Intelligent fibers and self-powered wearable devices for ophthalmic applications: a frontier review
Abstract
The combination of intelligent fibers (IFs) and self-powered technologies provides new opportunities for wearable systems to interface with precision organs such as the eye and brain. In contrast to conventional ophthalmic diagnostics, which typically rely on bulky, externally powered equipment and offer only intermittent measurements, IF-based wearable devices leverage inherent flexibility, biocompatibility, and multifunctionality to support the minimally invasive, long-term in vivo monitoring and neural modulation. From a clinical perspective, the coupling of fiber design with autonomous energy strategies is central to enabling closed-loop ophthalmic platforms that operate without continuous external support. In this review, we examine recent progress across fiber materials, structural design, power solutions, and system-level integration, with emphasis on breakthroughs that have enabled applications including dynamic intraocular pressure monitoring, tear fluid biochemical analysis, visual function restoration, and neural interfaces. We further discuss the remaining challenges and emerging trends related to biocompatibility, energy autonomy, scalable manufacturing, and clinical translation, providing a forward-looking perspective on the development of next-generation IF-based diagnostic and therapeutic platforms.
Keywords
INTRODUCTION
The in-depth integration of the Internet of Things (IoT), artificial intelligence (AI), and biomedical engineering is transforming flexible wearable devices from simple peripheral accessories into new functional platforms that can easily integrate with the human body to achieve continuous health monitoring[1] and accurate medical intervention[2-5]. The next generation of biomedical electronics faces major challenges in ophthalmology, and there is an urgent need to develop new diagnostic and therapeutic platforms that are minimally invasive and can function in the body for extended periods[6]. The mechanical property mismatch between traditional ophthalmic devices and ocular tissues not only affects patient comfort but also compromises the continuity of monitoring data[7]. Besides, the limited energy supply restricts device functionality, lifetime, and clinical applicability.
In this context, intelligent fibers (IFs) offer distinct advantages in ophthalmic applications. Their inherent flexibility enables conformal adaptation to the curved ocular surface, while their permeability supports essential metabolic exchange for corneal tissue. The one-dimensional geometry of fibers further affords a practical route for minimally invasive intervention[8]. More importantly, through deliberate material selection and structural design, IFs can achieve multifunctional integration while ensuring biocompatibility, thereby providing a viable foundation for prolonged contact with or implantation within ocular tissues[9,10].
This review concentrates on the advances in two prominent frontiers in ophthalmics: IFs devices and self-powered systems. Here, the “intelligent fibers” describes the capability of a device to detect physiological signals, execute adaptive responses such as regulated drug delivery or optical modulation, and operate within a closed-loop manner. In parallel, the adoption of self-powered technologies is fundamentally reshaping the design of ophthalmic wearable devices[11]. By harvesting energy from biological sources, including ocular movement[12], ocular temperature[13], or tear fluid[14,15], these devices have the potential to overcome limitations of conventional batteries, such as size constraints, periodic replacement, and safety concerns. However, it is crucial to note that most of these self-powered approaches are currently at a low-to-mid Technology Readiness Level (TRL), primarily demonstrated in controlled laboratory settings or in simplified ex vivo models. A significant translational challenge lies in the fact that the microwatt-level, intermittent power generated by these harvesters is often insufficient to directly and continuously drive a complete operational cycle, which includes high-fidelity sensing, on-board processing, and robust wireless data transmission that typically requires milliwatt-level peak power. Therefore, achieving genuine long-term, dynamic monitoring necessitates not only advancements in harvesting efficiency but also revolutionary progress in ultra-low-power electronics, intelligent power management, and system-level energy budget co-design[16]. It is evident that the convergence of intelligent functionality and self-power represents a critical step toward realizing next-generation, self-sustaining ophthalmic platforms, offering the potential for enhanced patient comfort, extended device lifetime, and truly continuous, real-time physiological monitoring[17].
Current research in ophthalmology is shaped by well-defined clinical demands. Continuous intraocular pressure (IOP) monitoring is critical for improving glaucoma care[18-20], while real-time analysis of tear composition offers valuable information for dry eye syndrome[21] and diabetes[22]. However, conventional diagnostic approaches remain episodic, providing only fragmented information that is insufficient to capture dynamic progression. In this context, ophthalmic wearable devices based on IFs and self-powered have emerged as a promising platform. By enabling sustained physiological sensing at the ocular surface, these devices support a transition from intermittent clinical assessment to continuous and personalized health management[23] - a transition that opens a new pathway for the prevention[24], diagnosis[25], and treatment[26] of ophthalmic diseases.
We first outline the fundamental design principles and material innovations that enable IFs to form effective interfaces with delicate ocular tissues. The discussion then examines advanced fabrication and manufacturing strategies, evaluating their ability to achieve precise structural control, flexibility, and functional integration while considering scalability for future production. A focus is placed on the synergistic integration of fibers with self-powered strategies, which is critical for moving toward intelligent, portable, and continuous monitoring platforms. We subsequently review key progress made by IFs in ophthalmic diagnosis and treatment, including flexible sensing architectures for dynamic IOP monitoring, continuous tear composition analysis, and neural interfaces for visual restoration. Finally, we critically address the challenges that must be overcome for clinical translation, encompassing long-term biocompatibility, signal stability, system reliability, and viable regulatory pathways. Collectively, this work illustrates the potential of IF technologies to shift eye care from intermittent intervention to continuous, dynamic, and personalized health management [Scheme 1], aiming to provide theoretical insights to support the development of next-generation intelligent ophthalmic platforms.
Scheme 1. Contrast between clinical protocols and novel IFs. IFs can be integrated with multi-source self-powered strategies, highlighting the transformative potential advancing ophthalmic diagnosis and therapy toward minimally invasive, continuous, and intelligent wearable devices. All the elements are from Biorender (Created in BioRender, Joe, Z. (2026). https://BioRender.com/tvsqz0t). IFs: Intelligent fibers.
DESIGN RATIONALE, MATERIAL INNOVATION AND FABRICATION OF INTELLIGENT OPHTHALMIC FIBERS
The origin of IF technology can be traced back to the “fibers as devices” concept proposed by Yoel Fink and co-workers at Massachusetts Institute of Technology in the late 1990s[27], which marked the transformation of fibers from passive transmission media to active functional devices. The transition of fibers from passive substrates to active, intelligent components underpins their application in ophthalmic devices. This progression is driven by the need for long-term, stable integration with the delicate and dynamic ocular surface. Achieving this requires a unified design strategy that combines biocompatible and functional materials, mechanically compliant fiber structure, and advanced fabrication methods for integrating multiple functions within a single fiber.
Material innovation for functional ocular interfacing
High-performance conductive fibers
Recent advancements in conductive fibers have paved the way for their application in sensitive areas such as the eye. For instance, a synergistic strategy combining laser etching and dopamine doping has been employed to induce selective phase separation in poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) (PEDOT:PSS)[28], which allows for precise tuning of its electrical conductivity, ranging from 297 to 4,525 S·cm-1 [Figure 1A]. This process facilitates the reorganization and enhanced crystallization of PEDOT-rich domains [Figure 1B], ensuring that the composite maintains suitable mechanical properties while achieving ultrahigh conductivity. As a result, it is well-suited for conformal integration with the dynamic and curved ocular surface. Carbon nanotube (CNT) fibers developed by Zhang et al. achieve a remarkable dynamic strength of up to 14 GPa while maintaining high electrical conductivity (~2.9 × 103 S·cm-1)[29], which may help ensure exceptional mechanical reliability and signal transfer stability in the dynamic ocular environment. Additionally, Zheng et al. developed flexible fibers fabricated based on silver-coated liquid metal particles (Ag@LMPs), which exhibit outstanding mechanical durability and electrical stability in flexible electronic circuits, with only ~7% resistance change (R/R0 ≈ 1.07) under torsion[30]. These fibers are ideal for use in wearable devices, including eye-tracking and ocular sensors, where dynamic deformation is common.
Figure 1. Fabrication and characterization of functional fiber materials. (A) Structural transition from PEDOT:PSS to laser/DA-treated LPPD; (B) Conformational change of PEDOT and increased domain richness. (A and B) are reproduced from Reference[28]. Copyright 2024, Wiley-VCH; (C) Concept of an OC device for neural signal monitoring; (D) Fabrication step of the OC device involving film fixation and metal patterning; (E) Microstructural differences between OC and NS devices; (F) Optical images of OC and NS devices after water immersion; (G) Metal area structure of both devices after water soaking. (C-G) are reproduced from Reference[33]. Copyright 2024, Elsevier; (H) Preparation routes for functionalized fibers via wet spinning; (I) Cross-sectional SEM and elemental mapping of an AlgCa fiber; (J) Cross-sectional SEM and elemental mapping of an AlgMX fiber. (H-J) are reproduced from Reference[35]. Copyright 2025, American Chemical Society. PEDOT:PSS: Poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate); DA: dopamine; LPPD: laser/DA-treated PEDOT:PSS-derived fiber; OC: oriented crystallization; NS: normal silk; SEM: scanning electron microscopy; AlgCa: calcium alginate fiber; AlgMX: MXene-alginate fiber.
Beyond these intrinsic conductive fibers, another critical pathway involves patterning traditional conductive metals (e.g., copper, gold) into microscale, fiber-like traces on flexible polymer substrates. These flexible electronic metal fibers function as embedded, passive sensing elements whose electrical properties (resistance, inductance, capacitance) are modulated by mechanical deformation[31,32]. This approach leverages mature microfabrication processes to demonstrate a complementary route to achieving device intelligence and system-level functionality within the stringent constraints of ophthalmic wearables.
Furthermore, the promising electrical and mechanical properties of conductive fibers such as PEDOT:PSS, CNTs, and liquid metals must be balanced against stringent requirements for long-term safety under direct, chronic ocular exposure. Key concerns encompass the potential leaching of components such as doping agents from PEDOT:PSS or metal ions from liquid systems, as well as the stability and biological impact of material degradation products within the saline, protein-rich ocular environment. For nanomaterials such as CNTs, additional risks are related to fiber morphology, residual catalyst content, and possible inflammatory responses. Even with inherent flexibility, mechanical mismatch and repetitive micro-motion at the tissue interface can provoke chronic inflammation. Therefore, before these materials can be adopted for clinical use, they must undergo comprehensive in vitro cytotoxicity and in vivo chronic implantation studies. Surface functionalization or encapsulation is frequently necessary to establish a stable, bio-inert interface that ensures both performance and safety over extended periods.
Biocompatible and functional fibers
For ocular applications, fiber materials must exhibit excellent biocompatibility and specific biological functions. Research has moved away from traditional bio-inert materials, focusing instead on novel composite fibers that combine degradability with electronic functionality, meeting both safety and functionality requirements in the eye. For example, silk fibroin (SF) is recognized as an ideal material for in vivo bioelectronic devices due to its strong mechanical properties, tunable degradation rate, and good biocompatibility[33] [Figure 1C-G]. However, its relatively low conductivity often necessitates blending or coating with conductive materials, which may complicate processing and affect long-term interfacial stability. Additionally, advanced processing methods, such as high temperature, pressure, and spray drying, allow the production of active powders with long-term storage stability and re-solubility, offering new material choices for developing ocular drug delivery systems.
Furthermore, nanocellulose, especially crystalline nanocellulose from tunicates, stands out for its purity, optical transparency, and compatibility with 3D bioprinting, offering possibilities for constructing complex ocular tissue engineering scaffolds. Nevertheless, its mechanical strength in hydrated states and its integration with functional electronic components remain practical challenges. Finally, chitosan and its derivatives are valuable for ocular drug delivery, thanks to their exceptional mucoadhesion and biocompatibility[34]. Various chitosan-based formulations, such as nanoparticles and nanogels, have been proven to enable sustained drug release and improve corneal permeability. Yet, the variable viscosity, batch-to-batch consistency, and potential for causing blurred vision upon gelation are important limitations that must be addressed. Overall, while each biocompatible fiber offers distinct advantages, their translation into durable, multifunctional ocular devices often involves a balance between degradability, electronic performance, mechanical robustness, and manufacturability.
Multifunctional and stimuli-responsive fibers
The next generation of IFs goes beyond static properties by incorporating dynamic, stimuli-responsive features. These materials can be engineered to respond to changes in pH, temperature, or specific biomarkers. Recent advancements in flexible hydrogel optical fibers based on polyethylene glycol diacrylate (PEGDA)-gelatin systems demonstrate transformative potential for intraocular photomedicine. These fibers exhibit high biocompatibility, mechanical softness, and efficient light transmission, along with inherent antibacterial properties. A notable example from Chen et al. illustrates this multifunctional approach. They developed a hydrogel-based fiber probe through an ion-chelation-assisted wet-spinning process, which combines neural recording, optogenetic stimulation, and fluid delivery within a single minimally invasive device[35] [Figure 1H-J]. This study presents a hydrogel-based multifunctional fiber probe [hydrogel-based fiber (HybF)] with high conductivity
Structural and interfacial properties of micro-/nano-fibers
Scaling fiber diameters down to the micro- and nanometer scales confers distinct functional advantages for ophthalmic applications, primarily due to an increased surface area, ultra-fine morphology, and tunable nanoscale porosity. The exceptional flexibility and minimal dimensions of these fibers enable them to establish conformal, low-mass interfaces with delicate ocular tissues such as the cornea, sclera, and retinal surface[36]. This intimate contact reduces mechanical mismatch, minimizes foreign-body sensation and immune response, and supports truly micro-invasive implantation strategies[37]. Furthermore, the interconnected nanoporous network characteristic of electrospun or self-assembled nanofiber mats shows potential to facilitate efficient diffusion of gases, nutrients, and tear components. In biosensing applications, for example, nanofibrous substrates provide a greatly expanded surface area for immobilizing biorecognition elements while enhancing analyte wicking and transport, collectively leading to superior sensitivity, faster responses, and lower detection limits compared to conventional dense films or larger-diameter fibers[38].
