.; Chen, Y.; Zhao, L.; Zhang, J.; Luo, H. Constructions and properties of physically cross-linked hydrogels based on natural polymers. Polym. Rev. 2022, 63, 574-612.
118. Zhang, T.; Ping, W.; Suo, M.; et al. Stimuli-responsive hydrogels potentiating photothermal therapy against cancer stem cell-induced breast cancer metastasis. ACS. Nano. 2024, 18, 20313-23.
119. Wu, Y.; Chang, X.; Yang, G.; et al. A physiologically responsive nanocomposite hydrogel for treatment of head and neck squamous cell carcinoma via proteolysis-targeting chimeras enhanced immunotherapy. Adv. Mater. 2023, 35, e2210787.
120. Liu, W. S.; Lu, Z. M.; Pu, X. H.; et al. A dendritic cell-recruiting, antimicrobial blood clot hydrogel for melanoma recurrence prevention and infected wound management. Biomaterials 2025, 313, 122776.
121. Gong, Y.; Yuan, W.; Liu, S.; et al. Physical encapsulation and chemotherapy: a synergistic hydrogel strategy for tumor suppression. Adv. Healthc. Mater. 2025, 14, e2500511.
122. Guan, T.; Chen, Z.; Wang, X.; et al. Harnessing Mn2+ ions and antitumor peptides: a robust hydrogel for enhanced tumor immunotherapy. J. Am. Chem. Soc. 2025, 147, 6523-35.
123. Raza, M. A.; Lim, Y. M.; Lee, S. W.; Seralathan, K. K.; Park, S. H. Synthesis and characterization of hydrogels based on carboxymethyl chitosan and poly(vinylpyrrolidone) blends prepared by electron beam irradiation having anticancer efficacy, and applications as drug carrier for controlled release of drug. Carbohydr. Polym. 2021, 258, 117718.
124. Elshahawy, M. F.; Mohamed, R. D.; Ali, A. E.; Raafat, A. I.; Ahmed, N. A. Electron beam irradiation developed cinnamon oil- (polyvinyl alcohol/gum tragacanth)/graphene oxide dressing hydrogels: antimicrobial and healing assessments. Int. J. Biol. Macromol. 2024, 277, 134384.
125. Cheng, Y. H.; Huang, H. P.; Chen, H. H. Mucoadhesive phenylboronic acid-grafted carboxymethyl cellulose hydrogels containing glutathione for treatment of corneal epithelial cells exposed to benzalkonium chloride. Colloids. Surf. B. Biointerfaces. 2024, 238, 113884.
126. Devi V K, A.; Shyam, R.; Palaniappan, A.; Jaiswal, A. K.; Oh, T. H.; Nathanael, A. J. Self-healing hydrogels: preparation, mechanism and advancement in biomedical applications. Polymers. (Basel). 2021, 13, 3782.
127. Duan, Y.; Ding, L.; Meng, X.; et al. A therapeutic strategy integrating ultrasound-guided microwave ablation with nanocomposite hydrogels to enhance autophagy and suppress tumor growth in hepatocellular carcinoma. Acta. Biomater. 2025, 198, 413-27.
128. Annala, A.; Ilochonwu, B. C.; Wilbie, D.; Sadeghi, A.; Hennink, W. E.; Vermonden, T. Self-healing thermosensitive hydrogel for sustained release of dexamethasone for ocular therapy. ACS. Polym. Au. 2023, 3, 118-31.
129. Stevanović, M.; Jović, M.; Filipović, N.; et al. Multifunctional nanomaterial-integrated hydrogels for sustained drug delivery: from synthesis and characterization to biomedical application. Gels 2025, 11, 892.
130. Li, Z.; Yang, L.; Zhang, D.; et al. Mussel-inspired “plug-and-play” hydrogel glue for postoperative tumor recurrence and wound infection inhibition. J. Colloid. Interface. Sci. 2023, 650, 1907-17.
131. Ge, J.; Caldona, E. B.; Advincula, R. C. Functional stimuli-responsive polymers on micro- and nano-patterned interfaces. Polymer 2025, 317, 127958.
132. Wang, X.; Wang, Q. Enzyme-laden bioactive hydrogel for biocatalytic monitoring and regulation. Acc. Chem. Res. 2021, 54, 1274-87.
133. Yin, W.; Ke, W.; Lu, N.; et al. Glutathione and reactive oxygen species dual-responsive block copolymer prodrugs for boosting tumor site-specific drug release and enhanced antitumor efficacy. Biomacromolecules 2020, 21, 921-9.
134. Wang, L.; Ye, C.; Xue, X.; et al. 3D-printed breast prosthesis that smartly senses and targets breast cancer relapse. Adv. Sci. (Weinh). 2024, 11, e2402345.
135. Prince, E.; Cruickshank, J.; Ba-Alawi, W.; et al. Biomimetic hydrogel supports initiation and growth of patient-derived breast tumor organoids. Nat. Commun. 2022, 13, 1466.
