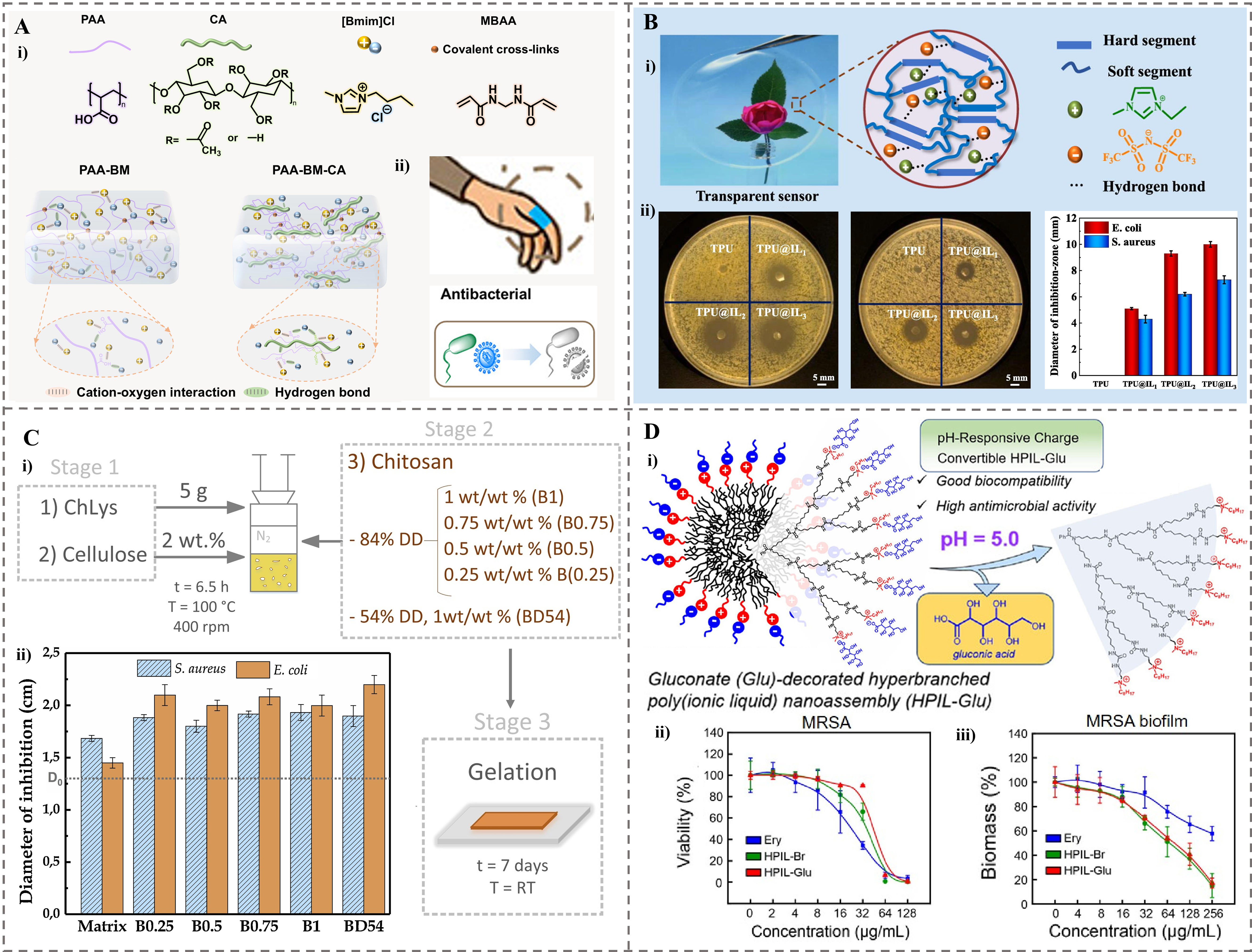
fig4
Figure 4. The ionogels were used as antibacterial agents. (A) Wearable ionogels with antibacterial effects. (i) Molecular structure of raw materials. (ii) Schematic illustration of interwoven networks of different samples[74]. Copyright 2025, Elsevier B.V; (B) Transparent antibacterial TPU@IL ionogels. (i) Optical transparency and schematic structure. (ii) Photos and measured diameters of the inhibition zones of TPU@IL ionogels with different mass ratios for E. coli and S. aureus[38]. Copyright 2021, Elsevier B.V; (C) A fully bio-based bionogel. (i) The formulation of bionogels reinforced with chitosan. (ii) Diameters of inhibition obtained from the antibacterial tests for S. aureus and E. coli[128]. Copyright 2019, Elsevier Ltd; (D) pH-responsive charge conversion feature of HPIL-Glu with high antimicrobial activity. (i) Schematic illustration of the responsiveness. (ii) Concentration-dependent bactericidal effect of HPIL-Br, HPIL-Glu, and erythromycin against planktonic MRSA bacteria. (iii) Concentration-dependent MRSA biofilm eradication effect of HPIL-Br, HPIL-Glu, and erythromycin[129]. Copyright 2024, American Chemical Society. TPU: Thermoplastic polyurethane; IL: ionic liquid; E. coli: Escherichia coli; S. aureus: Staphylococcus aureus; HPIL-Glu: Glu-decorated hyperbranched poly(ionic liquid); HPIL-Br: quaternary ammonium bromide-tethered HPIL; MRSA: methicillin-resistant Staphylococcus aureus; PAA: poly(acrylic acid); CA: cellulose acetate; MBAA: N, N′-methylenebis (acrylamide); PAA-BM: ionogels made by acrylic acid and [BMIM]Cl ionic liquid; PAA-BM-CA: ionogels made by acrylic acid, [BMIM]Cl ionic liquid and cellulose acetate; DD: degree of deacetylation; RT: room temperature.