At the cellular level, nano-fibrous scaffolds closely replicate the topological features of the extracellular matrix. This biomimetic architecture delivers critical physical cues that can guide cellular behavior, such as promoting oriented corneal epithelial regeneration or directing axon growth in designed neural interfaces[39]. Such precise control over tissue integration and repair represents a functional advance that is difficult to achieve with conventional materials, underscoring the unique role of nano-fibers in accelerating the next generation of ophthalmic devices.
Structural design of fibers for enhanced ophthalmic functionality
In biomedical applications, both thin-film[40,41] and fiber-based materials have been widely explored for wearable and implantable devices. However, in ophthalmic applications, thin films often struggle to maintain stable conformality on the highly curved and dynamic ocular surface. They also face the risk of delamination when multiple functional layers are integrated. In contrast, fiber-based materials have advantages that address these limitations.
It is noteworthy that there exists a category of materials whose macroscopic devices present as a thin film, yet their internal structures consist of a porous network of micro-/nanofibers (e.g., aerogel fibers, electrospun fibers), which are classified as fiber-based materials in this work. The flexibility and one-dimensional geometry of fiber-based materials allow them to adapt to the cornea and sclera, ensuring reliable mechanical compliance during blinking and eye movements[42]. Additionally, the fiber format could enable complex multifunctional integration through coaxial or multi-core designs, consolidating sensing, stimulation, optical guidance, and fluidics into a single, minimally invasive structure. While thin-film processing excels in planar patterning, fibers provide a more robust and tissue-compatible solution for developing ophthalmic devices, particularly for conformal sensing, intraocular implants, and chronic neural interfaces. Table 1 provides a comparative overview of fibers and thin-film platforms for ophthalmic applications.
Comparison of fiber-based and thin-film-based platforms for ophthalmic devices
| Parameter | Fiber-based platforms | Film-based platforms | Implication for ophthalmology |
| Conformability and 3D integration | Natural compliance with curved surfaces (cornea, sclera). Can be woven/knitted into 3D constructs | Good on gentle curves; limited on highly curved or complex 3D structures | Fibers enable better fit and stable interfacing on the spherical eyeball |
| Mechanical robustness | High intrinsic flexibility and durability under repeated bending/flexion | Prone to cracking under cyclic strain | Essential for long-term reliability in dynamic ocular environment |
| Multifunctional integration | Enables multi-core, coaxial designs (conductor, insulator, waveguide in one fiber) | Requires layer-by-layer stacking, increasing thickness and delamination | Fibers allow creation of “all-in-one” devices |
| Power/data transmission | Suitable for optical/electrical transmission lines | Requires separate waveguides or wires | Fibers simplifies connection to external sensors |
| Manufacturing and scalability | Continuous, high-throughput production | Microfabrication is highly mature for 2D patterning | Fibers offer a promising route for scalable production |
Structural innovation in IFs serves as a crucial role in linking material properties to specific ophthalmic functions. At the micro-scale, geometric parameters of fibers, such as curvature, porosity, and channel topology, can be tailored to influence their physicochemical behavior and interactions with biological interfaces. For example, Varsha Sharma et al. engineered ion channels with varying tortuosity on cellulose fiber substrates[43], which shows preliminary success in mimicking the ion transport architecture of neural synapses [Figure 2A]. For ophthalmic applications, a fiber curvature designed to match the corneal surface could potentially improve conformal contact and signal stability for continuous IOP monitoring and might provide a foundation for future bio-interfaced devices that interact with retinal or neural circuits. Lu et al. developed a coaxial multilayer fiber structure that addressed the challenge of isolating electrodes from electrolytes[44], leading to fiber batteries that combine high energy density and flexibility. This structural strategy points toward a potential pathway for flexible, miniaturized power sources suitable for implantable or wearable ophthalmic electronics. The nacre-mimetic porous structure developed by Zhang et al. may allow for controlled drug diffusion via engineered porosity, supporting tailored release profiles for therapies targeting conditions such as glaucoma or post-operative inflammation[45] [Figure 2B]. While these aerogels are macroscopically presented as film devices, they are internally composed of a three-dimensional nanofiber network. This unique microstructure not only preserves ease of processing but also provides essential advantages such as high specific surface area, high permeability, and excellent mechanical resilience.
Figure 2. Optimization of fiber structures and designs. (A) Ion transmission structures mimicking neural synapses are realized by constructing ion channels with different twisting degrees on a cellulose fiber substrate[43]. Copyright 2025, American Chemical Society; (B) A lightweight, semi-transparent fiber filmwith a nacre-inspired multi-level channel structure[45]. Copyright 2023, Wiley‐VCH; (C) Schematic illustrating the transformation of a two-dimensional film with micro-patterned components into a one-dimensional fiber; (D) SEM image of a 320-channel optical fiber; (E) Wireless S-NeuroString for intestinal monitoring; (F) Structure of the S-NeuroString with integrated sensors and stimulation electrodes; (G) Representative sensor outputs showing intestinal contraction patterns. (C-G) are reproduced from Reference[46]. Copyright 2025, Springer Nature. SEM: Scanning electron microscopy; S-NeuroString: Spiral-NeuroString; PEDOT: poly(3,4-ethylenedioxythiophene); SCQs: spring-back-assisted BC-PMSQ; BC: bacterial cellulose; PMSQ: polymethylsilsesquioxane.
At the macro-scale, a central challenge lies in integrating high-density functional components into an ultra-flexible, minimally invasive fiber format. The “spiral transformation” technique represents a pivotal structural innovation to address this challenge. Khatib et al. at Stanford University engineered a one-dimensional tubular fiber (S-NeuroString) by rolling planar thin films into an ultra-slim profile of just 230 micrometers in diameter, yet integrating up to 1,280 channels [Figure 2C and D][46]. The resulting fiber exhibits a tunable modulus (1.2-81 MPa) that matches biological tissues. While validated in intestinal models [Figure 2E-G], its core material properties and structural design establish a direct and compelling blueprint for ophthalmic applications. For ophthalmic use, this macro-architectural strategy has three key advances: First, its ultra-slender, compliant profile is ideally suited for micro-invasive intraocular or periocular implantation, such as placement in the suprachoroidal space (between the sclera and choroid) or as a conformal optic nerve interface, drastically reducing surgical trauma compared to rigid implants. Second, the structure’s inherent mechanical compliance and high channel density are ideal for creating durable, high-fidelity neural interfaces for vision restoration. Finally, this integrated platform demonstrates the feasibility of combining electrodes, microfluidic channels, and optical waveguides within a single fiber. It offers a compelling design rationale for future ultraflexible, highly integrated ophthalmic neural interfaces.
SYNERGISTIC SELF-POWER STRATEGIES FOR AUTONOMOUS OPERATION
The integration of fiber-based materials with self-powered technologies is a cornerstone for advancing ophthalmic diagnostics and therapeutics toward minimal invasiveness, long-term in vivo operation, and precise modulation. This synergy could enable the “intelligent” functions of sensing and response by providing the necessary energy autonomy. Crucially, the unique structural and material properties of IFs enhance the efficiency, stability, and applicability of self-powered mechanisms in the ocular environment. For instance, the one-dimensional geometry and high surface area of micro-/nano-fibers significantly improve photon capture and charge collection in fibers[47], while their flexibility and tunable mechanics are suitable for converting the subtle, low-frequency energy of eye movements into usable electrical power[48]. Furthermore, the porous, three-dimensional network of nanofiber provides an idea scaffold for enzymatic biofuel cells, increasing the interfacial area for electrochemical reactions in tear fluid[49]. Thus, the link between form and function suggests that the concurrent design of the fiber substrate and its self-powered mechanism is critical for developing efficient, stable, and biocompatible autonomous devices.
Biomechanical self-powered shows significant potential for powering ophthalmic devices. For instance, eye-movement-driven nanogenerators can capture mechanical energy from periocular muscle activities such as blinking[50], generating electrical output with a power density of 10-100 µW/cm2 to operate sensing and communication modules[12]. A representative example is the blinking-powered contact lens (BPCL), which employs an integrated rectifier to convert intermittent eyelid motion into usable electrical energy, offering a practical route toward self-sustaining ocular interfaces [Figure 3A]. Advances in bioinspired thermoelectric materials further complement this approach. Intelligent textiles modified with polydopamine/polyaniline nanostructured coatings have achieved a substantially enhanced Seebeck coefficient of -30.2 µV·K-1, potentially enabling energy harvesting from eye surface temperature gradients[13]. Besides, tear fluid-driven biofuel cells, which enzymatically catalyze the electrochemical reaction between glucose and oxygen in tears[13-15,51], can produce a sustained stable output power density of 1-10 µW/cm2. Innovative designs such as dual-electrode systems that combine glucose oxidase (GOx) for cathode charging with self-reducing conductive polymer anodes exemplify the ongoing progress toward efficient, tear-based micro-power sources [Figure 3B].
Figure 3. Miniaturized self-powered strategies for smart ophthalmic fiber. (A) Working principle of a BPCL across different blinking stages (i-iv)[50]. Copyright 2023, The Author(s); (B) Tear fluid biofuel cell utilizing enzymatic and self-reduction reactions[15]. Copyright 2023, Elsevier; (C) Schematic of a FDSSC integrated with a light-diffusion and conversion layer[55]. Copyright 2024, Wiley-VCH; (D) Schematic of a fiber-based device capable of harvesting ambient electromagnetic wave energy and its potential application scenarios; (E) Photograph and (F) exploded view of the SMWSS device. (D-F) are reproduced from Reference[57]. Copyright 2025, Elsevier. BPCL: Blinking-powered contact lens; FDSSC: fiber-shaped dye-sensitized solar cells; SMWSS: self-powered multi-mechanism wireless sensing scheme; IoT: Internet of Things.
Significant progress has been made in lightpowered self-sustaining systems for implantable and flexible bioelectronics. Photovoltaic systems based on monocrystalline silicon[52], graphene[53], and organic semiconductors[54] enable efficient conversion of visible light to electricity, offering a viable power source for periocular wearable or implantable devices. Fiber-shaped dye-sensitized solar cells (FDSSCs), which integrate light diffusion and conversion layers, improve photon capture by controlling refraction and scattering[55]. This enhances self-powered efficiency of fiber electrode surfaces [Figure 3C]. Furthermore, emerging wireless energy transmission systems, using near-field coupling, can efficiently harvest ambient electromagnetic energy[56,57], presenting a promising route toward fully battery-free implantable ophthalmic electronics. Fiber-based devices capable of capturing stray electromagnetic waves (e.g., self-powered multi-mechanism wireless sensing scheme, SMWSS) have demonstrated feasibility in powering miniaturized sensors without the need for direct wiring or onboard batteries, highlighting a versatile strategy for sustainable operation in ocular environments [Figure 3D-F].
These diversified energy strategies, as summarized in Table 2, exhibit considerable potential in targeted clinical scenarios, particularly in the long-term management of chronic ocular conditions such as glaucoma. The choice of strategy depends on the application’s “intelligent” requirements: continuous IOP monitoring may utilize biomechanical energy harvested from blinking, whereas a glucose-sensing lens could integrate biochemical power from tears, forming a fully self-contained feedback loop. With continuous advances in materials science and micro-/nanofabrication, such self-sustaining power solutions are steadily progressing toward the realization of fully autonomous, long-term implantable ophthalmic devices[58,59], thereby providing reliable technical support for the personalized management of chronic eye diseases.
Summary of self-power strategies for ophthalmic IFs
| Power strategy | Technical principle | Application scenarios | Power density (Typical) | Advantages | Ref. |
| Biomechanical self-powered | Converts eye movement into electricity | Glaucoma monitoring, implantable sensors | 10-100 µW/cm2 | Sustainable, seamless integration | [12,50] |
| Biochemical self-powered | Uses tear fluid to generate power | Diabetes management, tear composition analysis | 1-10 µW/cm2 | Easy to obtain | [13-15,41] |
| Light self-powered | Converts light into electricity | Periocular devices, superficial implants | 50-200 μW/cm2 | Wide energy source, eye-compatible | [52-55] |
| Wireless energy transmission | Harvests ambient electromagnetic energy | Brain-computer interfaces, long-term monitoring | 1-50 μW/cm2 | Portable, senseless | [56,57] |
Implementing self-powered strategies fundamentally depends on aligning the harvested energy with the device’s total power budget, a balance dictated by the intended duty cycle of continuous monitoring. Core ophthalmic functions exhibit a broad range of power demands. For example, a minimalist continuous IOP sensor may operate on 1-10 µW, while integrating wireless data transmission can raise the requirement to 10-50 µW. Energy harvesters driven by blinking, with an output of 10-100 µW/cm2, can theoretically satisfy these needs given sufficient active area and conversion efficiency. However, advanced systems aiming for multimodal sensing, active feedback, and robust communication may demand hundreds of microwatts[60], challenging standalone biomechanical or biochemical sources.