136. Xu, Z.; Liu, L.; Duan, J.; et al. A host/guest assembled hyaluronic acid-based supramolecular hydrogel with NIR-steered degradation capacity for enhanced tumor therapy through programmable drug release. Carbohydr. Polym. 2025, 368, 124098.
137. Liang, X.; Li, X.; Wu, R.; et al. Breaking the tumor chronic inflammation balance with a programmable release and multi-stimulation engineering scaffold for potent immunotherapy. Adv. Sci. (Weinh). 2024, 11, e2401377.
138. Luo, F. Q.; Xu, W.; Zhang, J. Y.; et al. An injectable nanocomposite hydrogel improves tumor penetration and cancer treatment efficacy. Acta. Biomater. 2022, 147, 235-44.
139. Peppas, N. A.; Merrill, E. W. Crosslinked poly(vinyl alcohol) hydrogels as swollen elastic networks. J. Appl. Polym. Sci. 2003, 21, 1763-70.
140. Ilochonwu, B. C.; Urtti, A.; Hennink, W. E.; Vermonden, T. Intravitreal hydrogels for sustained release of therapeutic proteins. J. Control. Release. 2020, 326, 419-41.
141. Chang, D.; Park, K.; Famili, A. Hydrogels for sustained delivery of biologics to the back of the eye. Drug. Discov. Today. 2019, 24, 1470-82.
142. Richbourg, N. R.; Peppas, N. A. The swollen polymer network hypothesis: quantitative models of hydrogel swelling, stiffness, and solute transport. Prog. Polym. Sci. 2020, 105, 101243.
143. Li, J.; Mooney, D. J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 1-17.
144. Campbell, K. T.; Wysoczynski, K.; Hadley, D. J.; Silva, E. A. Computational-based design of hydrogels with predictable mesh properties. ACS. Biomater. Sci. Eng. 2020, 6, 308-19.
145. Shirakura, T.; Smith, C.; Hopkins, T. J. J.; et al. Matrix density engineering of hydrogel nanoparticles with simulation-guided synthesis for tuning drug release and cellular uptake. ACS. Omega. 2017, 2, 3380-9.
146. Tram, N. K.; Maxwell, C. J.; Swindle-Reilly, K. E. Macro- and microscale properties of the vitreous humor to inform substitute design and intravitreal biotransport. Curr. Eye. Res. 2021, 46, 429-44.
147. Cooper, R. C.; Wang, J.; Yang, H. Injectable dendrimer hydrogel delivers melphalan in both conjugated and free forms for retinoblastoma. Biomacromolecules 2024, 25, 5928-37.
148. Huo, Y.; Wang, Q.; Liu, Y.; et al. A temperature-sensitive phase-change hydrogel of topotecan achieves a long-term sustained antitumor effect on retinoblastoma cells. Onco. Targets. Ther. 2019, 12, 6069-82.
149. Xie, L.; Yue, W.; Ibrahim, K.; Shen, J. A long-acting curcumin nanoparticle/in situ hydrogel composite for the treatment of uveal melanoma. Pharmaceutics 2021, 13, 1335.
150. Li, L.; Zeng, Z.; Chen, Z.; et al. Microenvironment-triggered degradable hydrogel for imaging diagnosis and combined treatment of intraocular choroidal melanoma. ACS. Nano. 2020, 14, 15403-16.
151. Kim, D. I.; Lee, H.; Kwon, S. H.; Sung, Y. J.; Song, W. K.; Park, S. Bilayer hydrogel sheet-type intraocular microrobot for drug delivery and magnetic nanoparticles retrieval. Adv. Healthc. Mater. 2020, 9, e2000118.
152. Taich, P.; Moretton, M. A.; Del Sole, M. J.; et al. Sustained-release hydrogels of topotecan for retinoblastoma. Colloids. Surf. B. Biointerfaces. 2016, 146, 624-31.
153. Cocarta, A. I.; Hobzova, R.; Sirc, J.; et al. Hydrogel implants for transscleral drug delivery for retinoblastoma treatment. Mater. Sci. Eng. C. Mater. Biol. Appl. 2019, 103, 109799.
154. Hobzova, R.; Kodetova, M.; Pochop, P.; et al. Hydrogel implants for transscleral diffusion delivery of topotecan: In vivo proof of concept in a rabbit eye model. Int. J. Pharm. 2021, 606, 120832.
155. Chen, M.; Hu, J.; Gao, H.; et al. An immunotherapeutic artificial vitreous body hydrogel to control choroidal melanoma and preserve vision after vitrectomy. Sci. Adv. 2023, 9, eadh1582.
156. Lin, Y.; Luo, W.; Jiang, B.; et al. The effect of GelDex-S58 hydrogel on anti-conjunctival scarring after glaucoma filtration surgery. iScience 2023, 26, 107633.
157. Wang, C.; Wang, J.; Zhang, X.; et al. In situ formed reactive oxygen species-responsive scaffold with gemcitabine and checkpoint inhibitor for combination therapy. Sci. Transl. Med. 2018, 10, eaan3682.
158. Song, H.; Su, Q.; Nie, Y.; et al. Supramolecular assembly of a trivalent peptide hydrogel vaccine for cancer immunotherapy. Acta. Biomater. 2023, 158, 535-46.
