fig2
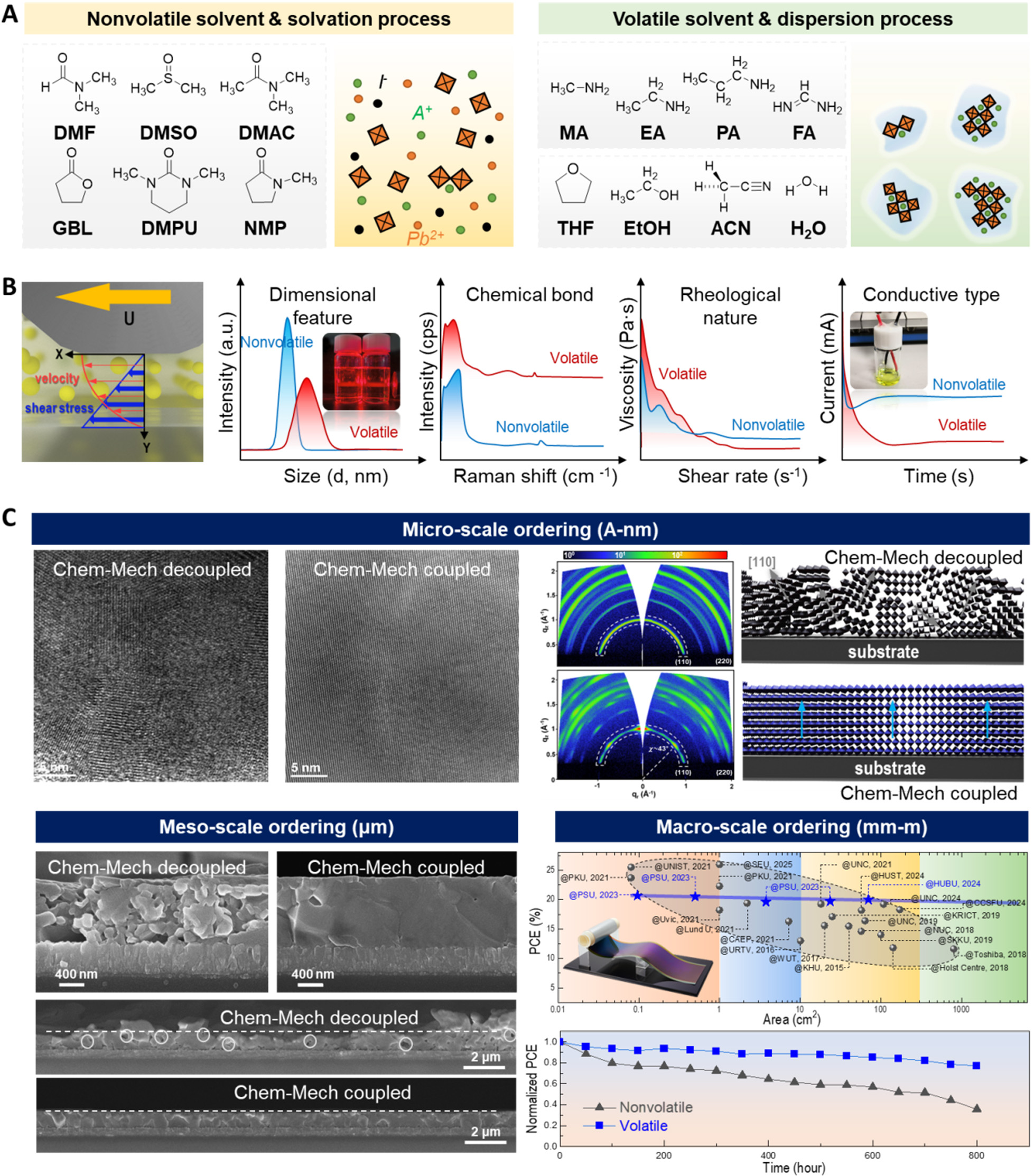
Figure 2. (A) Comparison of solvent systems used in perovskite precursor inks. Left: non-volatile Lewis-base solvents illustrated with a schematic of the ionization-solvation process leading to stable Pb-solvent adducts. Right: volatile/reactive solvent systems illustrated with the formation of transient, gel-like intermediates composed of dynamic colloidal dispersions; (B) Lateral distribution of mechanical shear stress during blade shearing and contrasting responses of volatile versus non-volatile precursor systems. From left to right: dimensional characteristics (DLS spectra), dominant chemical interactions (Raman spectra), rheological behavior (dynamic viscosity vs. shear-rate curves), and charge-transport characteristics (current-time transients)[35]. Reprinted with permission. Copyright 2024, Springer Nature; (C) Multi-scale ordering enabled by chemical-mechanical coupling compared with chemically dominated (decoupled) crystallization. Top left: HRTEM images showing nanoscale lattice coherence. Top right: 2D GIWAXS patterns with schematic illustrations of orientational differences. Bottom left: cross-sectional SEM images revealing mesoscale grain alignment and film densification. Bottom right: PCE-area scalability of state-of-the-art perovskite modules and long-term operational stability of devices fabricated from volatile versus non-volatile precursor systems[35]. Reprinted with permission. Copyright 2024, Springer Nature. DLS: Dynamic light scattering; HRTEM: high-resolution transmission electron microscopy; 2D: two-dimensional; GIWAXS: grazing-incidence wide-angle X-ray scattering; SEM: scanning electron microscopy; PCE: power conversion efficiency.