To reconcile intermittent energy harvesting from ambient or body with stable device operation, power management is essential. A foundational strategy is the work-upload-sleep duty cycle[61]. The device stays in a low-power sleep state most of the time. It wakes periodically to perform sensing and may transmit data in bursts before returning to sleep. This lowers the average power draw, better matching the average power available from sources such as blinking or thermal gradients. If ambient energy alone cannot support the desired function or duty cycle, a hybrid energy system can be used. Such a system might combine a primary ambient harvester, such as a tear glucose biofuel cell or a solar cell, with a secondary source, such as an external charging device. Their variable output is consolidated and smoothed using an integrated energy storage buffer, such as a micro-supercapacitor, which supplies the peak power needed and coordinates the process using an energy management circuit (EMC)[62]. It is responsible for performing maximum power point tracking from each energy harvester, coordinating the efficient charging and discharging of the storage buffer, and dynamically allocating power to various subsystems based on real-time energy availability. Therefore, an effective harvesting strategy must be co-designed from the beginning with the device’s functional specifications, duty cycle, and power management to improve reliable and autonomous operation.
INTEGRATION AND ENCAPSULATION FOR LONG-TERM OCULAR COMPATIBILITYS
In the development of ophthalmic IFs devices, integration and encapsulation strategies are crucial for ensuring stable, long-term operation in the complex ocular environment. Current research is focused on achieving a balance between device functionality and biocompatibility through advanced material processing and micro-/nanofabrication techniques. In terms of functional integration, bottom-up approaches such as supramolecular self-assembled could provide the precise construction of nanostructured organic conductors, allowing for the formation of high-density functional circuits on fiber substrates[63] [Figure 4A-D]. However, scaling such intricate self-assembly processes to continuous, high-throughput manufacturing remains a significant challenge. Meanwhile, top-down methods, including thermal drawing of multimaterial preforms combined with lithographic patterning, have facilitated the integration of complex micro-optical and electronic components into fiber devices[64-67] [Figure 4E]. Thermal drawing is a matured and continuous process, yet the fabrication of the complex multimaterial and maintaining interfacial stability during drawing are key determinants of its scalability and cost for mass production. Additionally, advances in laser micromachining and scanning electrochemical probe lithography (SEPL) provide versatile tools for high-resolution surface texturing and localized material modification [Figure 4F][66].
Figure 4. Functional integration processes for IFs. (A) Schematic of the supramolecular self-assembly of planar nanographenes with peripheral substituents; (B) TEM and (C) AFM images of the resulting supramolecular nanofibers; (D) Molecular simulation of a double-helical self-assembled nanofiber, showing its helical pitch. (A-D) are reproduced from Reference[63]. Copyright 2021, American Chemical Society; (E) Schematic illustrating the thermal drawing of a multimaterial preform into continuous fibers, alongside the preparation of microtextured thermoplastic elastomers using a patterned mask[65]. Copyright 2022, The American Association for the Advancement of Science; (F) Diagram of the scanning electrochemical probe lithography process, demonstrating the principle of voltage-regulated local etching of N-GaAs and the resulting etched grooves produced under constant voltage[66]. Copyright 2024, Wiley-VCH. IFs: Intelligent fibers; TEM: transmission electron microscopy; AFM: atomic force microscopy; SEBs: poly(styrene-b-(ethylene-co-butylene)-b-styrene); N-GaAs: n-type gallium arsenide.
However, it is important to note that techniques such as SEPL, while offering flexibility for prototyping, are single-threaded processes with low throughput and high cost. Their feasible role in transformation might be limited to creating master templates for higher-volume replication methods rather than direct device manufacturing. Consequently, while these advanced integration and processing methods offer a promising technological pathway for multifunctional ophthalmic platforms, their successful clinical translation is critically dependent on scalability for mass production and implementing effective cost-control.
In recent years, encapsulation strategies using IFs in ophthalmic applications have also evolved significantly. These strategies have shifted from serving as passive physical barriers to multifunctional systems that integrate biocompatibility, mechanical compliance, signal transduction, and dynamic responsiveness. Bio-derived materials such as SF[68], collagen/gelatin[69], chitosan[70], and elastin-like polypeptides (ELPs)[71] form the foundation of these advanced encapsulation systems. Processed via micro-fabrication techniques into multidimensional structures, these materials enable gentle encapsulation and tunable degradation, eliminating the need for secondary surgical removal. For example, biomimetic Janus structures based on acellular corneal stroma (electro-assembled biomimetic collagen, EB-Col), which can be customized in curvature and thickness, may help ensure rapid, stable tissue adhesion in the moist ocular environment [Figure 5A and B][69]. This approach holds promise for corneal repair and device integration. Such systems support seamless tissue-device integration and long-term stability, making them suitable for both ocular surface devices and intraocular implants.
Figure 5. Encapsulation strategies for IFs devices. (A) Biomimetic Janus bioadhesive encapsulation based on collagen, achieving rapid and stable tissue adhesion while adapting to the moist and dynamic ocular environment; (B) Schematic showing that the Janus is encapsulated by constructing a coating composed of 4-arm-PEG-NHS and ε-PLL. (A and B) are reproduced from Reference[69]. Copyright 2025, Elsevier; (C) Fabrication and encapsulation process of a multilayer composite film and an electrothermal smart device[73]. Copyright 2025, American Chemical Society; (D) Integration of conductive components such as MXene nanosheets and PEDOT:PSS into the fiber matrix forms a composite system capable of simultaneous encapsulation and efficient signal transmission, suitable for contact-lens-based tear sensors and optic nerve interfaces[74]. Copyright 2024 The Author(s). IFs: Intelligent fibers; PEG: polyethylene glycol; NHS: N-hydroxysuccinimide; ε-PLL: ε-Poly-L-lysine; MXene: transition metal carbides and/or nitrides; PEDOT:PSS: poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate); ACS: artificial cornea stromal substitutes.
These fiber-based encapsulation layers have not only played a foundational role but have also been further functionalized to incorporate bioactive and responsive features. For example, surface modification with Arg-Gly-Asp (RGD) peptide improves corneal epithelial adhesion and integration[72], while the addition of anti-inflammatory agents could enable targeted immunomodulation. Multilayer composite films fabricated through sequential lamination and bonding processes provide robust, multifunctional encapsulation for complex devices such as electrothermal smart implants[73] [Figure 5C]. The integration of conductive components, including MXene nanosheets and PEDOT:PSS, into fiber matrices results in composite systems that offer both encapsulation and efficient signal transmission. These systems are particularly useful in applications such as contact-lens-based tear sensors and optic nerve interfaces[74] [Figure 5D]. Moreover, stimuli-responsive materials have been utilized to develop intelligent encapsulation layers capable of autonomously regulating drug release or barrier permeability in response to pathological signals[75,76].
As encapsulation systems evolve toward increasingly multifunctional integration, their long-term reliability faces severe challenges in the continuously moist ocular environment, especially when self-powered units become a part of the device. For energy harvesters that depend on electrostatic induction, including triboelectric nanogenerators (TENG), the conductive nature of the tear film promotes rapid charge dissipation and significant parasitic leakage. This effect can severely degrade their electrical output and long-term reliability. Although encapsulation provides a necessary protective barrier, it introduces a critical design trade-off. A barrier effective enough to prevent ionic and moisture ingress often results in increased device thickness and stiffness. This can in turn compromise the essential attributes of patient comfort and conformability to the delicate ocular tissue. Consequently, material selection and encapsulation strategy must be co-optimized. The objectives extend beyond biocompatibility to include advanced properties such as inherent hydrophobicity, superior ionic barrier performance, and long-term hydrolytic stability[77]. Exploring alternative self-powered principles that are inherently less susceptible to humidity may provide more viable pathways for long-term ocular implants.
In summary, current research is shifting the focus of fiber-based encapsulation from a simple protective barrier into an integrated platform that may enable synergistic sensing, feedback, and therapeutic intervention. At the same time, advances in integration and encapsulation technologies are helping to define the application potential of IFs devices in areas such as continuous IOP monitoring, minimally invasive imaging of the anterior chamber angle, and retinal function restoration. Advances in encapsulation technology hold promise for effectively integrating multifunctional materials with self-powered strategies, helping to meet the stringent requirements of ophthalmic wearable devices for humidity resistance, mechanical durability, and biocompatibility.
APPLICATIONS OF IFs WEARABLE DEVICES IN OPHTHALMOLOGY
In recent decades, the rapid evolution of IF technology has led to breakthroughs in ophthalmic medical devices. These advanced systems, which are compact[78], flexible[79], and biocompatible[80], are made from innovative materials, including conductive polymers, liquid metals, and bio-derived substrates. Their unique mechanical and structural properties allow them to conform seamlessly to the complex and delicate ocular tissues. This stable and minimally invasive interface may promote not only long-term in vivo monitoring but also active interventions, such as electrical stimulation[81] and drug delivery[82,83].
This represents a departure from conventional ophthalmic diagnostics, which are largely confined to episodic, clinic-based spot measurements such as tonometry or slit-lamp examinations. These established methods provide invaluable but static snapshots of ocular health, often missing critical dynamic fluctuations in parameters. In contrast, fiber-based platforms introduce a continuous, longitudinal monitoring that can be conducted in a home environment, offering a more comprehensive physiological profile for chronic disease management. Furthermore, the flexibility, lightweight, and biocompatibility of fibrous materials allow for the creation of devices that are far less intrusive and more comfortable for prolonged wear compared to the bulky or stimulating contact required by many standard instruments[84,85]. While traditional tools remain the cornerstone of definitive diagnosis and calibration, IFs might complement them by filling the critical data gap between clinical visits, thereby facilitating proactive and personalized ocular healthcare.
However, this transformative potential is balanced by current translational challenges. Key limitations - such as long-term signal stability, the need for reliable calibration protocols, and the achievement of full energy autonomy - represent significant engineering hurdles that must be overcome to match the proven reliability and standardized use of established clinical equipment. This section examines novel applications where fiber-based wearable devices show promise in diagnostic and therapeutic use in ophthalmology.
IOP monitoring fiber devices
Glaucoma, a major cause of irreversible blindness worldwide, is primarily characterized by progressive damage to the optic nerve. The key pathological mechanism involves elevated IOP causing mechanical stress, ultimately leading to the apoptosis of retinal ganglion cells. Consequently, continuous and accurate monitoring of IOP is an important indicator[86]. In the field of glaucoma management, IFs devices demonstrate unique advantages. Sensors based on the microfluidic principle indirectly estimate IOP by detecting changes in corneal curvature[87]. Their core is a micro-channel filled with fluid. When IOP changes cause pipeline deformation, the internal fluid pressure changes accordingly[88]. In contrast, fiber Bragg grating (FBG) sensors utilize the sensitivity of the grating period in the optical fiber to mechanical strain, converting IOP changes into wavelength shift signals, offering advantages such as immunity to electromagnetic interference and intrinsic safety[89,90]. Piezoelectric fiber sensors rely on the property of piezoelectric materials generating surface charges under stress, enabling capture of minute deformations of the eyeball. Importantly, when these piezoelectric or conductive materials are fabricated into micro-/nano-fibers, the resulting sensors exhibit exceptional sensitivity to minute strains due to their high surface-area-dependent piezoresponse and extreme mechanical flexibility[91]. This allows for the development of ultra-thin, conformal IOP sensing patches or the integration of highly sensitive elements into IFs devices[92]. The practical implementation of such sensors introduces two critical considerations. First, most piezoelectric materials require a high-voltage poling process to align their internal dipoles, which poses a significant integration challenge for miniaturized, implantable medical devices. Second, the long-term biological safety of the material is paramount. While traditional high-performance piezoceramics such as lead zirconate titanate (PZT) offer strong signals, their lead content presents a toxicity risk. Consequently, lead-free alternatives such as potassium sodium niobate (KNN) ceramics and biocompatible polymers such as polyvinylidene fluoride (PVDF) are increasingly favored for ocular applications[93]. Although these materials may have a lower inherent piezoelectric coefficient, their excellent biocompatibility, flexibility, and often lower poling voltage requirements make them more suitable for safe, chronic implantation.