160. Wang, K.; Chen, Y.; Ahn, S.; et al. GD2-specific CAR T cells encapsulated in an injectable hydrogel control retinoblastoma and preserve vision. Nat. Cancer. 2020, 1, 990-7.
161. Wang, G.; Wu, K.; Zhang, Z.; et al. Injectable hydrogel-mediated co-delivery of oncolytic adenovirus and melphalan for retinoblastoma control and vision preservation. Acta. Pharm. Sin. B. 2025, 15, 6638-56.
162. Yang, M.; Huang, Y.; Chen, Z.; et al. Synthetic carbon-based lanthanide upconversion nanoparticles for enhanced photothermal therapy. Nat. Commun. 2025, 16, 6343.
163. Lin, M.; Liu, X.; Li, J.; et al. Hybrid NIR-responsive liposome/hydrogel platform mediating chemo-photothermal therapy of retinoblastoma enhanced by quercetin as an adjuvant. Theranostics 2025, 15, 3995-4015.
164. Wang, S.; Chen, B.; Ouyang, L.; et al. A novel stimuli-responsive injectable antibacterial hydrogel to achieve synergetic photothermal/gene-targeted therapy towards uveal melanoma. Adv. Sci. (Weinh). 2021, 8, e2004721.
165. Xie, P.; He, J.; Ou, Y. Metal complex hydrogel as a dual functional photothermal platform for in vitro doxorubicin delivery in ocular melanoma treatment. Polym. Adv. Technol. 2025, 36, e70136.
166. Jager, M. J.; Shields, C. L.; Cebulla, C. M.; et al. Uveal melanoma. Nat. Rev. Dis. Primers. 2020, 6, 24.
167. Chiang, B.; Jung, J. H.; Prausnitz, M. R. The suprachoroidal space as a route of administration to the posterior segment of the eye. Adv. Drug. Delivery. Rev. 2018, 126, 58-66.
168. Wu, K. Y.; Fujioka, J. K.; Gholamian, T.; Zaharia, M.; Tran, S. D. Suprachoroidal injection: a novel approach for targeted drug delivery. Pharmaceuticals. (Basel). 2023, 16, 1241.
169. Ghassemi, F.; Shields, C. L.; Ghadimi, H.; Khodabandeh, A.; Roohipoor, R. Combined intravitreal melphalan and topotecan for refractory or recurrent vitreous seeding from retinoblastoma. JAMA. Ophthalmol. 2014, 132, 936-41.
170. Riazi-Esfahani, H.; Masoomian, B.; Khodabandeh, A.; et al. Addition of intravitreal carboplatin with melphalan for management of vitreous seeding in retinoblastoma. Graefes. Arch. Clin. Exp. Ophthalmol. 2023, 261, 1167-75.
171. Galardi, A.; Colletti, M.; Lavarello, C.; et al. Proteomic profiling of retinoblastoma-derived exosomes reveals potential biomarkers of vitreous seeding. Cancers. (Basel). 2020, 12, 1555.
172. Andreucci, E.; Peppicelli, S.; Ruzzolini, J.; et al. The acidic tumor microenvironment drives a stem-like phenotype in melanoma cells. J. Mol. Med. (Berl). 2020, 98, 1431-46.
173. Wang, T.; Zhang, Y.; Bai, J.; Xue, Y.; Peng, Q. MMP1 and MMP9 are potential prognostic biomarkers and targets for uveal melanoma. BMC. Cancer. 2021, 21, 1068.
174. Sudhakar, J.; Venkatesan, N.; Lakshmanan, S.; Khetan, V.; Krishnakumar, S.; Biswas, J. Hypoxic tumor microenvironment in advanced retinoblastoma. Pediatr. Blood. Cancer. 2013, 60, 1598-601.
175. Sradhanjali, S.; Tripathy, D.; Rath, S.; Mittal, R.; Reddy, M. M. Overexpression of pyruvate dehydrogenase kinase 1 in retinoblastoma: a potential therapeutic opportunity for targeting vitreous seeds and hypoxic regions. PLoS. ONE. 2017, 12, e0177744.
176. Miracco, C.; Toti, P.; Gelmi, M. C.; et al. Retinoblastoma is characterized by a cold, CD8+ cell poor, PD-L1- microenvironment, which turns into hot, CD8+ cell rich, PD-L1+ after chemotherapy. Invest. Ophthalmol. Vis. Sci. 2021, 62, 6.
177. Bai, H.; Bosch, J. J.; Heindl, L. M. Current management of uveal melanoma: a review. Clin. Exp. Ophthalmol. 2023, 51, 484-94.
178. Hu, J.; Liu, X.; Gao, Q.; Ouyang, C.; Zheng, K.; Shan, X. Thermosensitive PNIPAM-based hydrogel crosslinked by composite nanoparticles as rapid wound-healing dressings. Biomacromolecules 2023, 24, 1345-54.
179. Wu, K. Y.; Wang, X. C.; Anderson, M.; Tran, S. D. Advancements in nanosystems for ocular drug delivery: a focus on pediatric retinoblastoma. Molecules 2024, 29, 2263.