The integration of these sensing fibers with contact lens platforms creates a monitoring system that naturally conforms to the cornea. More advanced implementations, such as wireless multifunctional contact lenses (WMCLs), combine deformation sensing with telemetry, enabling continuous IOP tracking and external readout without affecting patient mobility. Notably, a prominent design strategy for such WMCLs employs flexible electronic metal fibers, precisely patterned metallic coil circuits on polyimide substrates as the core inductive sensing element. As shown in Figure 6A, deformation of the corneal surface alters the geometry of these embedded metal fibers, thereby shifting their resonant frequency for wireless IOP readout[94]. This approach exemplifies that the functional principle of fiber-like sensing could be implemented using high-precision planar fabrication, offering advantages in integration density and signal consistency. While the integration of a self-powered sensor with a glaucoma drainage device presents an attractive vision for autonomous monitoring, the decision to embed an energy harvester within the implant must be weighed against the alternative of powering the sensor via inductive coupling through the conjunctiva. The latter may offer a simpler and surgically lower-risk pathway.
Figure 6. IFs devices for ophthalmic sensing and visual restoration. (A) A flexible wireless contact lens for IOP monitoring, utilizing patterned electronic metal fibers as the inductive sensor[94]. Copyright 2024, American Chemical Society; (B) Flexible fiber-based contact lens with wireless power transmission, integrated glucose sensing, and LED visual feedback[95]. Copyright 2018, The American Association for the Advancement of Science; (C) Tellurium nanowire network (TeNWN) retinal prosthesis enabling broad-spectrum light absorption and subretinal photocurrent generation for visual restoration[98]. Copyright 2025, The American Association for the Advancement of Science; (D) High-density fiber arrays for precise cortical interfacing, including linear and bundled configurations[103]. Copyright 2024, The Author(s). IFs: Intelligent fibers; IOP: intraocular pressure; LED: light-emitting diode; TeNWN: tellurium nanowire network.
Tear fluid biochemical analysis fiber devices
Tear fluid, as a biofluid highly correlated with blood components, contains abundant health information. IFs integrated into the eyelid-facing side or the canthus of contact lenses enable continuous monitoring of tear composition. For instance, in diabetes management, integrated electrochemical sensors with GOx can reflect blood glucose levels indirectly by monitoring fluctuations in tear glucose concentration, with detection limits reaching ~10 µM[95]. For oxidative stress assessment, based on the absorption or fluorescence quenching of specific wavelength light by ascorbic acid (AA), real-time evaluation of corneal oxidative stress status could be provided[96]. For stress level monitoring, cortisol, a stress hormone, can be detected in tears via competitive immunoassays, providing an objective indicator for mental stress assessment[97]. A critical advancement is the use of nanofiber-based substrates as the sensing platform. The high porosity and vast surface area of nanofibrous networks enable (i) efficient capillary action for rapid tear uptake and retention; (ii) high-density immobilization of enzymes or antibodies, enhancing signal magnitude; and (iii) shortened diffusion paths for analytes, resulting in faster response times and lower detection limits compared to sensors built on conventional devices.
Fiber-based sensing systems are increasingly being designed with closed-loop functionality, allowing for real-time monitoring and immediate responses to physiological changes. For example, flexible smart contact lenses can wirelessly receive power through an integrated antenna. This power activates embedded sensors and micro-light-emitting diode (LED), allowing for direct visual feedback on the lens. In a typical example, an LED is programmed to turn off when the detected glucose concentration exceeds a predefined threshold[95] [Figure 6B]. This integration of fiber-based sensors and actuators into smart contact lenses demonstrates the potential of fibers to create multifunctional, responsive systems that can provide real-time feedback and actively manage physiological conditions.
Visual function restoration and enhancement
In the field of visual function restoration and enhancement, IF technology holds transformative potential. Flexible fiber electrode arrays may lead to the breakthrough of retinal prostheses implantation. For example, Wang et al. developed a tellurium nanowire network retinal prosthesis, through a single minimally invasive subretinal implantation and without the need for an external power supply, restored visible light vision in blind animal models. Remarkably, it also enabled the animals to perceive infrared light [Figure 6C]. This prosthesis operates across a wide spectrum, from visible light to the near-infrared II region (470-1,550 nm), and can spontaneously convert light signals into electrical signals, directly activating surviving nerve cells in the retina[98].
Electrodes or scaffolds composed of conductive nanofibers present a topology that more closely mimics the neural extracellular environment. The nanoscale morphology can reduce glial scar formation, improve charge injection efficiency, and provide directional cues for neurite outgrowth. In the context of optic nerve repair or cortical visual prostheses, aligned nanofiber scaffolds have shown promise in guiding axon regeneration and establishing more stable, high-fidelity neural connections compared to smooth electrodes[99].
The application of optical fibers in optogenetic modulation of retinal neurons provides another precise intervention strategy. By transfecting light-sensitive optogenetic proteins into specific retinal neurons and then using miniaturized optical fibers to deliver light of specific wavelengths, precise modulation of preserved neural circuits in degenerated retinas can be achieved[100]. Such systems are evolving towards fully implantable, wirelessly powered directions. For instance, some research combines upconversion nanoparticles with flexible optical fibers, achieving NIR light-triggered visible light responses for optogenetics through tissue[101,102], greatly enhancing treatment penetration and safety.
For intervention in higher visual pathways, fiber-shaped high-density micro-electrode arrays that interface with the visual cortex show unique value. These ultra-flexible electrode arrays, thinner than a human hair, can be implanted into the visual cortex with minimal trauma. When arranged in linear or bundled configurations (e.g., 100-fiber arrays), they achieve submillimeter spatial precision for selective stimulation of cortical columns (functional units within the visual cortex), directly eliciting phosphene phenomena, which are perceptions of light flashes or patterns induced by neural stimulation. This approach offers a direct brain-computer interface for bypassing damaged eyes and optic nerves [Figure 6D][103]. With increasing electrode density and biocompatibility, such brain-computer interface devices are offering the possibility for completely blind patients to bypass damaged eyes and optic nerves and communicate directly with the brain’s visual center.
However, it is worth noting that the challenge for any chronic neural interface is the foreign body response (FBR). Over time, even biocompatible materials can induce glial scarring, which forms an insulating layer between the electrode and target neurons. This encapsulation progressively increases interfacial impedance and attenuates signal quality, ultimately leading to functional degradation of the prosthesis. Therefore, the design of fiber-based neural interfaces must extend beyond acute biocompatibility to strategies that mitigate chronic FBR. This includes engineering fiber surfaces with anti-fibrotic coatings (e.g., drug-eluting systems), optimizing mechanical compliance to minimize micromotion, and exploring topological cues that discourage glial overgrowth while promoting neuronal integration[104,105].
Advantages of micro-/nano-fibers in ophthalmology
As detailed in the preceding sections, the transition from conventional fibers to those with micro- and nano-scale diameters is not merely a matter of dimensional scaling but represents a fundamental shift in functionality. The distinct structural characteristics of micro-/nano-fibers, including their exceptionally high surface volume ratio, tunable nano-porosity, biomimetic topography, and ultra-high flexibility, may enable performance enhancements that are critical for advanced ophthalmic applications. The comparative summary in Table 3 delineates how specific morphological and material properties of micro-/nano-fibers directly address key challenges and improvements in device performance across the fields of physiological monitoring, biochemical sensing, and neural interfacing.
Performance advantages of micro-/nano-fibers in ophthalmic applications
| Application | Representative fiber morphology | Structural advantage | Performance benefit |
| IOP monitoring (Section 3.1) | Piezoelectric nanofiber; Conductive polymer nano-fibers | Mechanical compliance; High transducer response | Enhanced sensitivity to minute corneal deformation; Reduced mechanical loading |
| Tear fluid biosensing (Section 3.2) | Highly porous nanofiber | Nanoporosity; Maximized specific surface area | Accelerated analyte diffusion; High density of immobilized biorecognition elements |
| Visual restoration (Section 3.3) | Ordered nanofiber scaffolds | Biomimetic topology | Promotion of directed neuronal growth |
In monitoring, their exceptional mechanical compliance and conformability could enable precision sensing with minimal interference, thereby mitigating fibrotic encapsulation and signal drift commonly seen with rigid implants. For biosensing, their intricately porous and interconnected architecture creates an optimized microenvironment that facilitates both high probe-loading capacity and rapid analyte diffusion, which are critical for sensitive and timely molecular detection. In the realm of intervention and restoration, their biomimetic topology and mechanical cues fosters harmonious integration with neural tissue by directing cell adhesion, proliferation, and axonal guidance. Thus, micro-/nano-fibers are not simply smaller versions of conventional fibers but constitute a distinct and enabling materials platform whose functionality is encoded in its geometry and material composition. Its targeted use is therefore pivotal for advancing toward next-generation intelligent ophthalmic systems, as it enables the convergence of minimally invasive, highly sensitive diagnostics, and biocompatible therapeutic interfaces within a multifunctional framework.
CRITICAL CHALLENGES AND PATHWAYS TOWARD CLINICAL TRANSLATION
The promising laboratory results described in the preceding sections were often achieved under controlled and idealized conditions. However, their translation into viable clinical solutions encounters a series of profound and interconnected challenges that define the current frontier of the field. A primary hurdle lies in achieving sustainable long-term biological integration, which extends far beyond short-term biocompatibility assessments. The inevitable chronic FBR can lead to progressive fibrotic encapsulation of implanted fibers. This encapsulation may electrically insulate sensors, mechanically constrain device functionality, alter local physiology, and potentially contribute to secondary complications such as elevated IOP, thereby reducing the long-term performance and safety envisioned. Mitigating this response requires moving beyond inert materials toward actively engineered interfaces. This includes developing fiber surfaces with anti-fibrotic coatings, topological cues that discourage glial cell adhesion, or dynamically responsive materials that can modulate the local immune environment.
Concurrently, engineering complete device autonomy and reliability presents a formidable systems-level challenge. Many compelling laboratory demonstrations depend on externalized power sources or wired data connections. Translating these into a fully implanted and self-sustaining system requires the co-optimization of energy harvesting, ultra-low-power operation, robust wireless communication, and fault-tolerant design to improve stable service without intervention, a goal that remains largely unmet. Key limitations include the significant gap between harvested power (µW) and the peak power required for wireless transmission (mW). Bridging this gap necessitates not only a more efficient self-powered strategy but also intelligent power management, such as efficiency duty cycling and hybrid systems that combine self-powered with periodic external recharging. This challenge is tightly linked to the critical issue of scalable and reproducible manufacturing. Fabricating these multi-material, micro-structured fiber devices with the consistency, reliability, and quality required for human implantation under Good Manufacturing Practice (GMP) standards represents a significant step beyond laboratory prototyping.
Furthermore, establishing definitive clinical validation pathways is essential. This necessitates a rigorous transition from proof-of-concept studies in animal models to comprehensive safety and efficacy testing in clinically relevant disease models. Success in this stage must then be followed by phased human clinical trials. These trials must evaluate not only biological and functional outcomes but also practical determinants, including patient quality of life, surgical implantation feasibility, long-term management, and overall cost-effectiveness. The regulatory pathway for such active, intelligent implantable devices is complex, requiring clear benefit over existing standards of care and risk management throughout the product lifecycle. Actively addressing these translational gaps is imperative for the field to evolve and deliver practical clinical impact. Ultimately, the convergence of advanced materials, systems engineering, scalable manufacturing, and clinical evaluation will determine the pace at which IFs solutions transition from prototypes to standard tools.
CONCLUSION AND OUTLOOK
This review provides a comprehensive look at the recent advances and future potential of IF technology in ophthalmic wearable devices. The integration of sensing, power, and communication capabilities in multifunctional devices has been made possible through innovations in material systems, structural designs, advanced fabrication techniques, and self-powered strategies. Collectively, these developments are driving ophthalmic care toward a new era of minimally invasive, continuous, and personalized treatments, supported by the unique flexibility, biocompatibility, and integration potential of IFs.
While the scientific and engineering progress in IFs is substantial, ideal laboratory results must be rigorously evaluated against the demands of clinical deployment. Several challenges remain, as many innovative self-powering strategies operate at power levels that fall short of supporting the continuous wireless operation required for full device autonomy. Furthermore, the long-term biocompatibility and functional stability of fiber-based interfaces in the dynamic, saline-rich ocular environment remain incompletely validated, with persistent risks such as biofouling, material degradation, and signal drift. The critical bottlenecks primarily lie in energy budget constraints, environmental durability, and interfacial integration strategies, which collectively limit the functional complexity and clinical viability of IFs. This is especially evident in the development of intelligent contact lenses, which confront formidable multi-faceted hurdles including the miniaturization of multimodal sensors, efficient micro-power management, and reliable wireless communication. Therefore, despite notable advancements, the path toward clinically viable, self-powered ophthalmic devices necessitates focused efforts to overcome these barriers.
Looking ahead, the convergence of several interdisciplinary domains is supposed to reshape the development of IFs ophthalmic devices, driven by three major trends. First, the frontier of materials science and structural engineering will move beyond single-function components toward multi-mechanism systems. Innovations will increasingly focus on self-powered designs, eliminating the need for bulky batteries, and on advanced biomimetic structures that replicate the nuanced mechanical and optical properties of eyes. Second, the deep integration of AI will transform passive devices into active diagnostic and therapeutic platforms. Specifically, the deployment of edge computing and lightweight, quantized neural networks directly on the device or in a paired module will allow for real-time, on-site data processing, thus achieving low latency, preserving data privacy, and reducing power consumption. This could lead to contact lenses or implants that not only continuously monitor but also predict individual risk of glaucomatous progression or even trigger closed-loop systems that adjust drug release profiles automatically in response to real-time biomarkers. Finally, the transition from laboratory prototypes to clinically viable products hinges on parallel advances in cross-scale manufacturing technologies. Emerging techniques such as microfluidic spinning for ultra-uniform fiber production, preform-based thermal drawing for complex multifunctional fibers, and novel hybrid processes that integrate nano-patterning with macro-scale assembly are critical. These technologies will accelerate clinical translation with high reproducibility, cost-effective and scalable production of the patient-specific ophthalmic devices.
In conclusion, the trajectory of IF technology in ophthalmology is undoubtedly promising, yet it requires balanced optimism. The field stands at a crossroads between compelling proof-of-concept studies and the stringent requirements of clinical utility. Success will depend not only on continued material and engineering innovation but also on deliberate efforts to address durability, user-centered design, cost-effectiveness, and regulatory pathways. True transformation will be measured not by technological sophistication alone, but by tangible improvements in patient outcomes, accessibility, and quality of life.
As these technologies continue to mature, IFs are set to become crucial in managing glaucoma, diagnosing dry eye syndrome, and restoring visual function. Ultimately, this technology promises to transform ophthalmic care from a traditional model of intermittent intervention to continuous health management, offering patients more precise, comfortable, and effective diagnostic and therapeutic solutions.
DECLARATIONS
Acknowledgments
We thank Dwad Dwad for assistance with the creation of the Graphical Abstract and Scheme 1 using BioRender.com [Created in BioRender. Joe, Z. (2026) https://BioRender.com/tvsqz0t].
Authors’ contributions
Made substantial contributions to the conception, design, theoretical framework and writing of the review: Qiao Z, Li Y
Performed critical literature search, data extraction, and integration: Cao M, Chen J, Zuo M
Provided administrative, technical, and material support: Jia R, Ju X, Zhang K
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorships
This work was supported by the Sichuan Provincial Science and Technology Program (2024NSFJQ0048).
Conflict of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Xindi, S.; Fengyuan, Z.; Lingyu, Z.; et al. Enhanced electromechanical conversion via in situ grown CsPbBr3 nanoparticle/poly(vinylidene fluoride) fiber composites for physiological signal monitoring. Soft. Sci. 2022, 2, 1.
2. Yu, X.; Chen, L.; Zhang, J.; et al. Structural design of light-emitting fibers and fabrics for wearable and smart devices. Sci. Bull. 2024, 69, 2439-55.
3. Ikram, N.; Patel, N. A.; Kvedar, J. C. Wearable health devices for pediatric ophthalmology. NPJ. Digit. Med. 2025, 8, 354.
4. RaviChandran, N.; Teo, Z. L.; Ting, D. S. W. Artificial intelligence enabled smart digital eye wearables. Curr. Opin. Ophthalmol. 2023, 34, 414-21.
5. Smith, M.; Cacucciolo, V.; Shea, H. Fiber pumps for wearable fluidic systems. Science 2023, 379, 1327-32.
6. Seo, H.; Chung, W. G.; Kwon, Y. W.; et al. Smart contact lenses as wearable ophthalmic devices for disease monitoring and health management. Chem. Rev. 2023, 123, 11488-558.
7. Chen, K.; Song, E.; Koh, W-G.; Myung, D. A cornea-mimetic electrospun fiber-reinforced hydrogel platform for ocular surface applications. Macromol. Res. 2025, 33, 725-35.
8. Omer, S.; Nagy, N.; Szőcs, E.; et al. Development of innovative electrospun nepafenac-loaded nanofibers-based ophthalmic inserts. Int. J. Pharm. 2023, 647, 123554.
9. Tummala, G. K.; Joffre, T.; Lopes, V. R.; et al. Hyperelastic nanocellulose-reinforced hydrogel of high water content for ophthalmic applications. ACS. Biomater. Sci. Eng. 2016, 2, 2072-9.
10. Qu, S.; Sun, L.; Zhang, S.; et al. An artificially-intelligent cornea with tactile sensation enables sensory expansion and interaction. Nat. Commun. 2023, 14, 7181.
11. Xiao, L.; Yin, B.; Geng, Z.; Li, J.; Jia, R.; Zhang, K. Flexible wearable devices based on self-powered energy supply. Nano. Energy. 2025, 142, 111157.
12. Pourshaban, E.; Banerjee, A.; Karkhanis, M. U.; et al. Eye tear activated mg - air battery driven by natural eye blinking for smart contact lenses. Adv. Mater. Technol. 2022, 8, 2200518.
13. Wang, Y.; Wu, S.; Yin, Q.; et al. Novel hybrid p- and n-type organic thermoelectric materials based on mussel-inspired polydopamine. ACS. Appl. Mater. Interfaces. 2021, 13, 23970-82.
14. Wang, Z.; Dong, Y.; Sui, X.; et al. An artificial intelligence-assisted microfluidic colorimetric wearable sensor system for monitoring of key tear biomarkers. npj. Flex. Electron. 2024, 8, 35.
15. Yun, J.; Li, Z.; Miao, X.; et al. A tear-based battery charged by biofuel for smart contact lenses. Nano. Energy. 2023, 110, 108344.
16. Zhang, L.; Yang, Y.; Jiang, Z.; et al. Demonstration of topological wireless power transfer. Sci. Bull. 2021, 66, 974-80.
17. Liu, C.; Feng, Z.; Yin, T.; et al. Multi-interface engineering of MXenes for self-powered wearable devices. Adv. Mater. 2024, 36, e2403791.
18. Pang, Y.; Li, Y.; Wang, X.; Qi, C.; Yang, Y.; Ren, T. L. A contact lens promising for non-invasive continuous intraocular pressure monitoring. RSC. Adv. 2019, 9, 5076-82.
19. Tai, Y.; Cheng, Q.; Lu, X.; et al. Recent advances in wearable and implantable intraocular pressure sensors for glaucoma prevention and therapeutic management. Adv. Mater. Technol. 2025, 10, e01191.
20. Li, C.; Li, Y.; Liu, X.; Xie, S. Flexible transparent capacitive pressure sensors for multimodal ophthalmic monitoring: intraocular pressure assessment, movement disorder diagnosis, and eye-tracking human-computer interaction system. Adv. Funct. Mater. 2025, 36, e20580.
21. Kannan, R.; Das, S.; Shetty, R.; Zhou, L.; Ghosh, A.; Deshpande, V. Tear proteomics in dry eye disease. Indian. J. Ophthalmol. 2023, 71, 1203-14.
22. Roostaei, N.; Hamidi, S. M. Plasmonic smart contact lens based on etalon nanostructure for tear glucose sensing. Sci. Rep. 2025, 15, 14948.
23. Xiao, R.; Yu, G.; Xu, B. B.; Wang, N.; Liu, X. Fiber surface/interfacial engineering on wearable electronics. Small 2021, 17, e2102903.
24. Hong, S.; Yu, T.; Wang, Z.; Lee, C. H. Biomaterials for reliable wearable health monitoring: Applications in skin and eye integration. Biomaterials 2025, 314, 122862.
25. Zha, X.; Zhou, N.; Hu, M.; et al. Piezoresistive MOF for intraocular pressure monitoring. ACS. Sens. 2025, 10, 6656-64.
26. Ran, S.; Chen, Y.; He, H.; Zhang, Q.; Liu, X.; Wang, Y. Short-peptide-induced diffusion-limited plasmonic aggregation with broad spectral tunability for wearable ophthalmic devices in color vision deficiency correction. Adv. Funct. Mater. 2025, 35, 2411649.
27. Abouraddy, A. F.; Bayindir, M.; Benoit, G.; et al. Towards multimaterial multifunctional fibres that see, hear, sense and communicate. Nat. Mater. 2007, 6, 336-47.
28. Zeng, M.; Ding, J.; Tian, Y.; et al. Phase separation manipulated gradient conductivity for a high-precision flexible pressure sensor. Adv. Funct. Mater. 2024, , 2411390.
29. Zhang, X.; Lei, X.; Jia, X.; et al. Carbon nanotube fibers with dynamic strength up to 14 GPa. Science 2024, 384, 1318-23.
30. Zheng, R.; Peng, Z.; Fu, Y.; et al. A novel conductive core - shell particle based on liquid metal for fabricating real - time self - repairing flexible circuits. Adv. Funct. Mater. 2020, 30, 1910524.
32. Joshi, B.; Samuel, E.; An, S.; Kim, S.; Yarin, A. L.; Yoon, S. S. Review of indium-free, transparent and flexible metallic fibers for wearable electronics. Chem. Eng. J. 2023, 475, 146189.
33. Yang, H.; Zhu, Z.; Ni, S.; et al. Silk fibroin-based bioelectronic devices for high-sensitivity, stable, and prolonged in vivo recording. Biosens. Bioelectron. 2025, 267, 116853.
34. Wang, T. J.; Rethi, L.; Ku, M. Y.; Nguyen, H. T.; Chuang, A. E. A review on revolutionizing ophthalmic therapy: Unveiling the potential of chitosan, hyaluronic acid, cellulose, cyclodextrin, and poloxamer in eye disease treatments. Int. J. Biol. Macromol. 2024, 273, 132700.
35. Chen, Z.; Zhang, Y.; Ding, J.; et al. Hydrogel-based multifunctional deep brain probe for neural sensing, manipulation, and therapy. ACS. Nano. 2025, 19, 21600-13.
36. Sakpal, D.; Gharat, S.; Momin, M. Recent advancements in polymeric nanofibers for ophthalmic drug delivery and ophthalmic tissue engineering. Biomater. Adv. 2022, 141, 213124.
37. Josyula, A.; Mozzer, A.; Szeto, J.; et al. Nanofiber-based glaucoma drainage implant improves surgical outcomes by modulating fibroblast behavior. Bioeng. Transl. Med. 2023, 8, e10487.
38. Paulraj, T.; Wennmalm, S.; Riazanova, A. V.; Wu, Q.; Crespo, G. A.; Svagan, A. J. Porous cellulose nanofiber-based microcapsules for biomolecular sensing. ACS. Appl. Mater. Interfaces. 2018, 10, 41146-54.
39. Song, S.; Zhang, Y.; Xu, D.; et al. Aligned nanofiber-based responsive sponge scaffolds for peripheral nerve regeneration. J. Nanobiotechnology. 2025, 23, 617.
40. Wang, D.; Xing, S.; Peng, F.; et al. Microenvironment-responsive electrocution of tumor and bacteria by implants modified with degenerate semiconductor film. Bioact. Mater. 2023, 20, 472-88.
41. Dinu, M.; Bita, B.; Parau, A. C.; Ristoscu, C.; Negut, I. Bioactive glass and melittin thin films deposited by MAPLE for titanium implant functionalization. Materials 2025, 18, 2410.
42. Lu, S.; Tan, Z.; Kong, S.; Zhang, D. Cost-effective gaze tracking system based on polymer fiber specklegrams. Opt. Lett. 2024, 49, 5027-30.
43. Sharma, V.; Anappara, A. A. Cellulose fiber tortuosity as a bioinspired design strategy for light-driven, self-powered ionotronic synapses. ACS. Appl. Mater. Interfaces. 2025, 17, 65781-92.
44. Lu, C.; Jiang, H.; Cheng, X.; et al. High-performance fibre battery with polymer gel electrolyte. Nature 2024, 629, 86-91.
45. Zhang, J.; Zheng, J.; Gao, M.; Xu, C.; Cheng, Y.; Zhu, M. Nacre-mimetic nanocomposite aerogels with exceptional mechanical performance for thermal superinsulation at extreme conditions. Adv. Mater. 2023, 35, e2300813.
46. Khatib, M.; Zhao, E. T.; Wei, S.; et al. High-density soft bioelectronic fibres for multimodal sensing and stimulation. Nature 2025, 645, 656-64.
47. Yan, W.; Qu, Y.; Gupta, T. D.; et al. Semiconducting nanowire-based optoelectronic fibers. Adv. Mater. 2017, 29, 1-17.
48. Wu, L.; Zhang, J.; Liu, M.; et al. Self-powered eye-tracking system by harvesting the energy of blinking. Cell. Rep. Phys. Sci. 2026, 7, 103026.
49. Li, X.; Wu, D.; Feng, Q.; et al. Flexible bioelectrode via in-situ growth of MOF/enzyme on electrospun nanofibers for stretchable enzymatic biofuel cell. Chem. Eng. J. 2022, 440, 135719.
50. Yao, G.; Mo, X.; Liu, S.; et al. Snowflake-inspired and blink-driven flexible piezoelectric contact lenses for effective corneal injury repair. Nat. Commun. 2023, 14, 3604.