180. Wang, T.; Ran, R.; Ma, Y.; Zhang, M. Polymeric hydrogel as a vitreous substitute: current research, challenges, and future directions. Biomed. Mater. 2021, 16, 042012.
181. Lin, Q.; Lim, J. Y. C.; Xue, K.; Su, X.; Loh, X. J. Polymeric hydrogels as a vitreous replacement strategy in the eye. Biomaterials 2021, 268, 120547.
182. Huibers, A.; Wong, A.; Burgmans, M.; et al. Management of liver metastases from uveal melanoma. Br. J. Surg. 2025, 112, znaf130.
183. Farhat, W.; Yeung, V.; Ross, A.; et al. Advances in biomaterials for the treatment of retinoblastoma. Biomater. Sci. 2022, 10, 5391-429.
184. Zang, C.; Tian, Y.; Tang, Y.; et al. Hydrogel-based platforms for site-specific doxorubicin release in cancer therapy. J. Transl. Med. 2024, 22, 879.
185. Elsawy, M. A.; Wychowaniec, J. K.; Castillo Díaz, L. A.; Smith, A. M.; Miller, A. F.; Saiani, A. Controlling doxorubicin release from a peptide hydrogel through fine-tuning of drug-peptide fiber interactions. Biomacromolecules 2022, 23, 2624-34.
186. Qu, S.; Tang, Y.; Ning, Z.; Zhou, Y.; Wu, H. Desired properties of polymeric hydrogel vitreous substitute. Biomed. Pharmacother. 2024, 172, 116154.