51. Kim, M.; Kwon, Y.; Ahn, Y. Paper-based mediatorless enzymatic microfluidic biofuel cells. Biosens. Bioelectron. 2021, 190, 113391.
52. Pourshaban, E.; Banerjee, A.; Deshpande, A.; et al. Flexible and semi-transparent silicon solar cells as a power supply to smart contact lenses. ACS. Appl. Electron. Mater. 2022, 4, 4016-22.
53. Huang, X.; Li, H.; Li, J.; et al. Transient, implantable, ultrathin biofuel cells enabled by laser-induced graphene and gold nanoparticles composite. Nano. Lett. 2022, 22, 3447-56.
54. Ding, J.; Tian, Y.; Zeng, M.; et al. High performance photoelectric neural electrode with laser-induced ordered reconstruction and tissue-like interface for acute seizure therapy. Chem. Eng. J. 2025, 506, 160263.
55. Song, J.; Gu, Y.; Lin, Z.; et al. Integrating light diffusion and conversion layers for highly efficient multicolored fiber-dye-sensitized solar cells. Adv. Mater. 2024, 36, e2312590.
56. Tohidinejad, Z.; Danyali, S.; Valizadeh, M.; Seepold, R.; TaheriNejad, N.; Haghi, M. Designing a hybrid energy-efficient harvesting system for head- or wrist-worn healthcare wearable devices. Sensors 2024, 24, 1218.
57. Si, J.; Yang, J.; Sun, D.; et al. Breakdown discharge effect enabled self-powered multi-mechanism wireless sensing scheme. Nano. Energy. 2025, 135, 110671.
58. Cui, X.; Wu, L.; Zhang, C.; Li, Z. Implantable self-powered systems for electrical stimulation medical devices. Adv. Sci. 2025, 12, e2412044.
59. Wang, Z.; Yao, S.; Wang, S.; et al. Self-powered energy harvesting and implantable storage system based on hydrogel-enabled all-solid-state supercapacitor and triboelectric nanogenerator. Chem. Eng. J. 2023, 463, 142427.
60. Yang, R.; Zhang, W.; Tiwari, N.; Yan, H.; Li, T.; Cheng, H. Multimodal sensors with decoupled sensing mechanisms. Adv. Sci. 2022, 9, e2202470.
61. He, M.; Du, W.; Feng, Y.; et al. Flexible and stretchable triboelectric nanogenerator fabric for biomechanical energy harvesting and self-powered dual-mode human motion monitoring. Nano. Energy. 2021, 86, 106058.
62. Toan, N.; Sui, H.; Li, J.; et al. Nanoengineered micro-supercapacitors based on graphene nanowalls for self-powered wireless sensing system. J. Energy. Storage. 2024, 81, 110446.
63. Kato, K.; Takaba, K.; Maki-Yonekura, S.; et al. Double-helix supramolecular nanofibers assembled from negatively curved nanographenes. J. Am. Chem. Soc. 2021, 143, 5465-9.
64. Shen, Y.; Wang, Z.; Wang, Z.; et al. Thermally drawn multifunctional fibers: toward the next generation of information technology. InfoMat 2022, 4, e12318.
65. Dong, C.; Leber, A.; Yan, D.; et al. 3D stretchable and self-encapsulated multimaterial triboelectric fibers. Sci. Adv. 2022, 8, eabo0869.
66. Xu, H.; Han, L.; Huang, J.; Du, B.; Zhan, D. Scanning electrochemical probe lithography for ultra-precision machining of micro-optical elements with freeform curved surface. Small 2024, 20, e2402743.
67. Hua, J. G.; Liang, S. Y.; Chen, Q. D.; et al. Free‐form micro - optics out of crystals: femtosecond laser 3D sculpturing. Adv. Funct. Mater. 2022, 32, 2200255.
68. Farokhi, M.; Mottaghitalab, F.; Reis, R. L.; Ramakrishna, S.; Kundu, S. C. Functionalized silk fibroin nanofibers as drug carriers: advantages and challenges. J. Control. Release. 2020, 321, 324-47.
69. Lei, M.; Ji, Y.; Zhang, S.; et al. Engineering a functionality-integrated artificial cornea stromal Substitute: Janus bio-adhesive implant with a collagen-based multi-scale biomimetic skeleton. Bioact. Mater. 2025, 51, 740-57.
70. Albarqi, H. A.; Garg, A.; Ahmad, M. Z.; Alqahtani, A. A.; Walbi, I. A.; Ahmad, J. Recent progress in chitosan-based nanomedicine for its ocular application in glaucoma. Pharmaceutics 2023, 15, 681.
71. Guo, H.; Lee, C.; Shah, M.; et al. A novel elastin-like polypeptide drug carrier for cyclosporine A improves tear flow in a mouse model of Sjögren’s syndrome. J. Control. Release. 2018, 292, 183-95.
72. Ahmadi, Z.; Yadav, S.; Kar, A. K.; et al. An injectable self-assembling hydrogel based on RGD peptidomimetic β-sheets as multifunctional biomaterials. Biomater. Adv. 2022, 133, 112633.
73. Zhang, R.; Liu, C.; Li, T.; et al. PEDOT:PSS conductive networks encapsulated within a thermally responsive VO2 nanoparticle/cellulose nanofiber matrix for electrothermally triggered dual-mode EMI shielding and smart electromagnetic management. ACS. Appl. Nano. Mater. 2025, 8, 14387-98.
74. Li, Y.; Cao, W.; Liu, Z.; et al. A personalized electronic textile for ultrasensitive pressure sensing enabled by biocompatible MXene/PEDOT:PSS composite. Carbon. Energy. 2024, 6, e530.
75. Cao, Y.; Zhang, C.; Shen, W.; Cheng, Z.; Yu, L. L.; Ping, Q. Poly(N-isopropylacrylamide)-chitosan as thermosensitive in situ gel-forming system for ocular drug delivery. J. Control. Release. 2007, 120, 186-94.
76. Ren, H.; Xu, J.; Lai, Y.; et al. pH sensitive loading and release of doxorubicin by chitosan-graphene quantum dots hybridized material. Int. J. Biol. Macromol. 2025, 319, 145375.
77. Liu, Y.; Yue, S.; Tian, Z.; et al. Self-powered and self-healable extraocular-muscle-like actuator based on dielectric elastomer actuator and triboelectric nanogenerator. Adv. Mater. 2024, 36, e2309893.
78. Wang, D.; Yang, A. Miniaturized optics from structured nanoscale cavities. Prog. Quant. Electron. 2024, 94, 100507.
79. Wang, S.; Fan, P.; Liu, W.; et al. Research progress of flexible electronic devices based on electrospun nanofibers. ACS. Nano. 2024, 18, 31737-72.
80. Rokade, K. A.; Kumbhar, D. D.; Patil, S. L.; et al. Cognifiber: harnessing biocompatible and biodegradable 1D collagen nanofibers for sustainable nonvolatile memory and synaptic learning applications. Adv. Mater. 2024, 36, e2312484.
81. Yan, L.; Zhao, B.; Liu, X.; et al. Aligned nanofibers from polypyrrole/graphene as electrodes for regeneration of optic nerve via electrical stimulation. ACS. Appl. Mater. Interfaces. 2016, 8, 6834-40.
82. Stahnke, T.; Siewert, S.; Reske, T.; et al. Development of a biodegradable antifibrotic local drug delivery system for glaucoma microstents. Biosci. Rep. 2018, 38, BSR20180628.
83. Khrystonko, O.; Rimpelová, S.; Burianová, T.; Švorčík, V.; Lyutakov, O.; Elashnikov, R. Smart multi stimuli-responsive electrospun nanofibers for on-demand drug release. J. Colloid. Interface. Sci. 2023, 648, 338-47.
84. Zhang, Y.; Hu, Y.; Liu, Q.; et al. Multiplexed optical fiber sensors for dynamic brain monitoring. Matter 2022, 5, 3947-76.
85. Zhang, J.; Kim, K.; Kim, H. J.; et al. Smart soft contact lenses for continuous 24-hour monitoring of intraocular pressure in glaucoma care. Nat. Commun. 2022, 13, 5518.
86. Seo, H.; Hong, Y. M.; Chung, W. G.; et al. Real-time in vivo monitoring of intraocular pressure distribution in the anterior chamber and vitreous chamber for diagnosis of glaucoma. Sci. Adv. 2024, 10, eadk7805.
87. Zhu, H.; Yang, H.; Zhan, L.; Chen, Y.; Wang, J.; Xu, F. Hydrogel-based smart contact lens for highly sensitive wireless intraocular pressure monitoring. ACS. Sens. 2022, 7, 3014-22.
88. Shean, R.; Yu, N.; Guntipally, S.; et al. Advances and challenges in wearable glaucoma diagnostics and therapeutics. Bioengineering 2024, 11, 138.
89. Kim, T. Y.; Mok, J. W.; Hong, S. H.; et al. Wireless theranostic smart contact lens for monitoring and control of intraocular pressure in glaucoma. Nat. Commun. 2022, 13, 6801.
90. Yang, H.; Zhu, H.; Liu, H.; et al. Intraocular pressure monitoring smart contact lens with high environmental stability. Adv. Funct. Mater. 2024, 34, 2400722.
91. Su, Y.; Chen, C.; Pan, H.; Yang, Y. Muscle fibers inspired high - performance piezoelectric textiles for wearable physiological monitoring. Adv. Funct. Mater. 2021, 31, 2010962.
92. Li, T.; Yuan, Y.; Gu, L.; et al. Ultrastable piezoelectric biomaterial nanofibers and fabrics as an implantable and conformal electromechanical sensor patch. Sci. Adv. 2024, 10, eadn8706.
93. Guo, W.; Tan, C.; Shi, K.; et al. Wireless piezoelectric devices based on electrospun PVDF/BaTiO3 NW nanocomposite fibers for human motion monitoring. Nanoscale 2018, 10, 17751-60.
94. Li, X.; Chen, W.; Li, H.; et al. Temperature self-compensating intelligent wireless measuring contact lens for quantitative intraocular pressure monitoring. ACS. Appl. Mater. Interfaces. 2024, 16, 22522-31.
95. Park, J.; Kim, J.; Kim, S. Y.; et al. Soft, smart contact lenses with integrations of wireless circuits, glucose sensors, and displays. Sci. Adv. 2018, 4, eaap9841.
96. Shi, Y.; Wang, L.; Hu, Y.; et al. Contact lens sensor for ocular inflammation monitoring. Biosens. Bioelectron. 2024, 249, 116003.
97. Liu, Y.; Su, X.; Fan, P.; Liu, X.; Pan, Y.; Ping, J. Computationally-assisted wearable system for continuous cortisol monitoring. Sci. Bull. 2025, 70, 2004-13.
98. Wang, S.; Jiang, C.; Yu, Y.; et al. Tellurium nanowire retinal nanoprosthesis improves vision in models of blindness. Science 2025, 388, eadu2987.
99. Jiang, L.; Lu, G.; Zeng, Y.; et al. Flexible ultrasound-induced retinal stimulating piezo-arrays for biomimetic visual prostheses. Nat. Commun. 2022, 13, 3853.
100. Kilias, A.; Canales, A.; Froriep, U. P.; Park, S.; Egert, U.; Anikeeva, P. Optogenetic entrainment of neural oscillations with hybrid fiber probes. J. Neural. Eng. 2018, 15, 056006.
101. Patel, M.; Meenu, M.; Pandey, J. K.; et al. Recent development in upconversion nanoparticles and their application in optogenetics: a review. J. Rare. Earth. 2022, 40, 847-61.
102. Wu, C.; Su, B.; Xin, N.; et al. An upconversion nanoparticle-integrated fibrillar scaffold combined with a NIR-optogenetic strategy to regulate neural cell performance. J. Mater. Chem. B. 2023, 11, 430-40.
103. Tian, F.; Zhang, Y.; Schriver, K. E.; Hu, J. M.; Roe, A. W. A novel interface for cortical columnar neuromodulation with multipoint infrared neural stimulation. Nat. Commun. 2024, 15, 6528.
104. Pakshir, P.; Younesi, F.; Wootton, K. A.; et al. Controlled release of low-molecular weight, polymer-free corticosteroid coatings suppresses fibrotic encapsulation of implanted medical devices. Biomaterials 2022, 286, 121586.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data






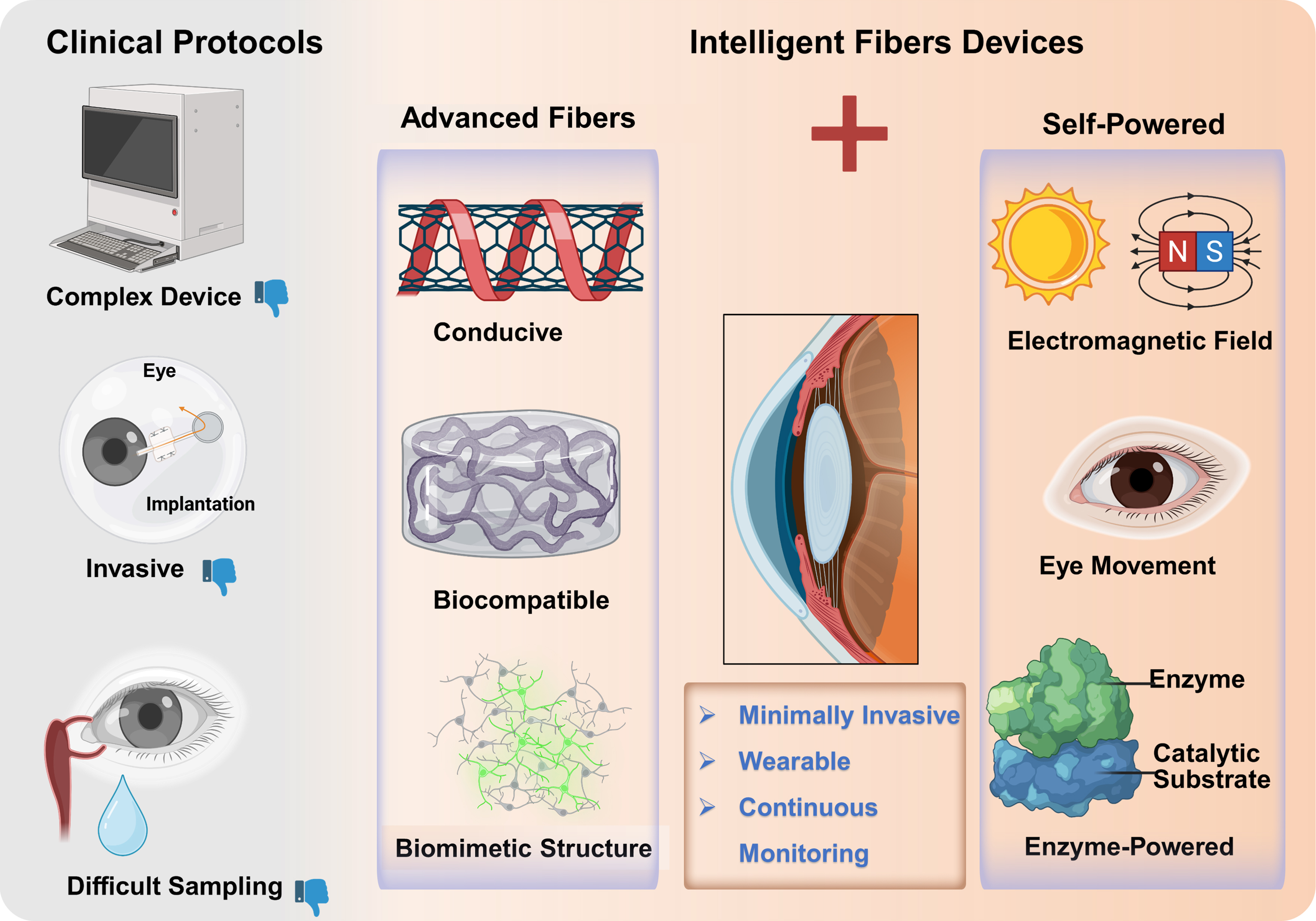