187. Chan, K. C.; Yu, Y.; Ng, S. H.; et al. Intracameral injection of a chemically cross-linked hydrogel to study chronic neurodegeneration in glaucoma. Acta. Biomater. 2019, 94, 219-31.
188. Booler, H.; Larsen, T.; Shelton, A.; Bansteev, V.; Mitra, M. S. Foreign body reaction, retinal degeneration, and epiretinal membranes associated with intravitreal administration of PLGA rods. Toxicol. Pathol. 2021, 49, 656-62.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data



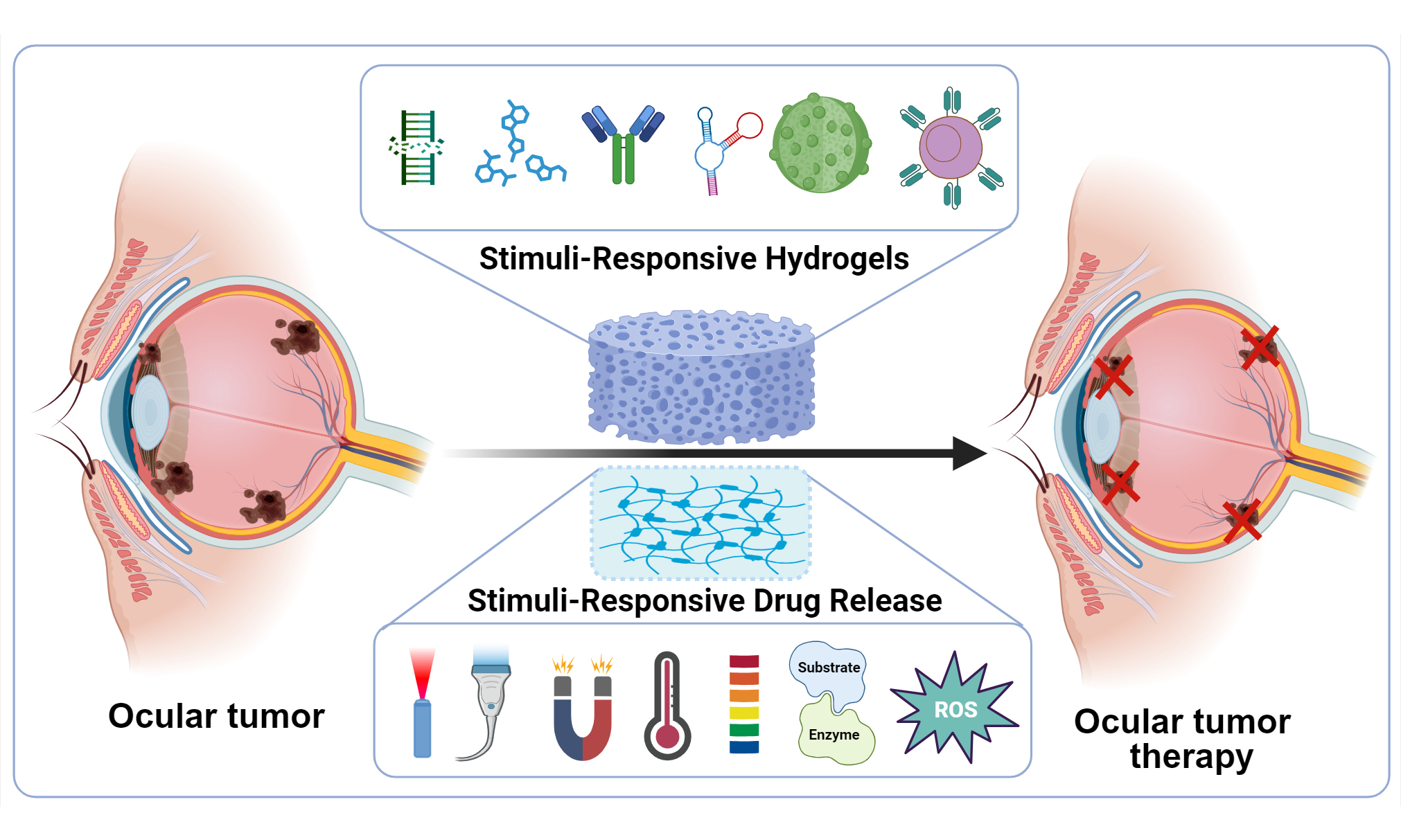
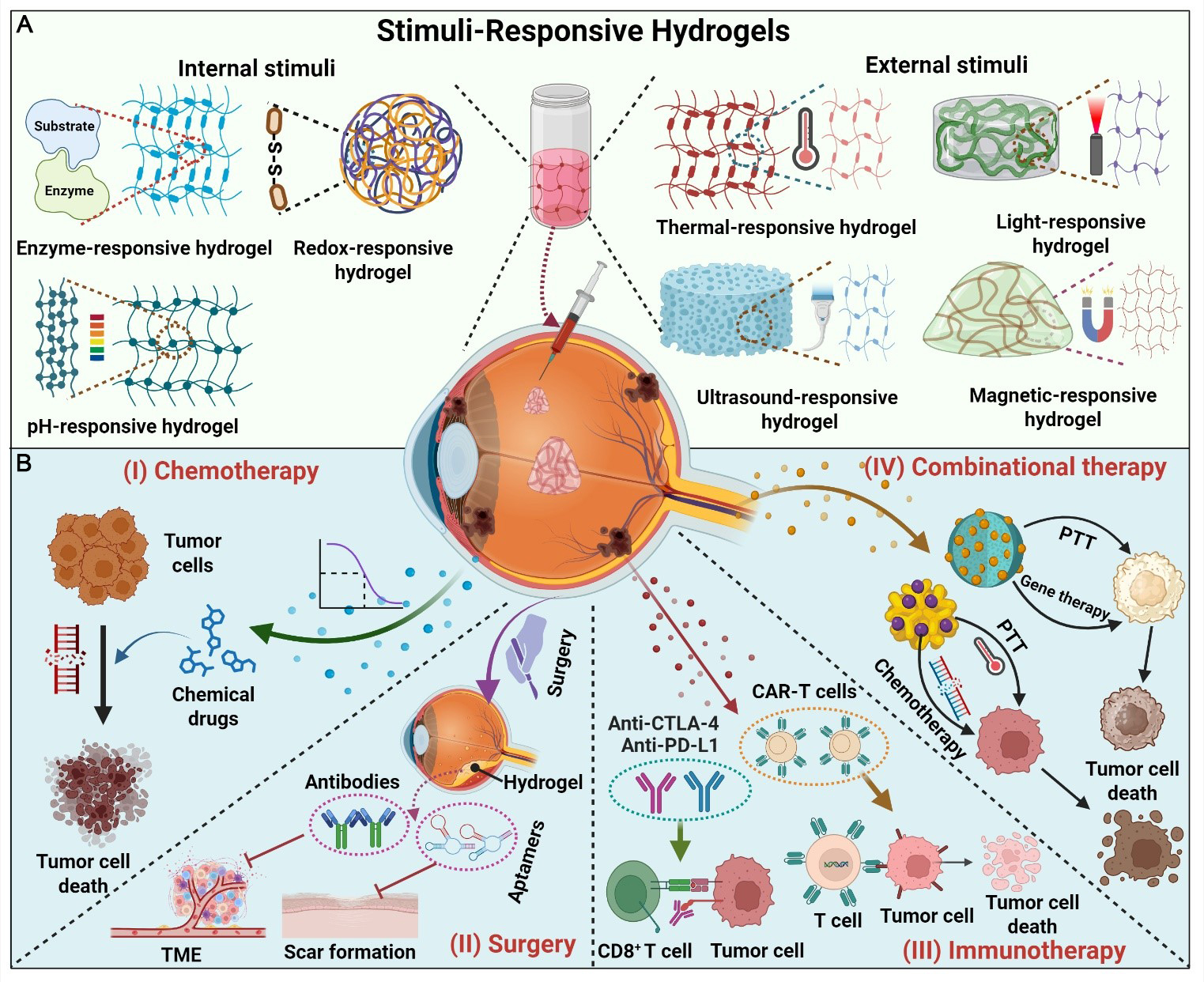
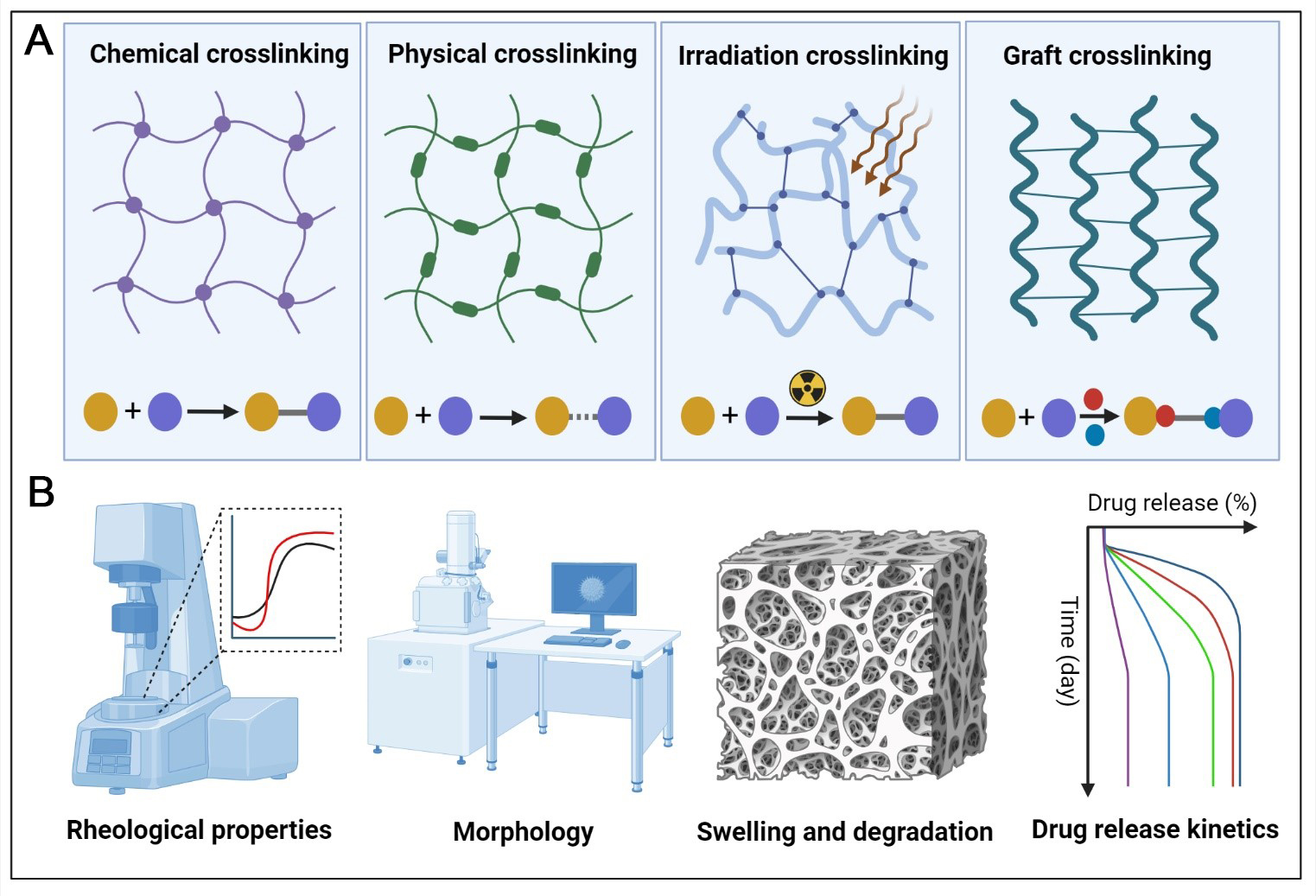
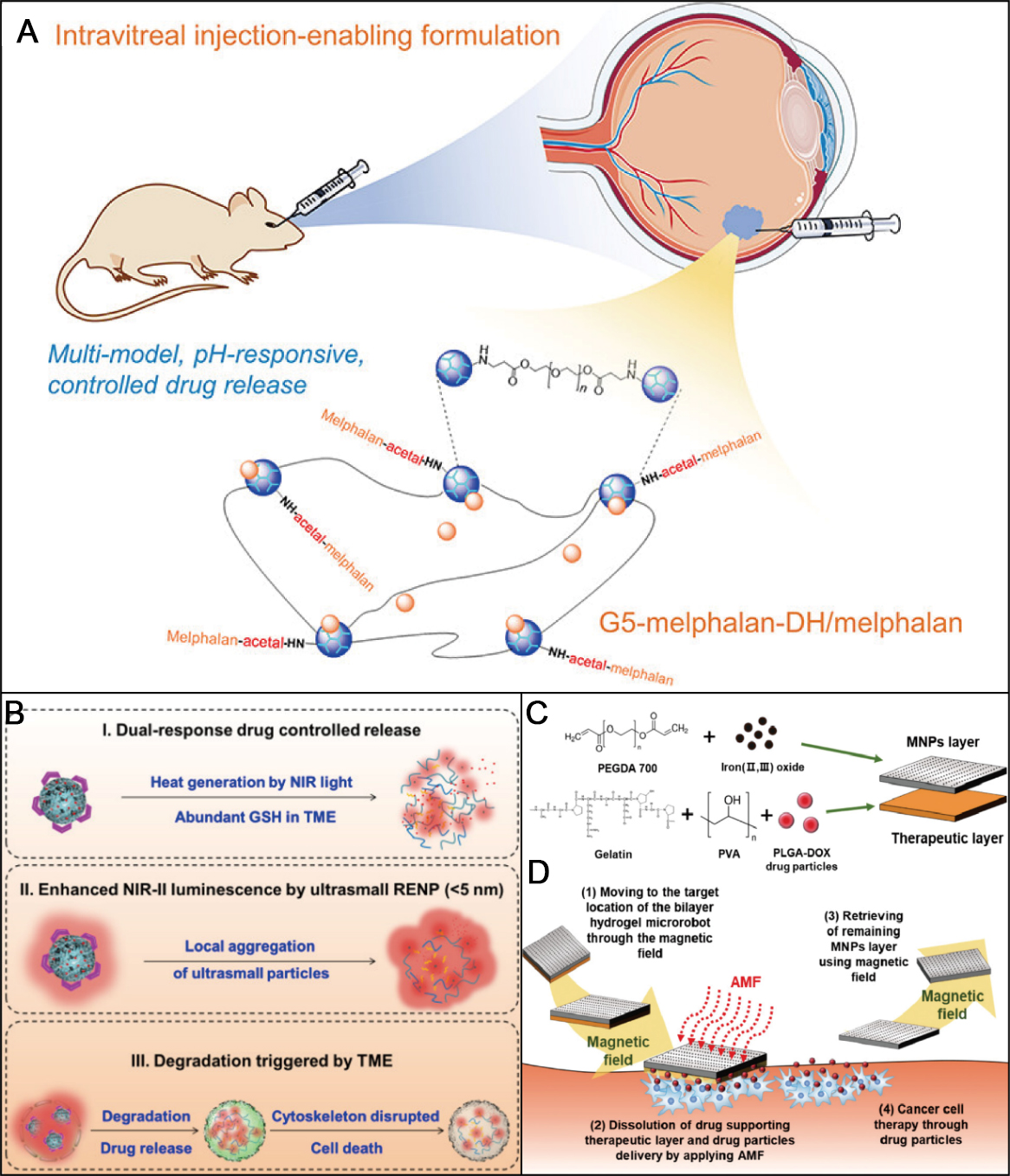
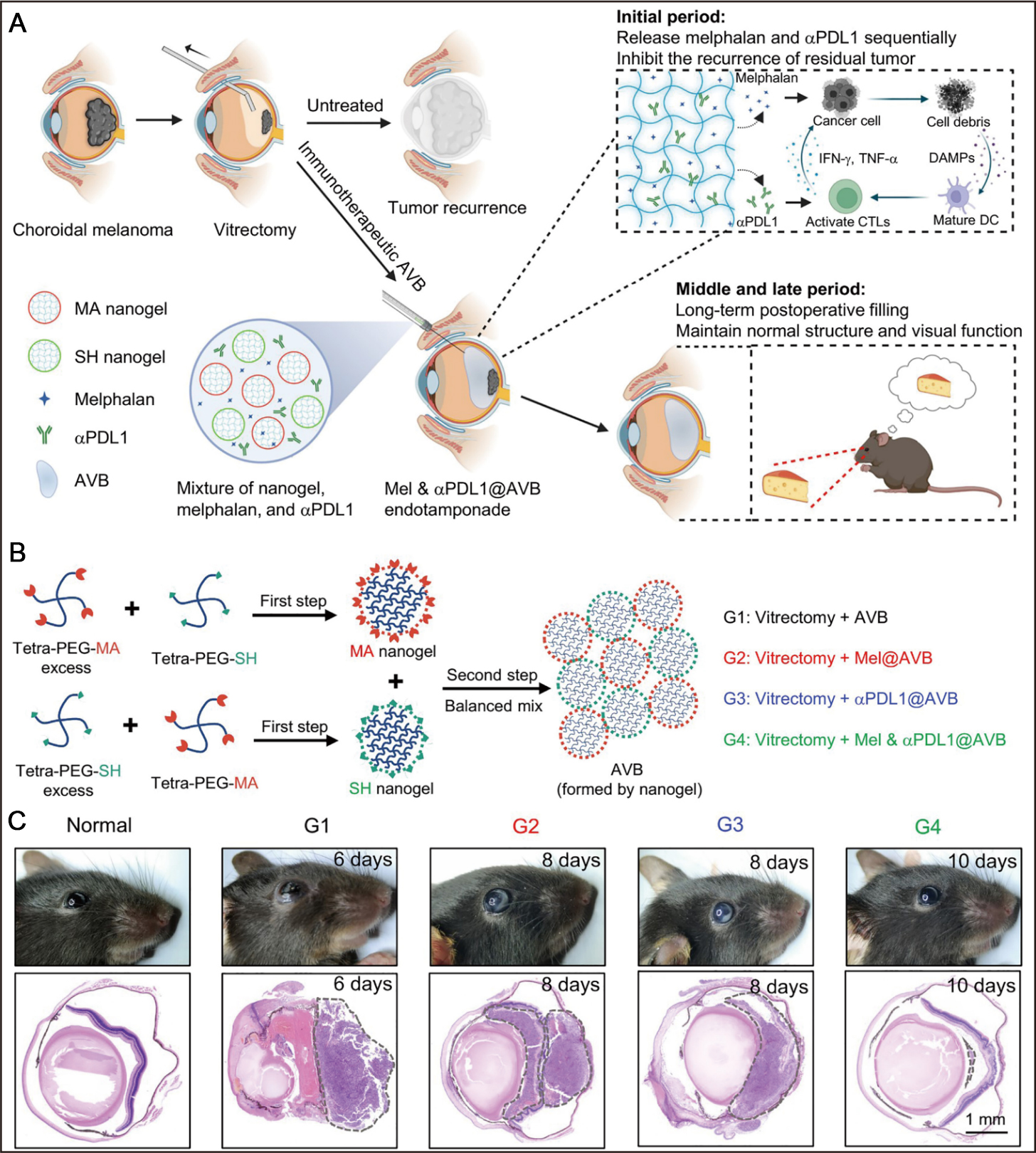
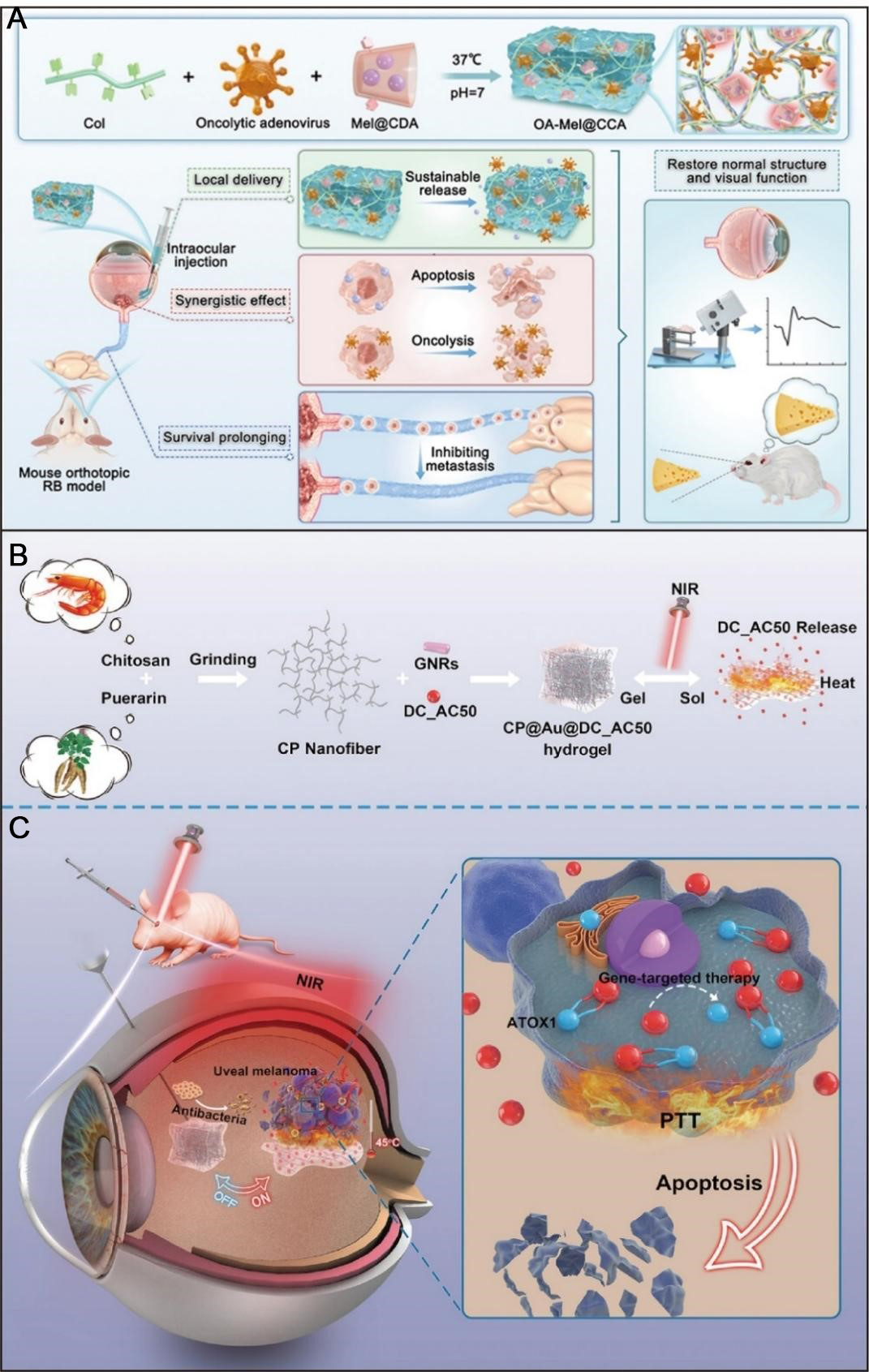








Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.