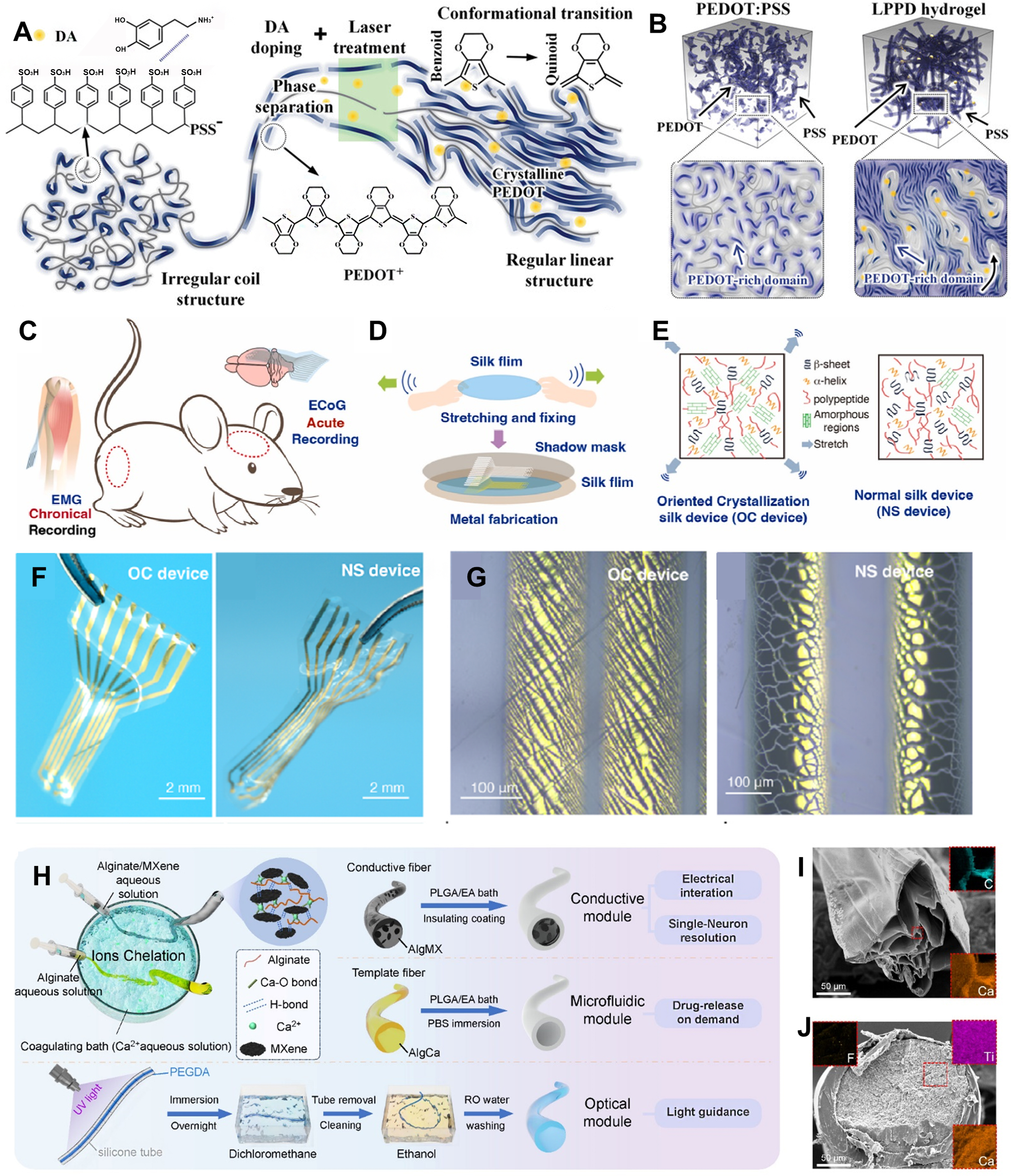
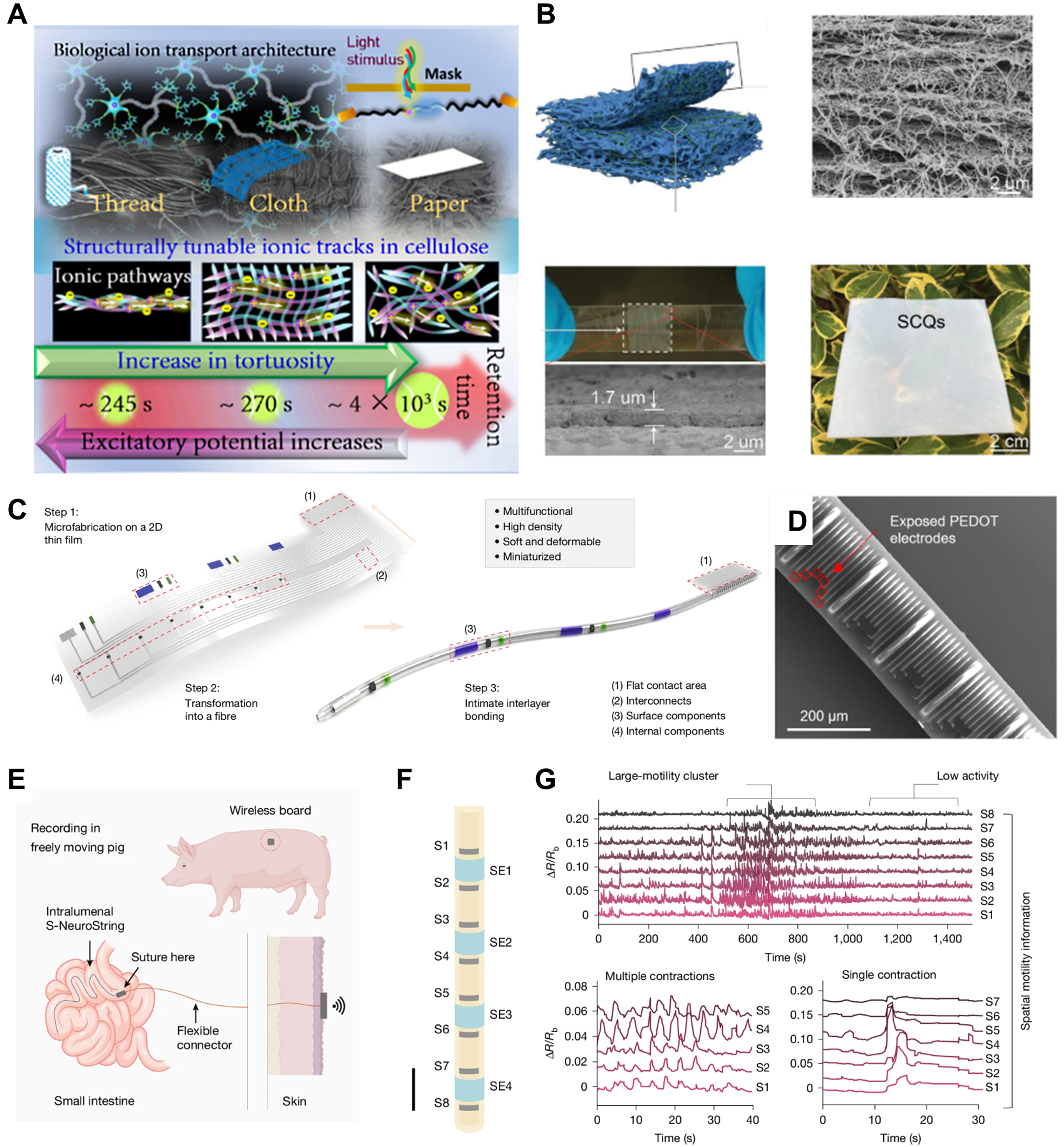
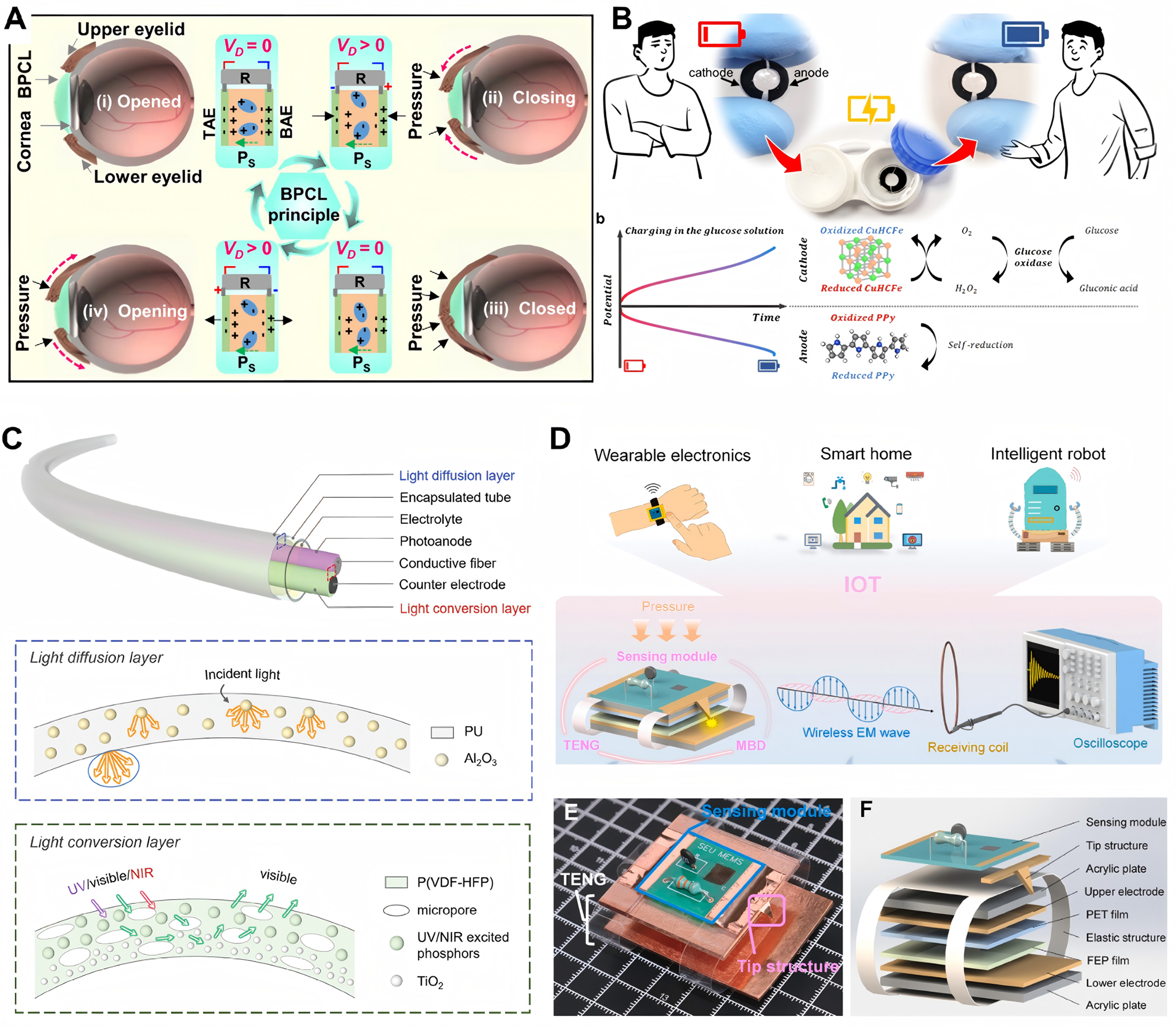
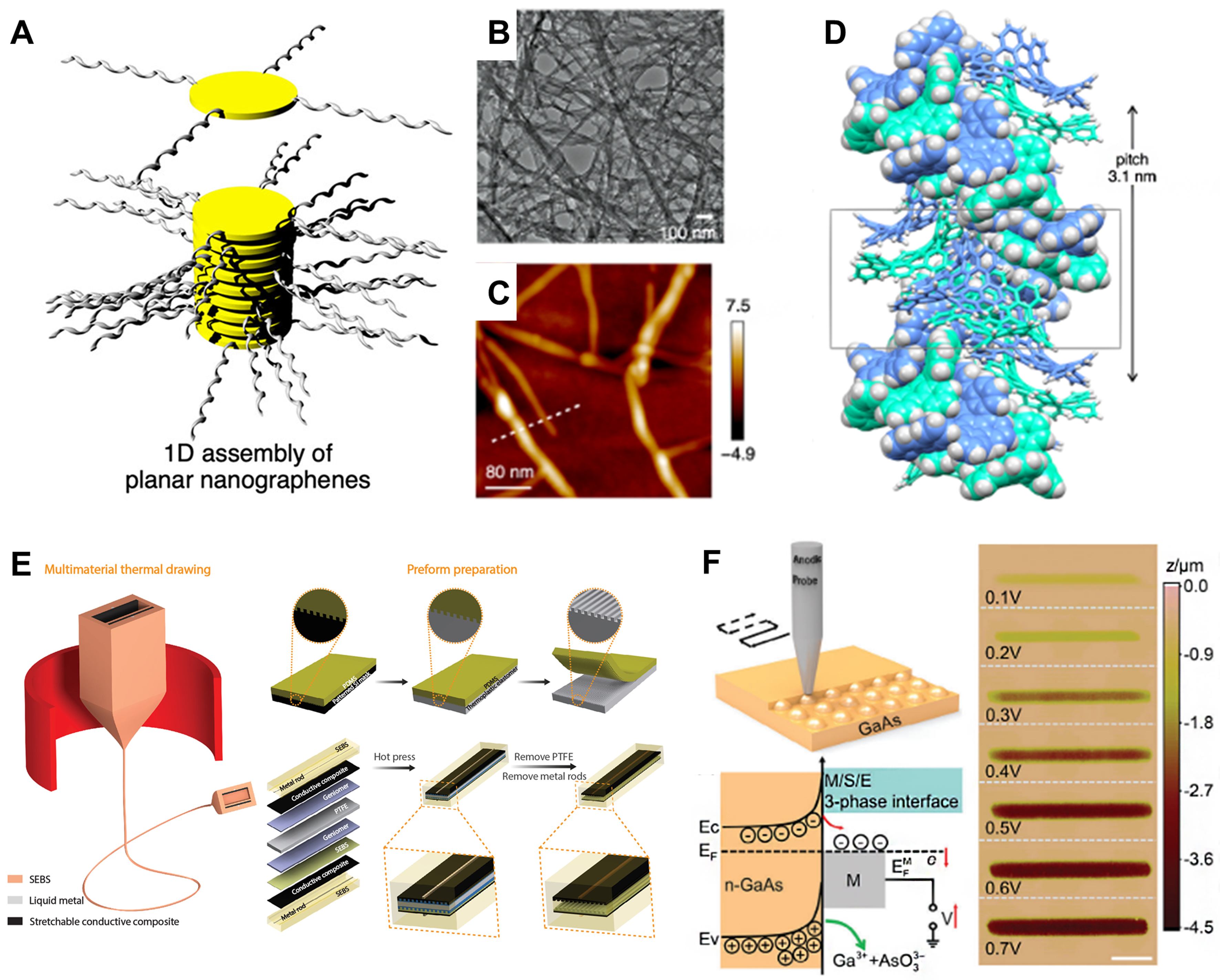
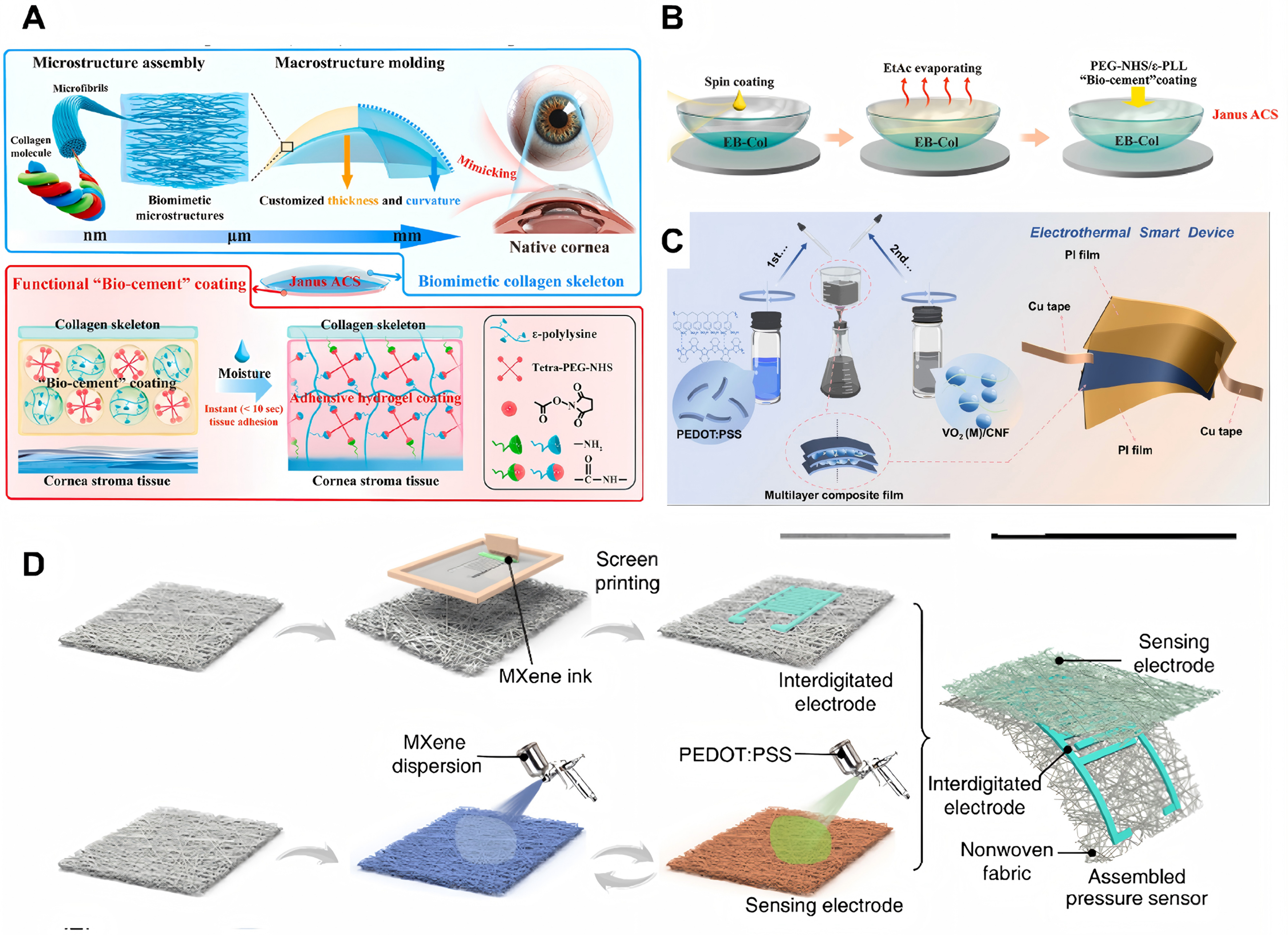
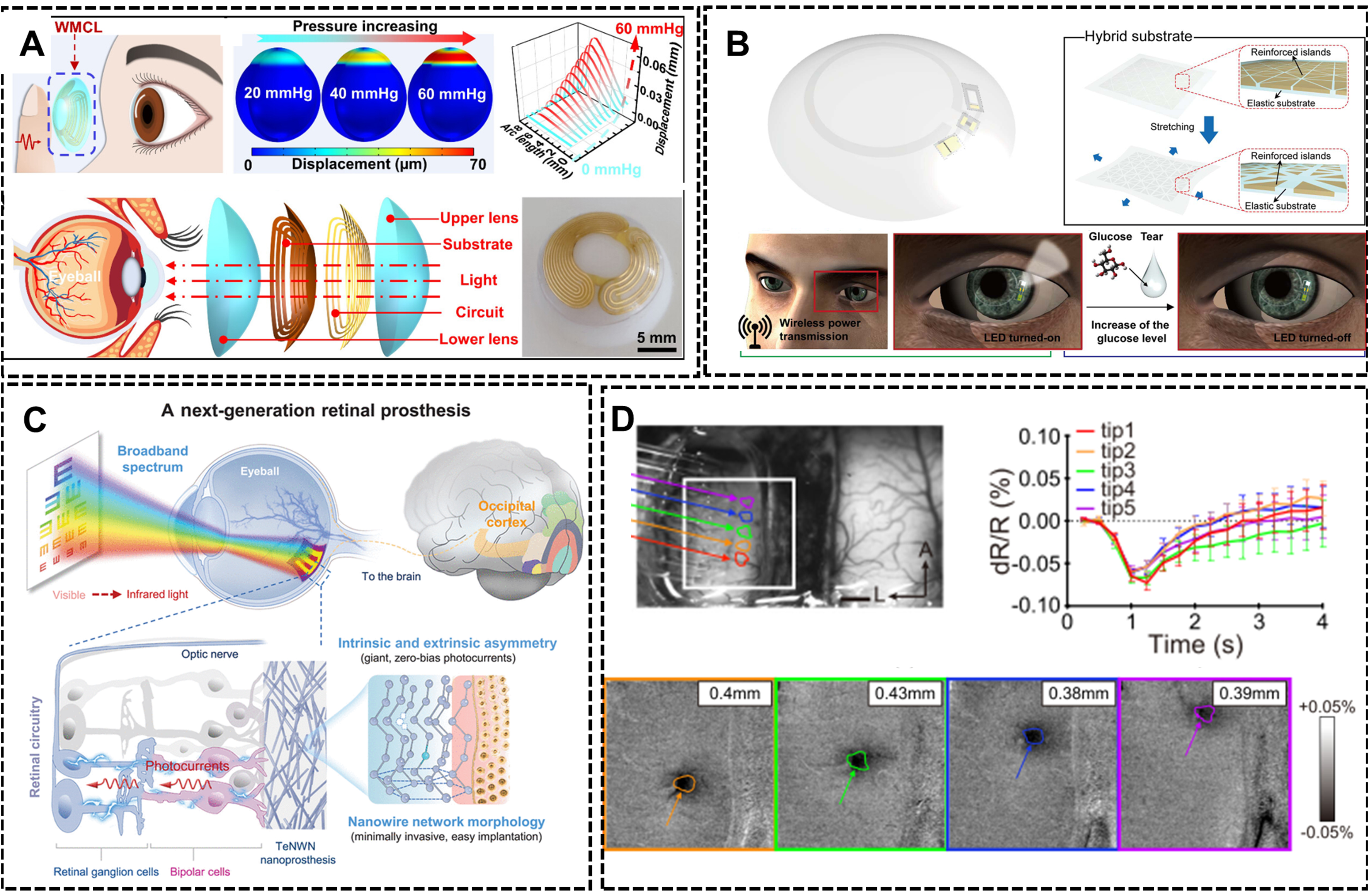












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.